Introduction
Bloodstream infections (BSIs) occur when microorganisms overcome the host’s normal defense mechanisms and spread through the bloodstream. These infections can lead to serious outcomes, such as sepsis, septic shock, and organ dysfunction, significantly increasing mortality and morbidity rates (1,2). In Europe, 1,200,000 episodes of BSIs are estimated to occur annually (3), whereas the incidence ranges between 122 and 220 cases per 100,000 population (4). The Turkish Society of Intensive Care Medicine and Sepsis Study Group conducted a multicenter study in 94 hospitals to assess sepsis in Turkish intensive care units (ICUs). The study reported prevalence rates of 10.9% for sepsis, 17.3% for severe sepsis, and 13.5% for septic shock, with high mortality rates of 55.7% for severe sepsis and 70.4% for septic shock (5).
BSIs can be caused by a variety of microorganisms. The most commonly isolated infectious agents are Gram-positive cocci and Gram-negative bacilli (6). Recent reports indicate a shift in the prevalence of microbial agents causing BSIs, with Gram-negative pathogens increasingly responsible for bacteremia (7). The order Enterobacterales includes various genera that primarily reside in the gut and share similar biochemical and genetic traits. Some members are significant opportunistic human pathogens responsible for various infections. Notable genera include Klebsiella pneumoniae, Escherichia coli, Proteus spp., Citrobacter spp., and Enterobacter spp. (8).
In hospitals, high antibiotic selective pressure contributes to the emergence of multidrug-resistant (MDR) pathogens (9), particularly Enterobacterales (e.g., E. coli and K. pneumoniae) (7,10). Infections caused by extended-spectrum β-lactamase (ESBL)-producing bacteria are challenging to the physicians (11). Carbapenems are commonly used to treat infections caused by these strains. However, their extensive use has resulted in carbapenem-resistant Enterobacterales (CRE), which are difficult to treat (12). Infections caused by CRE are associated with higher mortality rates, increased healthcare costs, and prolonged hospital stays (13).
Empirical antibiotic therapy is initiated promptly in suspected cases of sepsis, based on clinical and epidemiological data (14). Early intervention is critical, as the risk of mortality increases with each hour of delay (10). A thorough understanding of pathogen susceptibility patterns may assist clinicians in optimizing empirical therapy at an earlier stage (15). This study aimed to retrospectively evaluate the distribution and resistance patterns of Enterobacterales from blood cultures at Necmettin Erbakan University Meram School of Medicine Hospital in Konya, Türkiye, over a four-year period.
Materials and Methods
This retrospective, laboratory-based descriptive study was conducted at Necmettin Erbakan University Meram School of Medicine Hospital and was approved by the university’s Ethics Committee (Decision no. 2024/5400). Blood culture samples collected between January 2020 and January 2023 were included in the study.
Blood samples were collected in blood culture bottles and then loaded into the BACT/ALERT automated system (BioMérieux, Marcy L’Etoile, France) and incubated for five days. Samples that signaled positive were cultured on 5% sheep blood agar and eosin methylene blue agar and incubated at 37°C for 24–48 hours. Isolated microorganisms were initially identified based on colony morphology and Gram staining characteristics using conventional methods. Species-level identification was performed using the automated BD Phoenix system (Becton Dickinson, Sparks, MD, USA). Antimicrobial susceptibility testing was performed using the same system. Susceptibility to colistin and tigecycline was determined using the microdilution method. The minimum inhibitory concentration (MIC) values were interpreted according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) guidelines (version 15.0).
For patients with multiple blood culture samples, only the first isolate of the same bacterium was included in the analysis. For blood samples collected simultaneously from the right and left arms of the same patient, if the same bacterium was isolated from both cultures, the isolate was considered the causative pathogen. Samples considered contaminants were excluded from the study.
Extended-spectrum β-lactamase production was detected using a modified double-disk synergy test (DDST) with cefotaxime, ceftazidime, cefepime, and aztreonam discs placed 20 mm apart around a disc containing amoxicillin-clavulanic acid. Statistical analyses were performed using the Statistical Package for Social Sciences (SPSS), version 20.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were used to calculate the frequencies and percentages of age groups, gender, isolated organisms, antimicrobial susceptibility, and resistance rates.
Results
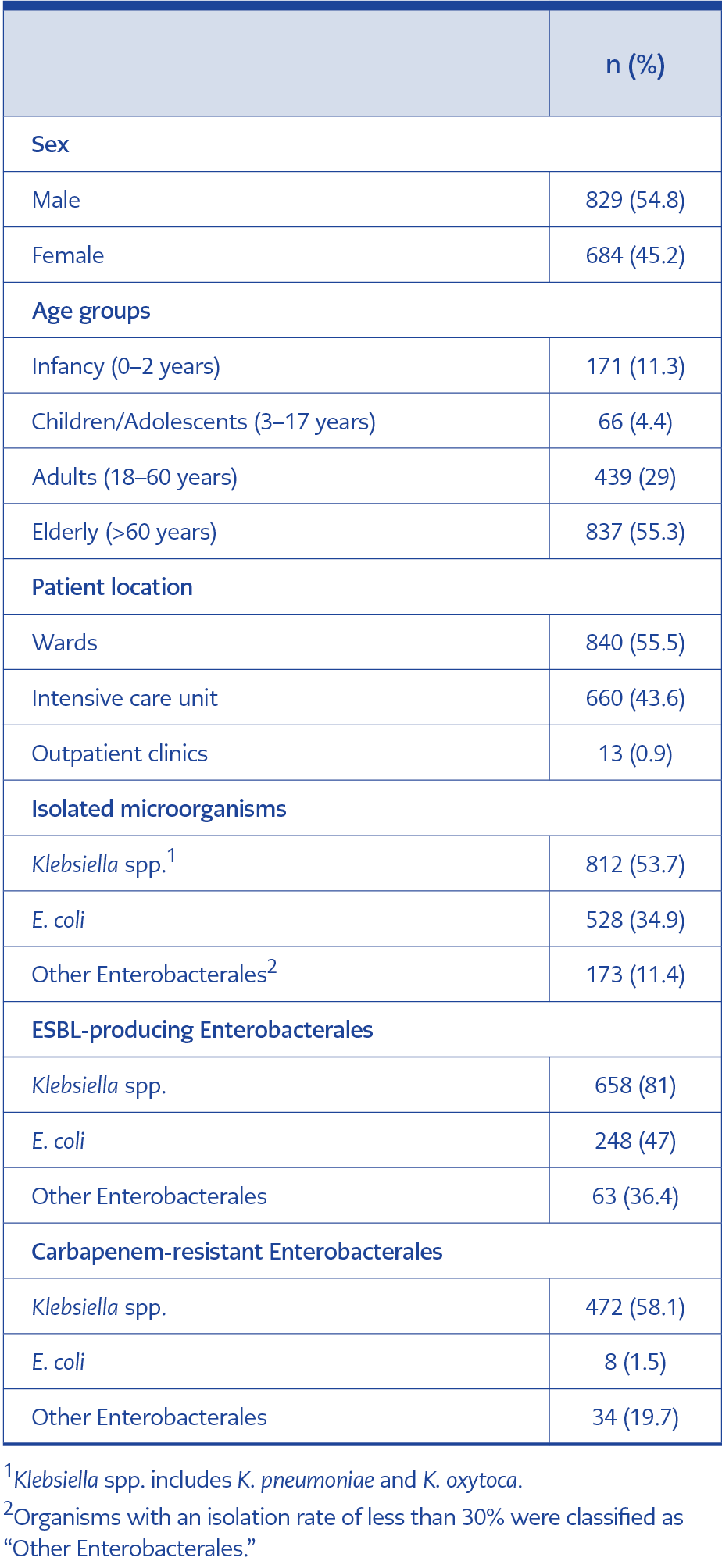
Table 1. General characteristics of patients with Enterobacterales bloodstream infections, 2020–2023
A total of 11,319 blood culture samples were analyzed during the study period. Table 1 presents the demographic and clinical characteristics of 1513 patients with Enterobacterales bloodstream infections identified between 2020 and 2023. Among the study population, 54.8% were male and 45.2% were female. Most patients were older adults (55.3%), followed by adults (29%), infants (11.3%), and children and adolescents (4.4%).
More than half of the isolates (55.5%) were isolated from patients in general wards, whereas 43.6% were recovered from patients in ICUs. Outpatient clinic visits constituted only 0.9% of the total patient population.
The majority of Klebsiella spp. isolates (81%) and nearly half of E. coli isolates (47%) were identified as ESBL producers, whereas other members of the Enterobacterales order exhibited significantly lower ESBL production rates. Carbapenem resistance was markedly higher in Klebsiella spp. (58.1%), compared to only 1.5% of E. coli isolates.
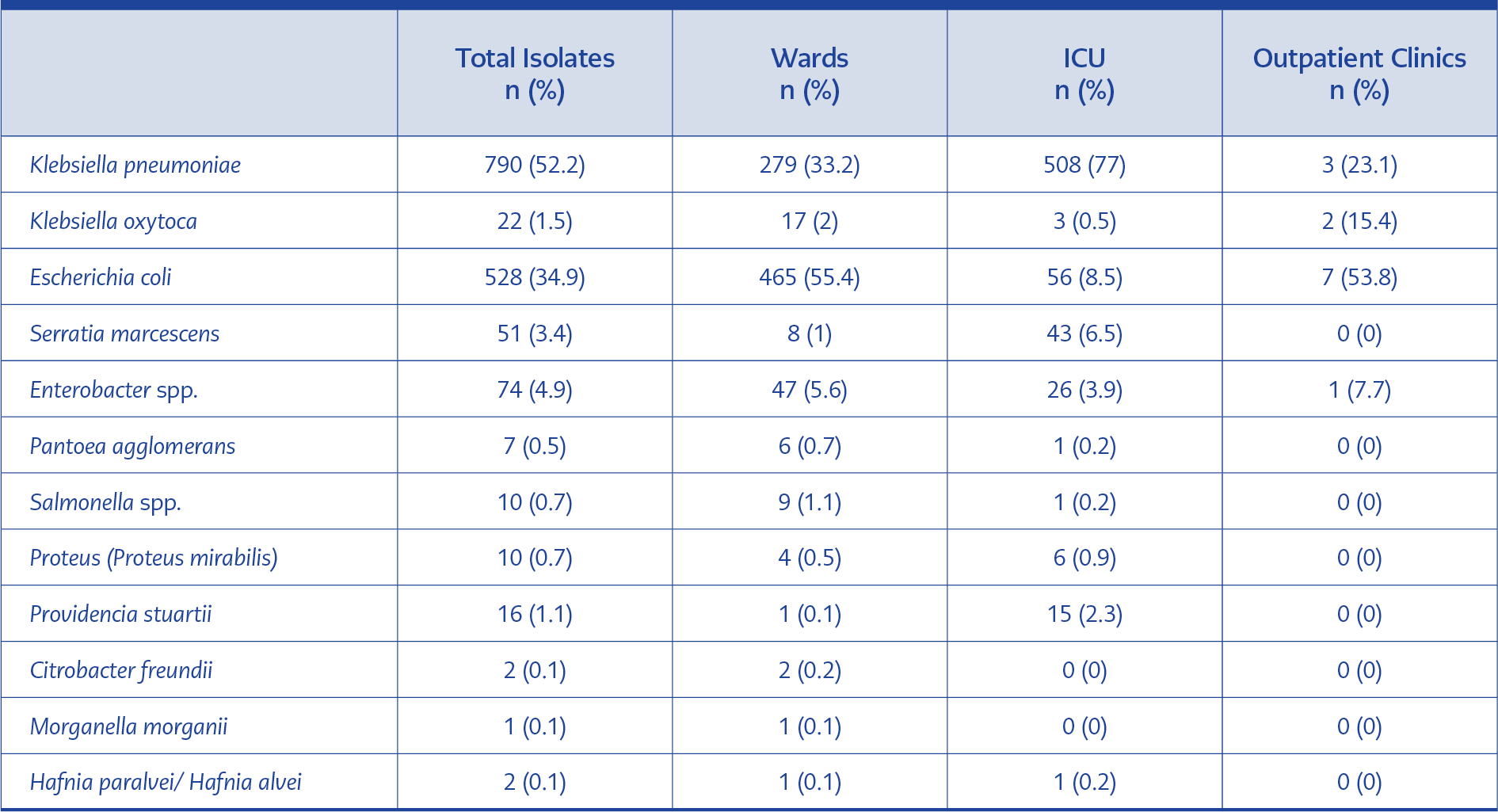
Table 2. The distribution of BSIs caused by various members of Enterobacterales in relation to different types of
patient care.
Among the identified species, K. pneumoniae and E. coli were the most dominant, accounting for 52.2% and 34.9% of the isolates, respectively. K. pneumoniae, Serratia marcescens, and Providencia stuartii were more frequently identified in ICU patients, whereas E. coli and other Enterobacterales species were primarily detected among patients from general wards and outpatient clinics, as detailed in Table 2.
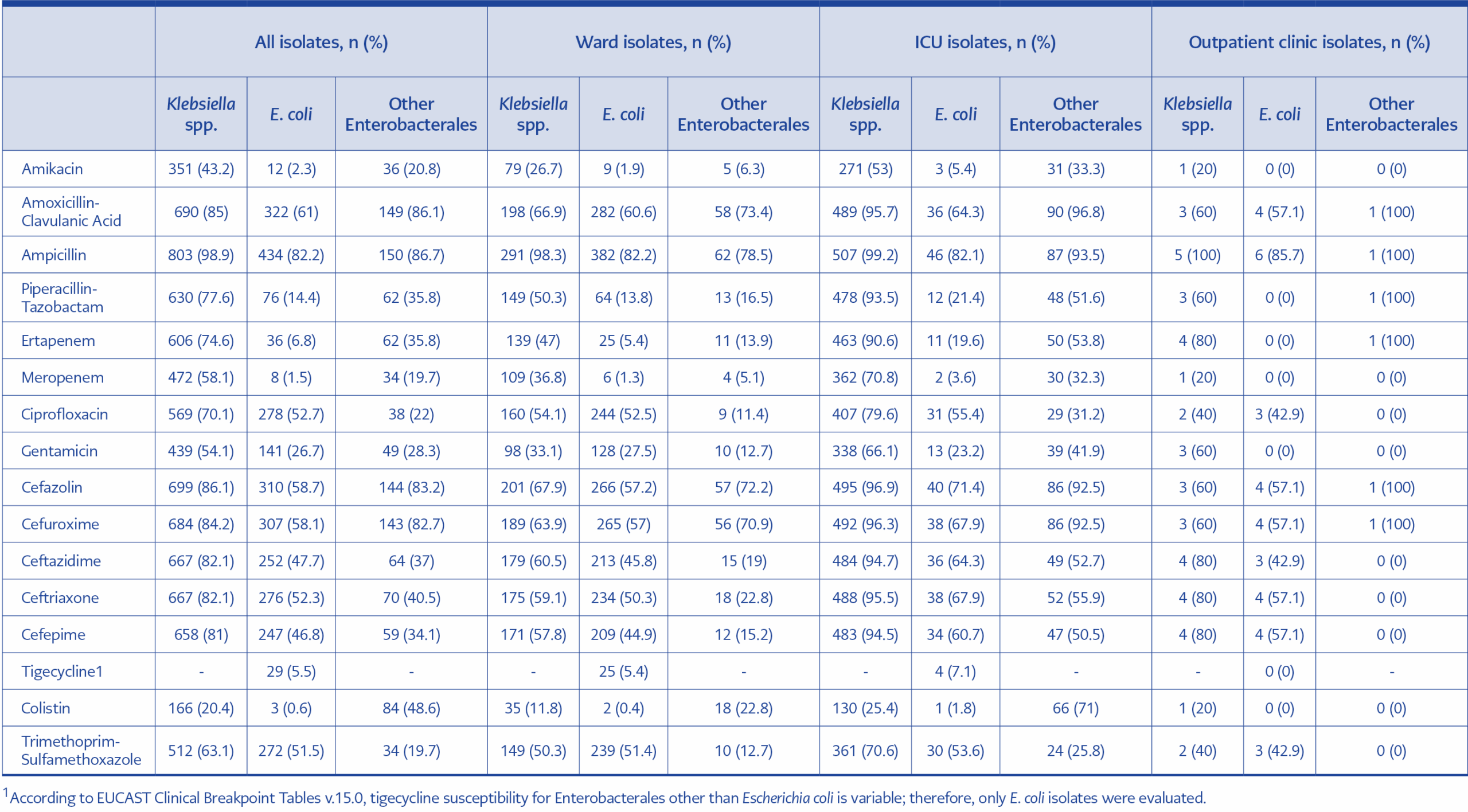
Table 3. Antimicrobial resistance rates of Enterobacterales causing bloodstream infections by patient location.
Table 3 presents the resistance patterns of Enterobacterales species across different patient settings. Almost all isolates demonstrated high resistance to ampicillin and amoxicillin/clavulanic acid. Resistance rates varied among species; Klebsiella spp. and E. coli demonstrated resistance rates of 20.4% and 0.6% to colistin, respectively. In addition, E. coli isolates exhibited low resistance to meropenem (1.5%), amikacin (2.3%), and tigecycline (5.5%). In contrast, other Enterobacterales species showed resistance rates of 19.7% to both meropenem and trimethoprim-sulfamethoxazole. Remarkably, Enterobacterales isolates obtained from ICU patients showed higher resistance rates compared to those from ward patients.
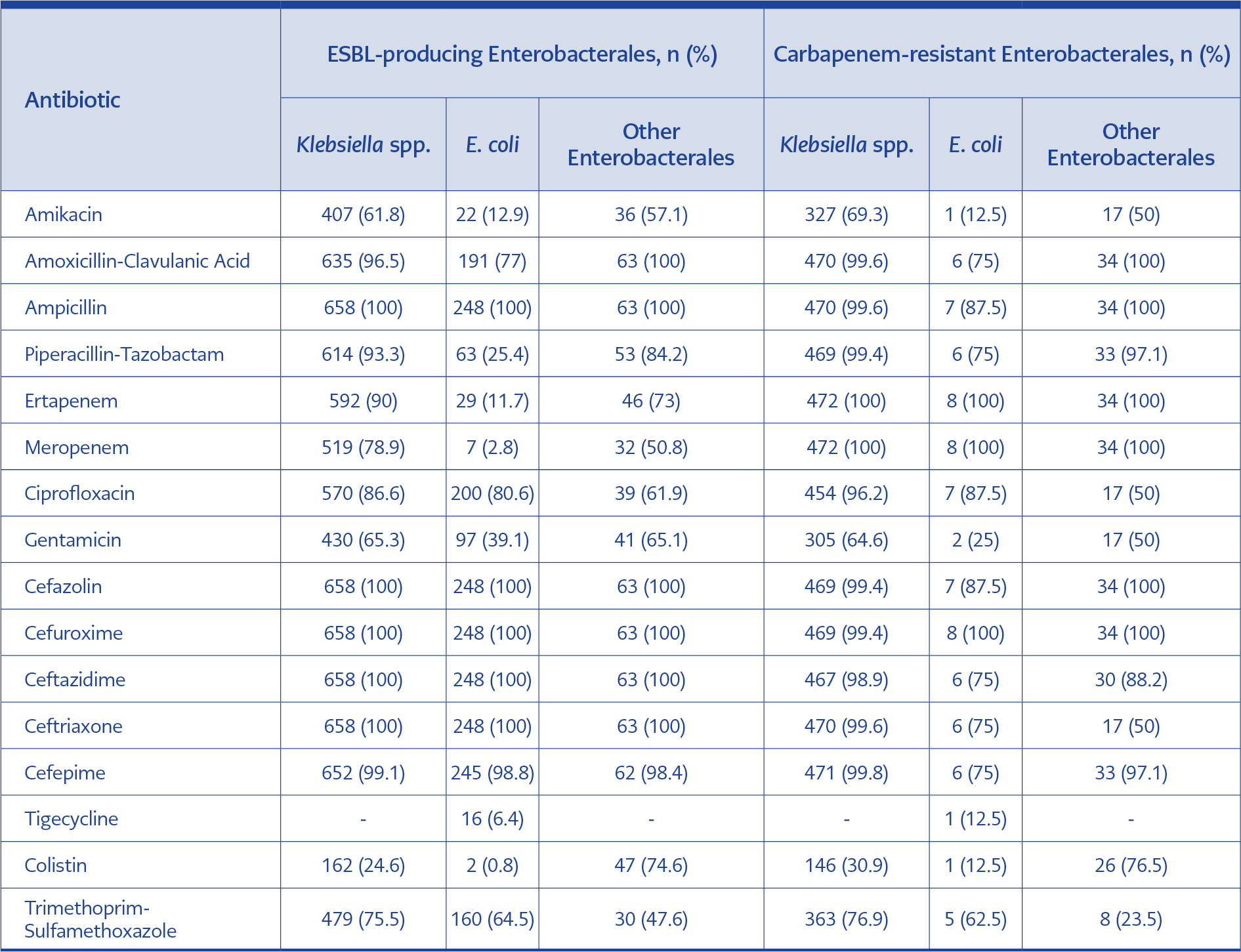
Table 4. Antimicrobial resistance patterns of ESBL-producing and carbapenem-resistant Enterobacterales.
Table 4 shows the resistance profiles of ESBL-producing Klebsiella spp., with the lowest resistance rate was observed for colistin (24.6%). Other Enterobacterales species exhibited a resistance rate of 47.6% to trimethoprim-sulfamethoxazole. Moreover, ESBL-producing E. coli isolates exhibited notably low resistance to several antibiotics, including colistin (0.8%), meropenem (2.8%), and tigecycline (6.4%).
Carbapenem-resistant Enterobacterales frequently exhibited resistance to antibiotics from multiple classes. Among these isolates, E. coli displayed a low colistin resistance rate (12.5%), whereas Klebsiella spp. showed higher resistance (30.9%). Other Enterobacterales species showed the highest colistin resistance (76.5%). In this group, the lowest resistance rate was observed for trimethoprim-sulfamethoxazole (23.5%).
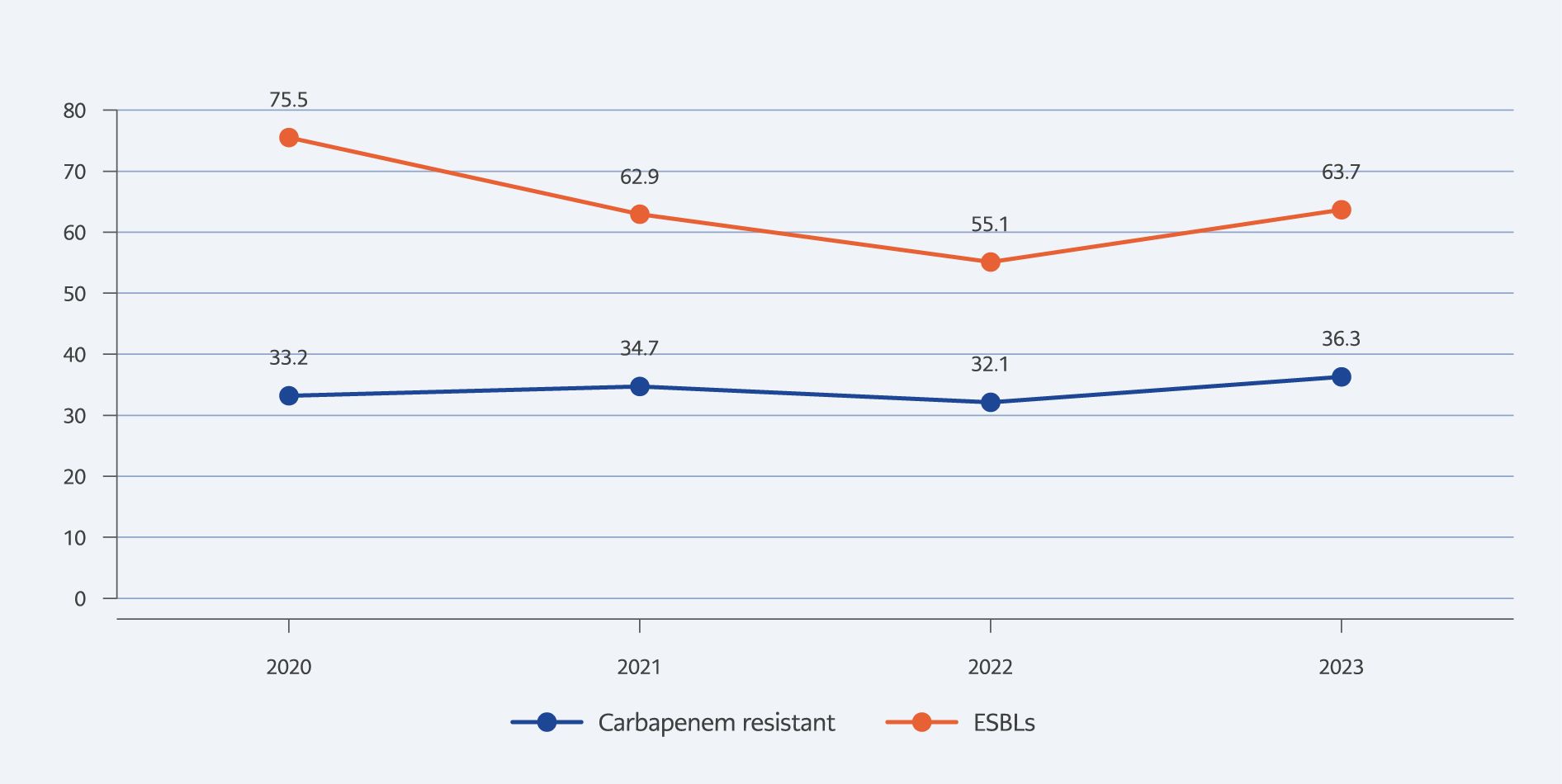
Figure 1. Percentage change in the distribution of ESBLs and carbapenem-resistant Enterobacterales over four years.
The Figure 1 illustrates the trends in ESBL-producing Enterobacterales and CRE over four years. Overall, ESBL rates consistently exceeded CRE rates, peaking at 75.5% in 2020 before declining and rising again to 63.7% in 2023. In contrast, CRE rates remained relatively stable throughout the study period but increased significantly to 36.3% in 2023.
Discussion
Bloodstream infections are prevalent worldwide and have significant direct and indirect social and economic impacts (16). They rank as the second most common condition linked to deaths caused by antimicrobial resistance, following respiratory infections (17). This study aimed to assess the distribution and susceptibility patterns of Enterobacterales responsible for BSIs and to determine the prevalence of ESBL production and carbapenem resistance among these species.
In our study, the isolation patterns of Enterobacterales from blood cultures showed that K. pneumoniae had the highest isolation rate (52.2%), followed by E. coli (34.9%). This finding contrasts with most studies conducted by other researchers (18–21), which typically identify E. coli as the predominant isolated species among Gram-negative pathogens causing BSIs. However, our results are comparable to those reported by Asena (22) and Mun et al. (23), suggesting that many patients may have developed infections associated with healthcare settings. Klebsiella spp. and other Enterobacterales were more frequently detected in ICU settings, while E. coli was more commonly isolated from patients in hospital wards. This observation aligns with a study conducted in Izmir (18).
Regarding resistance patterns, nearly all Klebsiella spp. demonstrated resistance to ampicillin (98.9%) and amoxicillin-clavulanic acid (85%). Similar findings were reported by Kłos et al. (7), who observed 100% resistance to ampicillin, and by Küçük et al. (6), who reported a resistance rate of 76.7% to amoxicillin-clavulanic acid. High resistance levels to multiple generations of cephalosporins have also been reported. Our findings are consistent with studies conducted in Türkiye (1,5), whereas a report from Poland indicated lower resistance rates for the same group of antibiotics (7). The resistance trend to penicillins and penicillin inhibitors among Klebsiella spp. may be attributed to mobile genetic elements carrying resistance genes. On the other hand, colistin exhibited the lowest resistance rate (20.4%), with most Klebsiella spp. remaining susceptible to this antibiotic. Our results are close to those reported by Kula et al. (1), who observed a colistin resistance rate of 22.6%. However, Müderris et al. (18) observed a significantly lower resistance rate of 4.5%. The extensive use of colistin for infections caused by MDR bacteria may contribute to the development of resistance.
E. coli isolates exhibited high resistance to ampicillin (82.2%), comparable to the estimates of earlier studies by Kula et al. (1) and Öksüz et al. (20). Colistin and meropenem were the most effective antibiotics against E. coli in vitro, with only 0.6% and 1.5% of isolates showing resistance, respectively. Our findings align with previous studies conducted in Türkiye (1,18). E. coli strains may remain susceptible to these antibiotics because developing resistance is energetically costly and hinders bacterial growth. Furthermore, resistance genes associated with these antibiotics remain uncommon among E. coli isolates.
The results of the present study revealed that the majority of Klebsiella spp. isolates were ESBL-producing, accounting for 81% of isolates, while 47% of E. coli isolates produced ESBLs. These rates are higher than those reported in Poland, where ESBL prevalence was 66.7% in Klebsiella spp. and 25.7% in E. coli (7). Moreover, these values exceed those previously reported in Türkiye by Öksüz et al. (20) and Varışlı et al. (21). Until recently, antibiotic prescription and use in Türkiye were not strictly regulated, which may have contributed to misuse and overuse of antibiotics and, subsequently, to the emergence of resistant bacterial strains.
Colistin was the only antibiotic that remained consistently effective against ESBL-producing Klebsiella spp., which showed a low resistance rate of 24.6%. In contrast, ESBL-producing E. coli isolates demonstrated low resistance not only to colistin (0.8%) but also to meropenem (2.8%) and tigecycline (6.4%). Consistent with many previous studies, carbapenems remain the drugs of choice for treating infections caused by ESBL-producing bacteria (20,21). However, carbapenems were less effective against ESBL-producing Klebsiella spp., which frequently exhibited an MDR pattern with low susceptibility even to carbapenems, leaving colistin as a last-resort therapeutic option.
In our study, the carbapenem resistance rate in Klebsiella spp. was 58.1%, which aligns closely with the 53.1% reported by Aslan et al. (24). However, our findings were higher than those reported in earlier studies conducted in Türkiye (20,21, 25), where resistance rates ranged from 34.6% to 39.1%. Additionally, carbapenem resistance among E. coli isolates was 1.5%. In contrast, other retrospective studies reported rates of 3.6%, 4.7%, and 6.1% (20,21,24). Differences in carbapenem resistance may be related to variations in the study periods and settings. Furthermore, the extensive use of carbapenems to treat infections caused by MDR bacteria may have contributed to the increase in resistance observed in recent years.
Colistin remained the only effective agent against carbapenem-resistant Enterobacterales; however, susceptibility varied significantly among species. The lowest susceptibility was noted in carbapenem-resistant Klebsiella spp., with a resistance rate of 30.9%. Although this rate is lower than that reported by Aslan et al. (24), it remains concerning.
Analysis of ESBL and CRE trends over the four-year study period showed that ESBL rates consistently exceeded CRE rates. Notably, the highest ESBL rate was recorded in 2020 (75.5%). According to a report by the World Health Organization (WHO), approximately 75% of hospitalized patients with COVID-19 received antibiotics during the pandemic, with cephalosporins being among the most frequently prescribed agents (26). This situation may partially explain the increased prevalence of ESBL-producing Enterobacterales observed during that period. In contrast, the proportion of CRE remained relatively stable throughout the study period but increased markedly in 2023 to 36.3%. This value falls within the range reported by the European Centre for Disease Prevention and Control (ECDC), which varies between 0% and 58.3% (13). According to ECDC reports, carbapenem resistance among Enterobacterales has increased considerably since 2019.
Conclusion
This four-year analysis underscores significant shifts in the epidemiology and resistance patterns of Enterobacterales causing bloodstream infections. K. pneumoniae has emerged as the leading isolate, surpassing E. coli. Alarmingly high resistance rates to β-lactam antibiotics among Klebsiella spp. have limited therapeutic options, leaving colistin as the last reliable therapeutic alternative. Although colistin and carbapenems remain effective against E. coli, the increasing reliance on colistin for Klebsiella infections raises concerns regarding the potential emergence of further resistance. Additionally, the surge in ESBL-producing isolates during the COVID-19 pandemic and the sharp increase in carbapenem resistance observed in 2023 highlight the consequences of excessive antibiotic use. Since part of the study period coincided with the pandemic, the findings may not be fully comparable to the non-pandemic periods. Continuous surveillance, strengthened infection control measures, improved antimicrobial stewardship programs, and the development of novel therapeutic strategies are essential to combat the mounting burden of MDR Enterobacterales in BSIs.
