Introduction
The uncontrolled and unmonitored overuse of antimicrobials in healthcare, agriculture, and animal husbandry has led to a global health crisis, resulting in resistance to both commonly used and reserve antibiotics. Gram-negative bacteria have developed resistance to higher-tier agents, posing a major concern (1). Multidrug resistant (MDR), extensively drug-resistant (XDR), and pandrug-resistant (PDR) strains are associated with increased hospital morbidity and mortality. In such cases, polymyxin antibiotics, particularly colistin, remain vital for the treatment of life-threatening infections (2,3). With carbapenem resistance among Enterobacterales reported to be 35–54% in India, colistin is frequently used as a last-line option despite its adverse effects (4). The increased use of colistin underscores the urgent need for dependable, rapid, and accurate in vitro susceptibility testing methods for consistent monitoring.
Accurate determination of antimicrobial susceptibility is essential to ensure the effectiveness of colistin therapy. However, methods such as disk diffusion or gradient strips are not ideal for colistin testing because it is a large molecule with poor diffusion through agar. This limitation, combined with electrostatic interactions between the drug and acidic or sulfate groups in the agar, results in unreliable zone diameters and inaccurate susceptibility results (5). Additionally, the VITEK® system (bioMérieux, Marcy-l’Étoile, France), widely used for automated antimicrobial susceptibility testing, may report unreliable minimum inhibitory concentration (MIC) values for colistin, with poor correlation to the broth microdilution (BMD) method (5–7).
To address these limitations, the Clinical and Laboratory Standards Institute (CLSI) has recommended more reliable methods for colistin susceptibility testing, including BMD, colistin broth disk elution (CBDE), and the colistin agar test (CAT) (8,9). Among these, the CAT is often preferred in laboratories with high sample loads because it enables simultaneous testing of multiple isolates across a range of antibiotic concentrations. However, this method still requires the preparation of a 0.5 McFarland bacterial suspension followed by a 1:10 dilution, which adds procedural complexity, increases technical workload and consumable use, and disrupts routine workflows primarily based on disk diffusion testing for other antibiotics. These challenges are particularly significant in high-throughput laboratories processing large numbers of samples daily. Recognizing the need to streamline and simplify colistin susceptibility testing, this study aimed to evaluate a simplified modification of the CAT.
Materials and Methods
The study was conducted in the Department of Microbiology, Maulana Azad Medical College, a 2000-bed tertiary care centre in New Delhi, India. Clinical Enterobacterales isolates were collected and archived between January 1, 2022, and December 31, 2024. Isolates were obtained from unique patients, and duplicate isolates from the same case or hospital outbreak were excluded. Bacterial identification was confirmed by MALDI-TOF (Vitek MS®, bioMérieux, Marcy-l’Étoile, France). Escherichia coli ATCC® 25922, Pseudomonas aeruginosa ATCC® 27853, and E. coli NCTC 13486 were used as quality control strains. All selected isolates were tested using three methods: (i) BMD, (ii) CLSI-recommended CAT, and (iii) modified CAT (mCAT).
Briefly, CLSI-approved CAT required Mueller-Hinton agar plates containing final concentrations of colistin sulfate (HiMedia®, Mumbai, India) of 0, 1, 2, and 4 μg/mL. A 0.5 McFarland bacterial suspension (approximately 1–2 × 108 CFU/mL of E. coli ATCC 25922) was prepared from three to five colonies obtained from a fresh (18–24 hours) nonselective agar plate and suspended in sterile saline. The suspension was then diluted 1:10 in sterile saline (approximately 1–2 × 107 CFU/mL) (8). Colistin agar test plates were inoculated with 10 μL of the diluted suspension (approximately 1–2 × 105 CFU per inoculum) using a calibrated sterile loop. Plates were incubated at 35°C for 16–20 hours under aerobic conditions. A maximum of 10 isolates were tested on each plate set, including control strains (9).
For the mCAT, the colistin concentration in the agar and the preparation of the 0.5 McFarland standard inoculum were maintained as in the original protocol. Inoculum size was modified as follows: instead of 10 μL of 1:10 diluted 0.5 McFarland inoculum, the recommended volume using a 1 μL calibrated sterile loop of undiluted 0.5 McFarland inoculum was used to inoculate each plate (still equivalent to approximately 1–2 × 105 CFU per inoculum). After inoculation, plates were incubated at 35°C for 16 to 20 hours under aerobic conditions.
For all methods, inocula were plated on sheep blood agar for purity checks and on growth control plates to confirm confluent growth. Interpretation of results followed CLSI criteria: MIC ≥4 μg/mL was considered resistant, and MIC ≤2 μg/mL was considered intermediate. Categorical agreement was calculated for CAT and mCAT using BMD as the reference standard. In the absence of a susceptible breakpoint for colistin, a very major error was defined as an isolate categorized as resistant by BMD but reported as intermediate by the test method (9,10).
Pilot testing demonstrated poor consistency and agreement of CAT and mCAT with BMD for P. aeruginosa and Acinetobacter spp.; therefore, these organisms were excluded from further evaluation.
Results
During the study period, 41,799 clinical bacterial isolates were processed for antimicrobial susceptibility testing, including 31,846 Gram-negative bacteria, of which 22,468 belonged to the order of Enterobacterales. Colistin resistance was detected in 128 Enterobacterales isolates (98 Klebsiella pneumoniae, 21 Enterobacter spp., nine E. coli), as well as in nine Acinetobacter spp. and seven P. aeruginosa isolates; however, 26 strains unfortunately could not be revived from stock vials.
A total of 306 Enterobacterales isolates were included for comparative evaluation, comprising 102 colistin-resistant isolates and 204 intermediate isolates. Among the resistant isolates, 79 were K. pneumoniae, 20 Enterobacter spp., and three E. coli. Carbapenem resistance was 49.9% (3488/6984) for K. pneumoniae, 59.6% (220/369) for Enterobacter spp., 35.2% (4700/13,370) for E. coli, 70.3% (3099/4406) for Acinetobacter spp., and 35.9% (1192/3314) for P. aeruginosa, highlighting the importance of accurate colistin susceptibility testing in our setting.
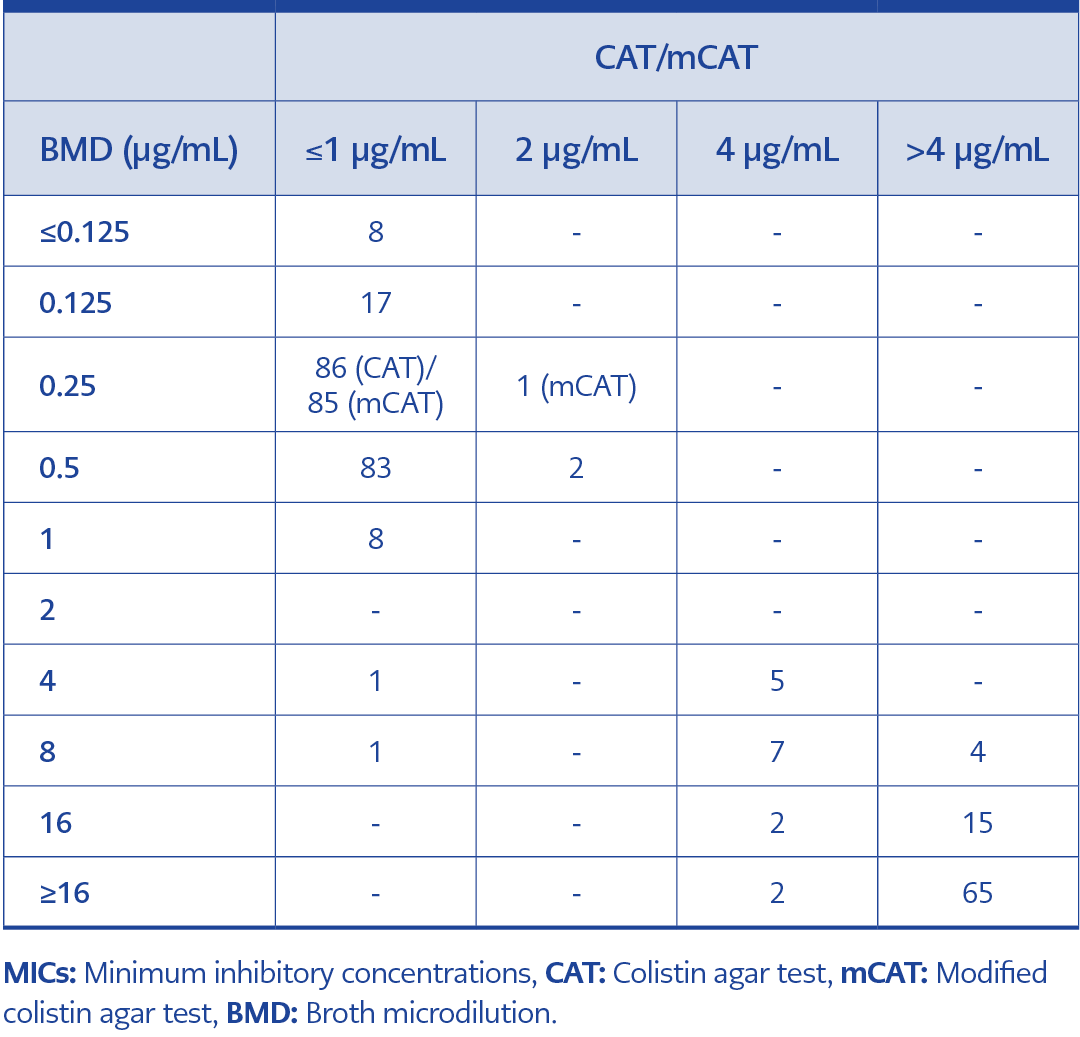
Complete (100%) categorical agreement was observed between CAT and mCAT. The detailed distribution of MIC values obtained by CAT and mCAT compared with BMD is presented in Table 1. A single MIC discrepancy was identified in a K. pneumoniae isolate, for which CAT reported an MIC of ≤1 μg/mL and mCAT reported 2 μg/mL. Compared with BMD, two isolates (both Enterobacter cloacae) produced very major errors, being categorized as intermediate by both CAT and mCAT despite resistance by BMD. Overall, CAT and mCAT demonstrated 99.3% categorical concordance with BMD (304/306 isolates).
Discussion
Colistin is frequently considered the last-line therapeutic option for treating infections caused by MDR and XDR Enterobacterales, P. aeruginosa, and Acinetobacter spp. (1). Consequently, accurate susceptibility testing and reporting are essential in clinical laboratories (9). In June 2019, the CLSI Antimicrobial Susceptibility Testing (AST) Subcommittee provisionally approved both the colistin broth disk elution (CBDE) and CAT methods for testing Enterobacterales and P. aeruginosa (11). However, currently recommended methods, including BMD and CAT, are labor-intensive and time-consuming, which limits their practicality in high-throughput laboratory settings.
In this study, we evaluated BMD-confirmed colistin-resistant and intermediate Enterobacterales isolates using CAT and mCAT. The inclusion of both resistant and intermediate isolates allowed validation of the proposed modification. Complete (100%) categorical agreement was observed between CAT and mCAT, and both CAT and mCAT demonstrated 99.3% categorical agreement with BMD (304/306 isolates).
To the best of our knowledge, and despite a thorough literature search, a similar simple modification of CAT has not been previously reported. A multicenter study assessed CBDE and CAT methods for Enterobacterales, P. aeruginosa, and Acinetobacter spp. using two inoculum volumes—1 μL (CAT-1) and 10 μL (CAT-10)—prepared from a 1:10 dilution of a 0.5 McFarland standard and compared the findings with CBDE. The categorical agreement for Enterobacterales was 99.4% with CAT-1 and 99.7% with CAT-10. Similarly, P. aeruginosa showed agreement rates of 98.7% and 100%, respectively. Due to the presence of very major errors with Acinetobacter spp., the CLSI AST Subcommittee endorsed CBDE and the CAT-10 method specifically for testing Enterobacterales and P. aeruginosa. Notably, that study did not evaluate 1 μL of undiluted 0.5 McFarland inoculum (11).
In a study by Pasteran et al. (12), a colistin agar spot test using Mueller-Hinton agar plates supplemented with 3.0 μg/mL of colistin was inoculated directly with an undiluted 0.5 McFarland suspension. They reported a categorical agreement of 95.5% with the reference BMD method, along with 0.7% very major errors and 3.8% major errors.
Several studies have compared the CLSI-recommended CAT with the reference BMD method for colistin susceptibility testing and have reported a reasonable agreement. For example, a study conducted at the All India Institute of Medical Sciences (AIIMS), Patna, in August 2024 evaluated the merits and limitations of the CAT method by comparing its performance with that of the BMD method in 108 clinical isolates of K. pneumoniae and E. coli. The study reported lower categorical agreement rates of 90.74% for K. pneumoniae and 98.07% for E. coli, suggesting the possibility of intercenter variation (1).
Although agar dilution methods offer several advantages, the CAT method is labor-intensive compared with disk diffusion and must be prepared in-house, with a limited shelf life. The additional step of preparing a 1:10 dilution further increases workload and consumable use. Therefore, the mCAT method provides a more efficient workflow while maintaining reliable results for Enterobacterales. The modification proposed in this study represents a simple and low-cost approach, particularly suitable for low- and middle-income countries with heavy workloads and limited access to reference BMD. In our experience, laboratories testing an average of 20–25 Enterobacterales isolates daily may save up to 30 minutes of work per day by omitting this step, including time required to prepare additional sterile saline tubes and perform 1:10 dilutions, without significantly altering test outcomes. Greater time and resource savings may be achieved in laboratories processing higher numbers of isolates.
Although CAT is not recommended by CLSI for Acinetobacter spp., it is endorsed for P. aeruginosa. In our preliminary evaluations, mCAT was also assessed for Acinetobacter spp. and P. aeruginosa; however, further testing was discontinued due to technical issues and a high error rate. These observations suggested suboptimal concordance with BMD, although a formal comparative analysis was not performed.
Our study has several limitations. As a single-center study with limited species representation, broader multicenter studies are needed to strengthen the evidence. Although only de-duplicated isolates from unique patients were included, molecular typing was not performed to exclude potential clonal relatedness. Future studies may benefit from molecular confirmation of colistin resistance mechanisms (e.g., mcr-1, mcr-2, mgrB, pmrAB mutations).
In conclusion, although CLSI recognizes BMD as the reference standard for colistin susceptibility testing, its implementation requires significant technical expertise and specialized training. The CAT method offers a practical alternative for testing multiple isolates simultaneously; however, it may still be challenging in laboratories with limited resources or high sample volumes. In this study, the mCAT method demonstrated a clear advantage by reducing procedural workload while yielding results comparable to those of CAT and BMD.