Introduction
The global rise of antibiotic-resistant bacteria has emerged as a major public health concern. To address this issue, the World Health Organization (WHO) published its bacterial priority pathogens list in 2017 and updated it in 2024. In the most recent classification, carbapenem-resistant Acinetobacter baumannii and Enterobacterales are designated as critical priority pathogens, while carbapenem-resistant Pseudomonas aeruginosa (CR-PA) is classified as high priority; collectively, these pathogens require the urgent development of novel antibiotics (1,2).
During the COVID-19 pandemic, the crisis of resistant bacterial infections deepened as antibiotic resistance increased and resistance to the “last-resort” antibiotic colistin (COL) became more widespread (3). Encouraged by the Infectious Diseases Society of America (IDSA) and other major organizations, several small and medium-sized enterprises, along with a limited number of pharmaceutical companies, have begun investing in the discovery of novel agents active against multidrug-resistant (MDR) Gram-negative bacteria. As a result of these efforts, several novel antibacterial agents have received approval from the U.S. Food and Drug Administration (FDA) (1).
Ceftolozane-tazobactam (TOL-TAZ) and ceftazidime-avibactam (CAZ-AVI) are two recently FDA-approved antibiotics with activity against Gram-negative bacteria. These agents combine cephalosporins with β-lactamase inhibitors to overcome resistance mechanisms, thereby expanding available therapeutic options (4).
Carbapenems have traditionally been among the most effective agents against infections caused by resistant Gram-negative bacteria, but the increasing prevalence of carbapenemase-mediated resistance has driven the development of alternative therapies. Meropenem-vaborbactam (MER-VAB), a combination of the carbapenem meropenem and vaborbactam, a non-β-lactam β-lactamase inhibitor, has emerged as a promising option for the treatment of challenging infections caused by carbapenem-resistant Enterobacterales (3).
Eravacycline (ERV), a synthetic fluorocycline derivative of the tetracycline class, has been developed as an innovative agent expected to provide a therapeutic alternative for resistant infections (5). It has demonstrated in vitro activity against Gram-negative pathogens, including carbapenem-resistant Enterobacterales, carbapenem-resistant A. baumannii, and Stenotrophomonas maltophilia, as well as Gram-positive pathogens (6). Although clinical evidence remains limited for most of these agents, several features support ERV as a non-β-lactam alternative for the treatment of resistant bacterial infections, including broad-spectrum activity and efficacy in MDR scenarios (5–7).
Delafloxacin (DLX) is a newly developed fluoroquinolone with activity against both Gram-positive and Gram-negative pathogens, demonstrating broad-spectrum efficacy, particularly against cystic fibrosis (CF) pathogens such as MDR P. aeruginosa (8,9). It exhibits excellent pulmonary and biofilm penetration and maintains enhanced activity under acidic conditions (8). It is currently being investigated as a therapeutic option for various bacterial infections (9).
In this study, the resistance profiles of Klebsiella pneumoniae, A. baumannii, and P. aeruginosa isolates obtained from patients admitted to the intensive care unit (ICU) were evaluated against ceftolozane/tazobactam, CAZ-AVI, MER-VAB, ERV, and DLX. While some of these antibiotics have recently been introduced into clinical use in our country, others have not yet become available. This study aimed to determine the resistance rates to these agents and thereby contribute to the optimization of therapeutic options and the development of evidence-based health policies.
Materials and Methods
The study protocol was approved by the Non-Interventional Clinical Research Ethics Committee of Uşak University Faculty of Medicine on May 15, 2025, with decision number 678-678-26. The study was conducted in accordance with the principles of the Declaration of Helsinki. This retrospective study included non-duplicate clinical isolates of K. pneumoniae, A. baumannii, and P. aeruginosa obtained from specimens collected from ICUs and submitted to the Medical Microbiology Laboratory of Uşak Training and Research Hospital between January 1, 2023, and December 31, 2024. To ensure that the analysis represented unique patients rather than repeated sampling, only the first isolate per patient per species was included. Subsequent isolates from the same patient showing the same identification and susceptibility profile, as well as surveillance cultures (e.g., rectal swabs), were excluded from the analysis.
Identification and Antimicrobial Susceptibility Testing
Bacterial isolates obtained from ICU patients were identified using the VITEK® 2 automated system (bioMérieux, Marcy-l’Étoile, France) and the VITEK® MS (bioMérieux, Marcy-l’Étoile, France). Susceptibility testing for TOL-TAZ, CAZ-AVI, MER-VAB, ERV, and DLX was performed on isolates identified as K. pneumoniae, A. baumannii, and P. aeruginosa using the VITEK® 2 automated system (bioMérieux, Marcy-l’Étoile, France) with the XN21 card. Colistin susceptibility was determined by broth microdilution. Minimum inhibitory concentration (MIC) values and susceptibility categories for the tested antibiotics were interpreted according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines (10). Since specific EUCAST breakpoints are currently undefined for ERV and DLX for certain species, susceptibility for these agents was interpreted based on the U.S. Food and Drug Administration (FDA) breakpoints to ensure accurate categorization. Quality control was performed using standard reference strains, including Escherichia coli ATCC 25922, P. aeruginosa ATCC 27853, and Enterobacter hormaechei ATCC 700323, in accordance with EUCAST recommendations.
Statistical Analysis
Data analysis was performed using IBM SPSS Statistics for Windows, version 27.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were presented as counts (n) and percentages (%) for categorical variables. Antibiotic resistance rates were determined for each bacterial species, and their distributions were evaluated across the study years (2023–2024). Differences in resistance rates between years were assessed using the Pearson chi-square test. A p-value of <0.05 was considered statistically significant. The distributions of MIC values obtained from antimicrobial susceptibility testing were graphically visualized using Microsoft Excel 2019 (Microsoft Corp., Redmond, WA, USA).
Results
Demographic and Clinical Characteristics
Between January 1, 2023, and December 31, 2024, a total of 1525 clinical specimens were submitted from ICU patients to the Medical Microbiology Laboratory of the study center. Of these specimens, 1007 (66.0%) were obtained from respiratory tract samples, 321 (21.0%) from blood, 168 (11.0%) from urine, and 29 (1.9%) from other specimen types. Regarding the distribution by ICU type, 537 samples (35.2%) originated from the General ICU, 495 (32.5%) from the Neurology ICU, 204 (13.4%) from the Anesthesiology and Reanimation ICU, 188 (12.3%) from the Chest Diseases ICU, and 101 (6.6%) from other ICUs. Microbiological cultures yielded 850 isolates of K. pneumoniae, 219 isolates of P. aeruginosa, and 456 isolates of A. baumannii.
Klebsiella pneumoniae Isolates
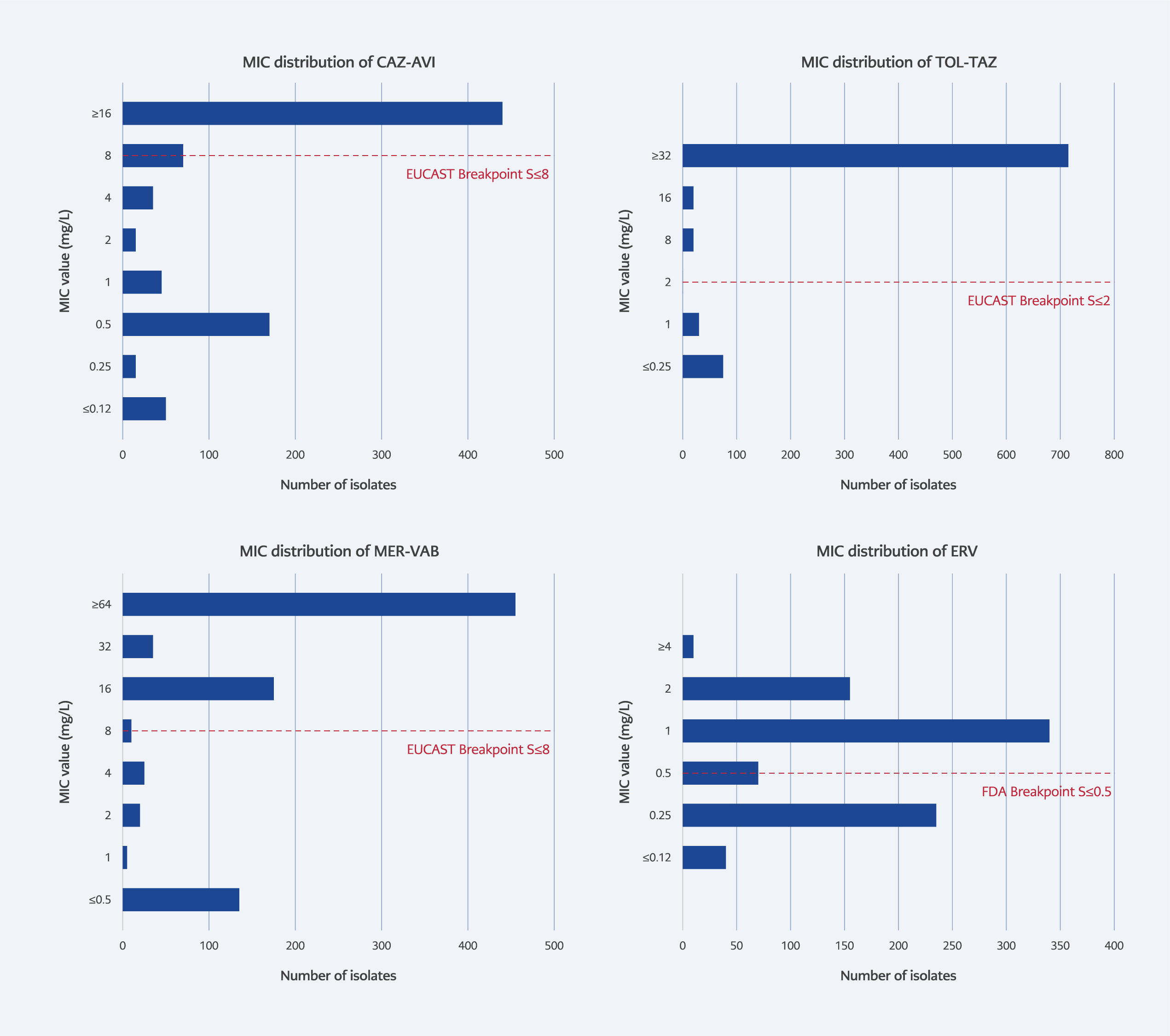
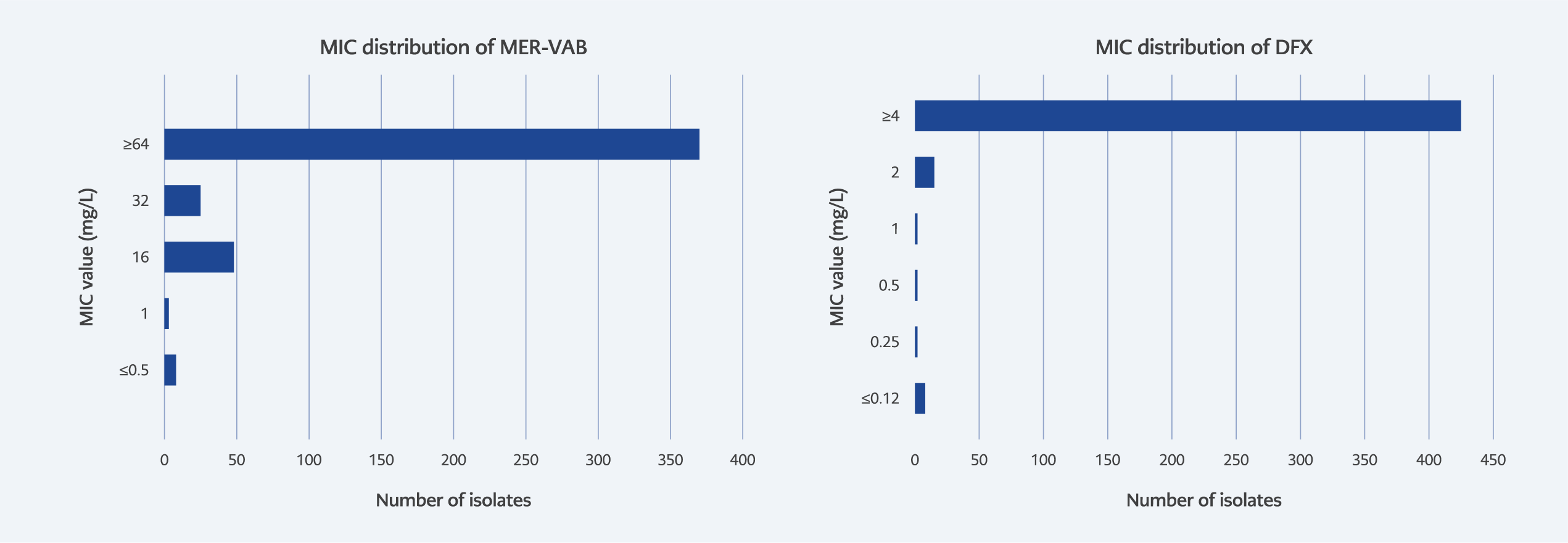
Among K. pneumoniae isolates, resistance rates were 52.2% for CAZ-AVI, 87.6% for TOL-TAZ, and 78.0% for MER-VAB. Since EUCAST guidelines do not currently define breakpoints for ERV and DLX for Enterobacterales (10), FDA breakpoints were applied (11). Accordingly, 40.6% of isolates were classified as susceptible to ERV (≤0.5 µg/mL). The MIC distribution of these novel antibiotics against K. pneumoniae isolates is presented in Figure 1.
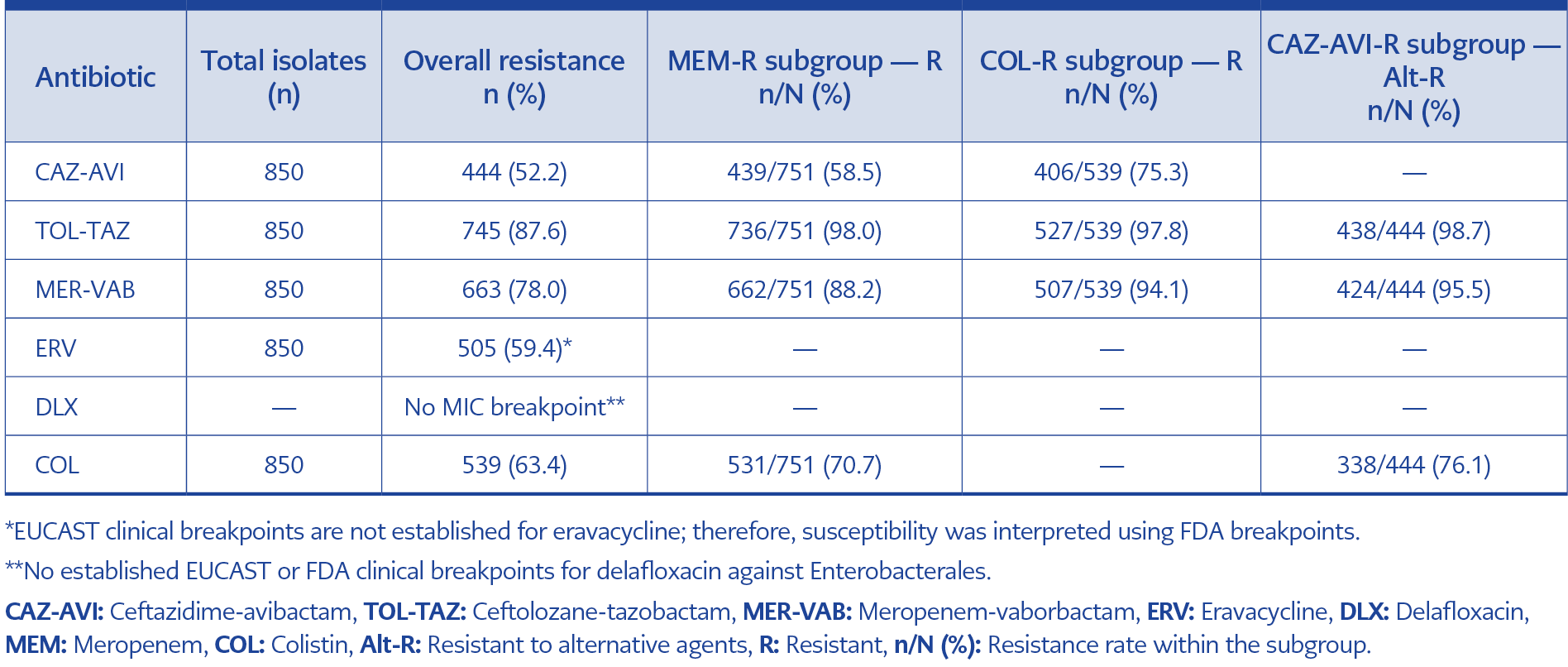
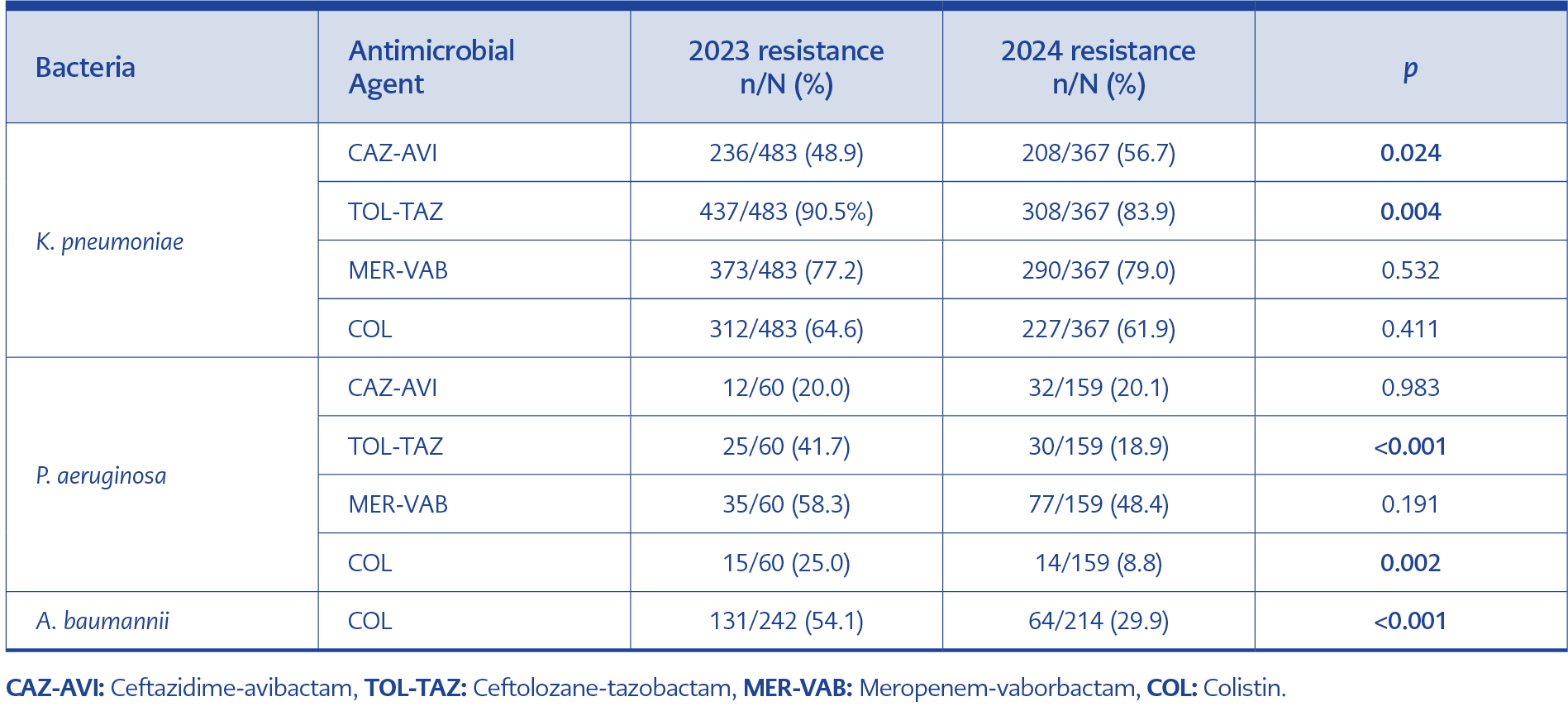
In the analysis of cross-resistance patterns, CAZ-AVI displayed the highest in vitro activity among meropenem-resistant isolates (41.5% susceptible), followed by COL (29.3%) (Table 1). Among COL-resistant isolates, 24.7% remained susceptible to CAZ-AVI. However, therapeutic options were severely limited for CAZ-AVI-resistant isolates, with susceptibility rates of only 4.5% for MER-VAB and 1.4% for TOL-TAZ. A comparison of resistance rates between years revealed a significant increase in CAZ-AVI resistance (p=0.024) and a significant decrease in TOL-TAZ resistance (p=0.004) in 2024 compared to 2023 (Table 3).
Pseudomonas aeruginosa Isolates
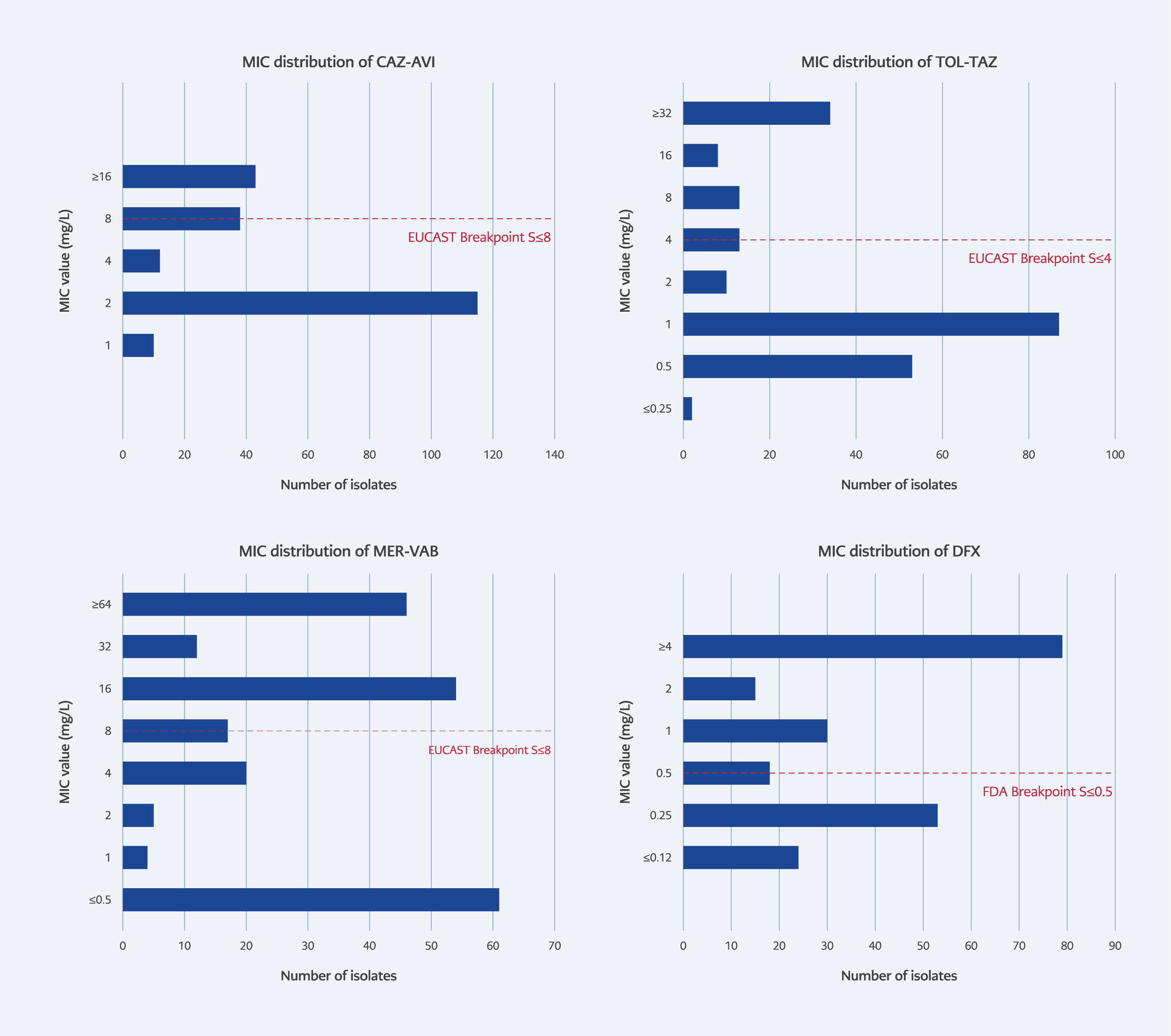
Resistance rates for P. aeruginosa were 20.1% for CAZ-AVI, 25.1% for TOL-TAZ, and 51.1% for MER-VAB. The MIC distribution of the novel antibiotics against P. aeruginosa isolates is shown in Figure 2.
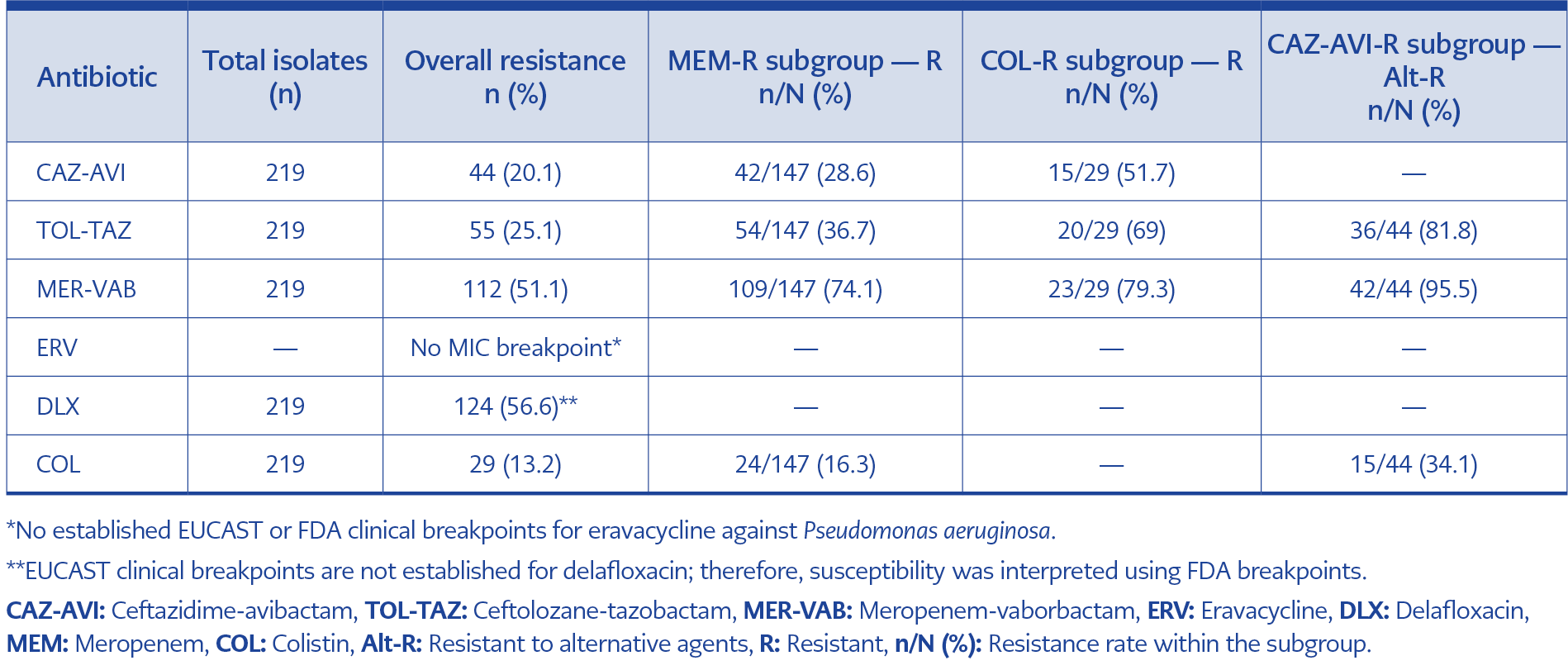
Among meropenem-resistant P. aeruginosa isolates, COL (83.7%) and CAZ-AVI (71.4%) exhibited high susceptibility rates (Table 2). For COL-resistant isolates, nearly half (48.3%) remained susceptible to CAZ-AVI. In contrast to K. pneumoniae, COL retained substantial activity (65.9%) against CAZ-AVI-resistant P. aeruginosa strains. Resistance to COL and TOL-TAZ decreased significantly in 2024 compared to 2023 (p=0.002 and p<0.001, respectively) (Table 3).
Acinetobacter baumannii Isolates
For A. baumannii isolates (n=456), no clinical breakpoints are currently available in the EUCAST guidelines for CAZ-AVI, TOL-TAZ, MER-VAB, DLX, or ERV (10). Meropenem resistance was observed in 97.6% of the isolates, while COL resistance was detected in 42.8%. The MIC distributions of the novel antibiotics in A. baumannii isolates are presented in Figure 3. A significant decrease in COL resistance was observed in 2024 compared to 2023 (p<0.001) (Table 3).
Discussion
The rapid global rise in antibiotic resistance has led to an urgent need for novel antimicrobial agents effective against carbapenem-resistant Gram-negative bacteria. In this context, novel antibiotics such as CAZ-AVI, TOL-TAZ, MER-VAB, ERV, and DLX offer promising therapeutic options against MDR isolates. However, the development of optimal treatment strategies for these agents should rely not only on their pharmacological properties but also on up-to-date local resistance data. Studies assessing resistance rates to these novel antibiotics in Türkiye remain limited.
For the treatment of infections caused by carbapenem-resistant Enterobacterales strains, agents such as CAZ-AVI, MER-VAB, imipenem-relebactam (IMI-REL), cefiderocol, and ERV are recommended (1). Among these, CAZ-AVI was introduced for clinical use in Türkiye in October 2019 (12). Global surveillance programs have provided critical benchmarks for these agents. In the SENTRY surveillance program, susceptibility rates for P. aeruginosa were reported as 93.9% for CAZ-AVI and 96.8% for TOL-TAZ. However, a geographic gradient was observed, with susceptibility rates declining to 82.9% in Eastern Europe. Similarly, for K. pneumoniae, CAZ-AVI susceptibility was found to be 100% in the USA and 99.2% in Western Europe, whereas this rate dropped to 92% in Eastern European countries (13). Furthermore, in a systematic review and meta-analysis by Wang et al. (14), which analyzed data from the global Study of Monitoring Antimicrobial Resistance Trends (SMART) and other studies, CAZ-AVI resistance rates were reported as 10.2% for K. pneumoniae, 86.8% for A. baumannii, and 22.8% for P. aeruginosa.
In studies conducted in Türkiye, Bilgin et al. (15) reported CAZ-AVI resistance rates of 7.5% among extended-spectrum β-lactamase (ESBL)-producing Enterobacterales isolates and 12.8% among carbapenem-resistant isolates collected between 2021 and 2022. Şenol et al. (16) found a resistance rate of 16.3% among 37 K. pneumoniae strains isolated from ICU patients in the first half of 2021, while a contemporaneous study by Arıcı et al. (12) reported a higher rate of 22.2% for K. pneumoniae. Another study evaluating year-to-year changes in antibiotic susceptibility among K. pneumoniae isolates from adult ICU blood cultures found that all isolates tested in 2021 were susceptible to CAZ-AVI. However, susceptibility rates declined to 93% in 2022, 87% in 2023, and 77% by 2024 (17). In addition to the temporal increase in resistance, reports of regionally high resistance rates have raised concerns regarding the use of CAZ-AVI. For example, Akbaş et al. (18), in a study conducted at Düzce University Faculty of Medicine, reported CAZ-AVI susceptibility rates of 44.6% among Enterobacterales isolates and 24.2% among P. aeruginosa isolates. In a comprehensive study at Dicle University, the susceptibility rate of CAZ-AVI among carbapenem-resistant K. pneumoniae isolates was reported as 57% (19).
In our study, CAZ-AVI susceptibility was 47.8% for K. pneumoniae isolates and 79.9% for P. aeruginosa isolates. Year-to-year comparison revealed a significant increase in resistance among K. pneumoniae isolates, from 48.9% in 2023 to 56.7% in 2024 (p=0.024). This significant temporal increase in CAZ-AVI resistance within just two years is clinically alarming. These findings may suggest that the introduction of this agent into clinical practice in our hospital could have created a selective pressure, accelerating the proliferation of resistant clones. Clinically, this rising trend threatens the utility of CAZ-AVI as a reliable empirical rescue therapy in our ICUs, necessitating stricter antimicrobial stewardship policies.
The high level of CAZ-AVI resistance observed in ICU isolates, particularly K. pneumoniae, suggests a potential association with the selection and dissemination of specific resistant clones within the study center. A previous study conducted in our hospital reported the spread of the ST395 K. pneumoniae clone, resistant to both carbapenems and COL, over an 18-month period (20). Although molecular characterization was not the primary scope of the current study, recent preliminary observations in our laboratory indicate the emergence of K. pneumoniae ST147 clones co-harboring New Delhi metallo-β-lactamase-5 (NDM-5) and oxacillinase-48 (OXA-48) carbapenemases (data not shown). Considering that CAZ-AVI was first introduced at our center in 2023, it is possible that selective antibiotic pressure may have favored the expansion of such high-risk clones, contributing to the elevated resistance rates observed. Further molecular surveillance is warranted to confirm these dynamics.
Ceftolozane-tazobactam is a novel antipseudomonal cephalosporin with a classical β-lactamase inhibitor. While TOL-TAZ retains activity against Enterobacterales strains producing AmpC β-lactamase (AmpC) and ESBLs, its efficacy remains limited against ESBL-producing K. pneumoniae (21). It is considered a promising agent for the treatment of CR-PA isolates. In the ERACE-PA Global Surveillance Program, susceptibility among carbapenemase-negative CR-PA isolates was reported as 88%, underscoring the potent activity of TOL-TAZ against carbapenem-resistant strains lacking carbapenemase production (22). However, its efficacy is significantly reduced in carbapenemase-producing isolates, underscoring the importance of accounting for molecular resistance mechanisms in clinical practice.
Given the increasing prevalence of CR-PA in Türkiye, TOL-TAZ may represent a valuable addition to treatment algorithms. Nonetheless, careful patient selection based on carbapenemase status and close monitoring of national resistance trends are essential before widespread implementation. In a study by Bilgin et al. (23), 49 of 52 ceftazidime-resistant P. aeruginosa isolates (94.2%) were susceptible to CAZ-AVI, while all but one (98.1%) were susceptible to TOL-TAZ. Furthermore, MIC50 values indicated that TOL-TAZ was twice as potent as CAZ-AVI. Another study conducted in Tokat reported a TOL-TAZ resistance rate of 8.2% among P. aeruginosa isolates according to EUCAST breakpoints (24). In our study, the overall resistance rate to TOL-TAZ among P. aeruginosa isolates was 25.1%, with a marked decrease from 41.7% in 2023 to 18.9% in 2024. This fluctuation underscores the dynamic nature of resistance in ICU settings, where local outbreaks can drastically alter susceptibility patterns within a short timeframe. The significant reduction in resistance suggests that the previous high rates might have been driven by a transient clonal outbreak that has since subsided, potentially restoring the clinical utility of TOL-TAZ for P. aeruginosa infections in our center. In contrast, resistance to TOL-TAZ among K. pneumoniae isolates was considerably higher, at 87.6%. In a study by Guzel et al. (25), susceptibility to CAZ-AVI among carbapenem-resistant Enterobacterales was 77%, whereas only 48% were susceptible to TOL-TAZ. Similarly, Terzi et al. (26) reported that 95% of carbapenemase-producing K. pneumoniae isolates were resistant to TOL-TAZ.
Meropenem-vaborbactam is designed to target Enterobacterales strains producing K. pneumoniae carbapenemase-type carbapenemases (3). It has demonstrated high efficacy in the treatment of infections caused by carbapenem-resistant K. pneumoniae isolates (27). However, vaborbactam is inactive against class B carbapenemases (e.g., NDM, imipenemase [IMP], Verona integron-encoded metallo-β-lactamase [VIM]) and class D carbapenemases, particularly OXA-48 (3), which substantially limits its clinical utility in regions where these enzymes are prevalent.
In our study, resistance to MER-VAB among K. pneumoniae isolates was high, at 78%. Within the meropenem-resistant subgroup, susceptibility declined to 11.8% and further decreased to 5.9% among COL-resistant isolates. Resistance to MER-VAB was also observed in 51.1% of P. aeruginosa isolates, with a susceptibility rate of 25.9% in the meropenem-resistant subgroup.
Consistent with these findings, Aydemir et al. (28) reported MER-VAB susceptibility in 60% of 30 carbapenemase-producing K. pneumoniae isolates. In a comprehensive study at Başakşehir Çam and Sakura Hospital, MER-VAB resistance was observed in 91% of carbapenem-resistant K. pneumoniae strains isolated from ICUs, with molecular analysis revealing that the majority (70 of 80) carried the OXA-48 gene, while the remaining isolates, except for one K. pneumoniae carbapenemase-positive strain, harbored the NDM gene (29). These findings, together with our own data, reflect the predominance of OXA-48 and NDM carbapenemases in Türkiye and indicate that the clinical efficacy of MER-VAB is substantially limited against these strains.
Eravacycline is a novel, fully synthetic tetracycline derivative developed for the treatment of complicated intra-abdominal infections. It demonstrates broad-spectrum activity against resistant Gram-negative pathogens, including ESBL-producing Enterobacterales and Acinetobacter spp., as well as methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci (VRE) (5).
In a study evaluating ERV susceptibility among carbapenem-resistant Enterobacterales, using the FDA breakpoint of ≤0.5 µg/mL for Enterobacterales as a reference (11), 54 of 80 isolates (67.5%) were reported as susceptible (30). In another study conducted in Taiwan on 1000 Enterobacterales isolates, the overall susceptibility rate to ERV was 67.7%, which decreased to 47.1% among the meropenem-resistant subpopulation (31). It should be noted that the EUCAST guidelines provide breakpoints only for E. coli, with no established values for other Enterobacterales species. Applying the FDA breakpoint for Enterobacterales to the 850 K. pneumoniae isolates included in our study, 345 isolates (40.6%) were found to be susceptible to ERV. Considering that the majority of our isolates were carbapenem-resistant, these susceptibility rates appear consistent with data reported from other countries. To date, no published data on ERV susceptibility have been reported from Türkiye.
Delafloxacin is a novel fluoroquinolone notable for its activity against pathogens proliferating under acidic conditions. However, emerging reports of DLX resistance underscore the need for cautious use (32).
In a study by Jordán-Chaves et al. (33), 29.7% of 102 P. aeruginosa isolates resistant to ciprofloxacin and levofloxacin were found to be susceptible to DLX according to the FDA criteria, while Millar et al. (34) reported a susceptibility rate of 35.7%. In our cohort, using the FDA susceptibility breakpoint of ≤0.5 µg/mL for P. aeruginosa, 95 of 219 isolates (43%) were susceptible. Notably, 102 of 106 ciprofloxacin-resistant isolates (96%) also exhibited DLX resistance, highlighting the need for detailed molecular investigations to elucidate underlying quinolone resistance mechanisms.
The primary strength of this study lies in its comprehensive scope, representing one of the first large-scale evaluations in Türkiye to provide simultaneous susceptibility data for five novel antimicrobial agents— TOL-TAZ, CAZ-AVI, MER-VAB, ERV, and DLX—using the specific VITEK® 2 XN21 panel (bioMérieux, Marcy-l’Étoile, France). The inclusion of a large dataset allows for a robust establishment of baseline resistance epidemiology for our region. However, these results should be interpreted within the context of certain limitations. First, as a single-center study, the findings may not fully reflect the resistance patterns of other regions in Türkiye. Second, due to the retrospective design and resource constraints, molecular characterization of resistance mechanisms (e.g., PCR for carbapenemase genes) and clonal relatedness analysis (e.g., pulsed-field gel electrophoresis [PFGE] or multilocus sequence typing [MLST]) could not be performed. The lack of molecular data limits the precise interpretation of high resistance rates, particularly for agents like CAZ-AVI and MER-VAB. Finally, since clinical outcome data were not collected, the in vivo correlation of these in vitro resistance patterns remains to be established.
In conclusion, our findings indicate that the in vitro activity of several novel antibiotics against resistant Gram-negative pathogens in Türkiye is limited. High resistance rates were observed for CAZ-AVI and MER-VAB among K. pneumoniae isolates, whereas TOL-TAZ and delafloxacin retained partial activity against P. aeruginosa. Among A. baumannii isolates, available treatment options were largely ineffective. These findings emphasize that antibiotic stewardship strategies should integrate local resistance data alongside pharmacological properties to guide effective therapy. Ultimately, integrating phenotypic and molecular data is crucial for continuous national surveillance to preserve the efficacy of novel antibiotics.