Introduction
Although case numbers of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) have declined since the virus first emerged on December 29, 2019, in Wuhan, China, it has caused significant morbidity and mortality worldwide (1,2). As of June 11, 2025, more than 777 million COVID-19 cases and over 7 million COVID-19-related deaths have been reported globally (3,4).
Patients with solid organ malignancy (SOM) are more susceptible to infections due to factors such as damage to normal anatomical barriers (e.g., skin and mucosal surfaces), obstructive processes, surgical interventions, central nervous system dysfunction, nutritional deficiencies, use of invasive medical devices, and treatments including chemotherapy and radiotherapy (5). In this population, the risk of developing severe COVID-19 infection is further increased by comorbidities, cancer type, stage of cancer, and antitumor therapies received (6,7). Numerous studies have shown that COVID-19 infection leads to higher complication, morbidity, and mortality rates in patients with SOM (6–9).
Several studies have focused solely on defining prognostic factors in patients with cancer (10–14). The present study aimed to investigate the clinical course of COVID-19 in patients with SOM and to determine its impact on mortality.
Materials and Methods
This retrospective case-control study included patients aged 18 years and older who tested positive for SARS-CoV-2 by polymerase chain reaction (PCR) and were followed on either an outpatient or inpatient basis at a tertiary care hospital between March 17, 2020, and September 29, 2021. The SOM group consisted of patients with ongoing active disease or those receiving treatment, while the control group comprised COVID-19 patients without SOM but with similar characteristics in terms of age, sex, comorbidities, and vaccination status. Exclusion criteria included patients with missing data, those with hematologic malignancies, a history of bone marrow or solid organ transplantation, or patients in whom complete remission or cure of SOM had been achieved.
Disease severity in all patients was classified according to the World Health Organization (WHO) COVID-19 severity definitions (15). The performance status of patients with SOM was evaluated using the Eastern Cooperative Oncology Group (ECOG) performance scale (16).
All data were obtained retrospectively from patient files, the electronic medical record system, and laboratory results. Demographic characteristics, clinical findings, laboratory parameters, treatment modalities, and clinical outcomes (including hospitalization, intensive care requirement, oxygen support, intubation, mortality) were recorded.
Statistical Analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, version 22.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics for continuous variables were presented as means ± standard deviations or medians (minimum-maximum), while categorical variables were expressed as numbers and percentages. The Kolmogorov-Smirnov test was used to assess the normality of data distribution. For comparisons of nominal variables between groups (cross-tabulations), the Chi-square and Fisher’s exact tests were applied. The Mann-Whitney U test was used to compare continuous variables between patients with and without SOM. Risk factors affecting mortality were evaluated using multivariate logistic regression analysis, with the backward likelihood ratio (LR) method employed. The diagnostic performance of C-reactive protein (CRP), procalcitonin, lymphocyte count, D-dimer, ferritin, and lactate dehydrogenase (LDH) was assessed by calculating the area under the receiver operating characteristic (ROC) curve (AUC). The optimal cutoff values were determined using Youden’s Index. The statistical significance was set at p<0.05.
Results
The study included 580 patients with COVID-19, with a median age of 64.5 years (range: 22–95), 63.4% of whom were male. Among all patients, 22.4% were managed on an outpatient basis, while 77.6% required hospitalization. Antibiotic treatment was administered to 49.1% of patients. Bacterial pneumonia was diagnosed in 37.4%, bloodstream infections in 5.7%, urinary tract infections in 6.0%, and soft tissue infections in 0.9%.
Among patients with SOM, the most common cancer types were genitourinary cancers (27.1%) and gastrointestinal cancers (25.0%). Distant organ metastasis was present in 40.2% of SOM patients. Of those with metastasis, 74.1% had single-organ metastasis, while 25.9% had multiple-organ involvement. The most frequent single-organ metastatic sites were the lung (32.6%), liver (30.2%), brain (11.6%), and bone (25.6%). Additionally, 52.8% of SOM patients had received antitumor therapy within the past 40 days. Among these, 22.9% underwent surgery, 18.8% received chemotherapy, 4.2% received radiotherapy, 2.1% immunotherapy, and 4.9% targeted therapy. All treatments were administered as monotherapy.
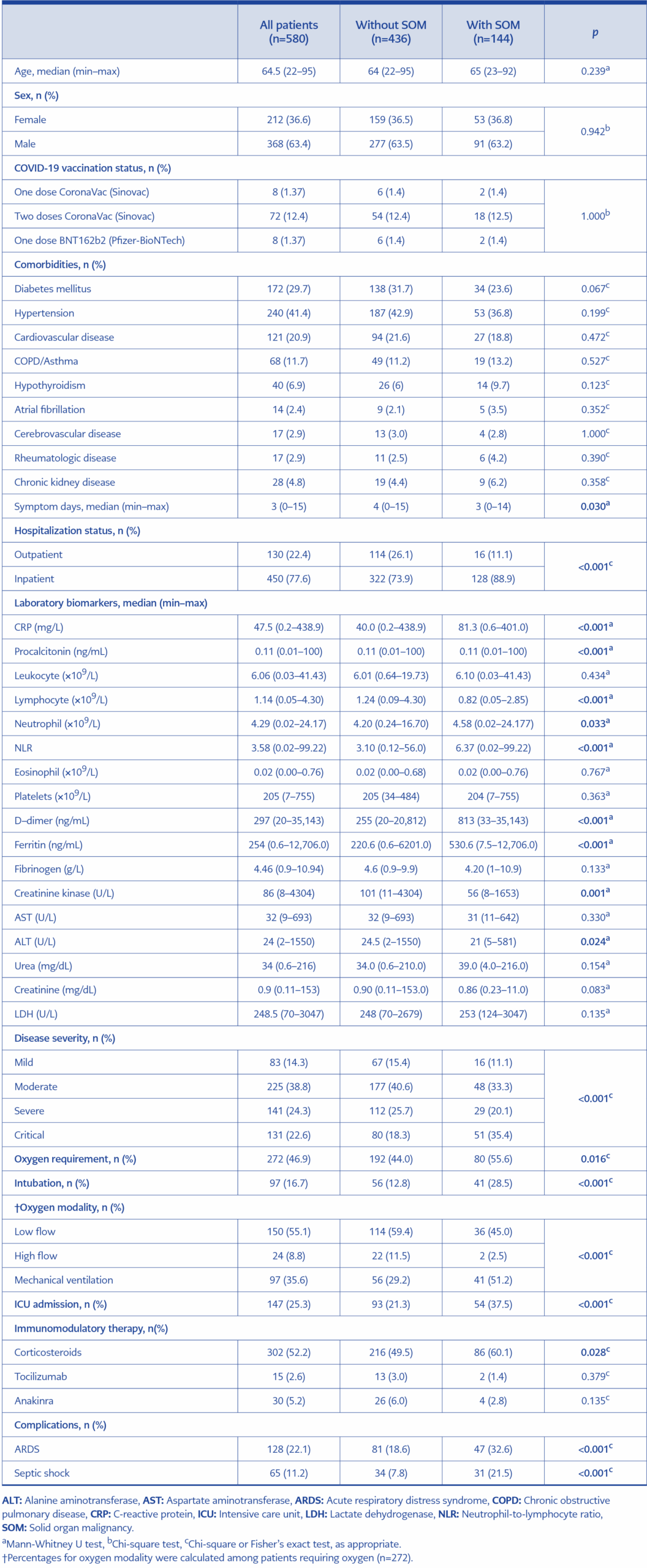
Table 1. Comparison of demographic and clinical characteristics and clinical course of COVID-19 patients with and without solid organ malignancy (SOM).
When compared to patients without SOM, those with SOM had significantly higher rates of hospitalization (88.9% vs. 73.9%, p<0.001), critical illness (35.4% vs. 18.3%, p<0.001), oxygen support (55.6% vs. 44.0%, p=0.016), intubation (28.5% vs. 12.8%, p<0.001), intensive care requirement (37.5% vs. 21.3%, p<0.001), corticosteroid therapy (60.1% vs. 49.5%, p=0.028), acute respiratory distress syndrome (ARDS) (32.6% vs. 18.6%, p<0.001), and septic shock (21.5% vs. 7.8%, p<0.001). No significant differences were observed between the two groups with respect to age, sex, vaccination status, or comorbidities (Table 1).
The median symptom onset duration at the time of hospital admission was three days. In patients with SOM, the median symptom onset duration was 3 (0–14) days, while in patients without SOM, the median symptom onset duration was 4 (0–15) days. The symptom onset duration was found to be significantly shorter in SOM patients (p=0.030) (Table 1).
Patients with SOM had significantly higher levels of CRP (p<0.001), procalcitonin (p<0.001), neutrophil count (p=0.033), neutrophil-to-lymphocyte ratio (NLR) (p<0.001), D-dimer (p<0.001), and ferritin (p<0.001), while lymphocyte counts were significantly lower (p<0.001). In all patients, the diagnostic performance of CRP, procalcitonin, lymphocyte count, D-dimer, ferritin, and lactate dehydrogenase (LDH) for predicting mortality was evaluated using ROC analysis. CRP >54.7 mg/L, procalcitonin >0.110 ng/mL, lymphocyte count <0.93 ×109/L, D-dimer >502.5 ng/mL, ferritin >451.5 ng/mL, and LDH >412.5 U/L were found to be significant predictors of mortality (p<0.001) (Table 1).
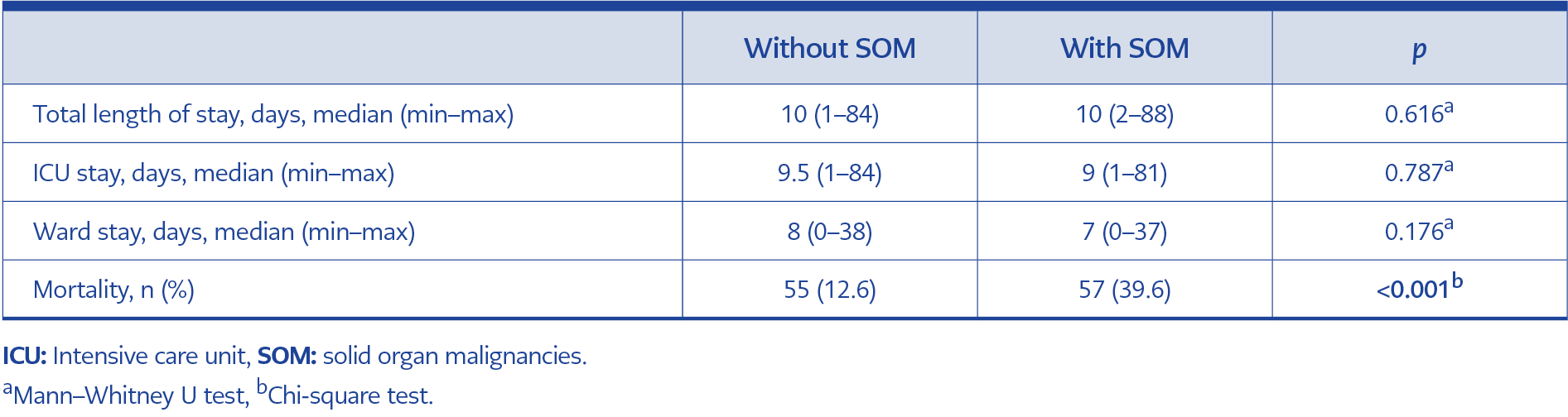
Table 2. Comparison of length of hospital stay and mortality in COVID-19 patients with and without SOM.
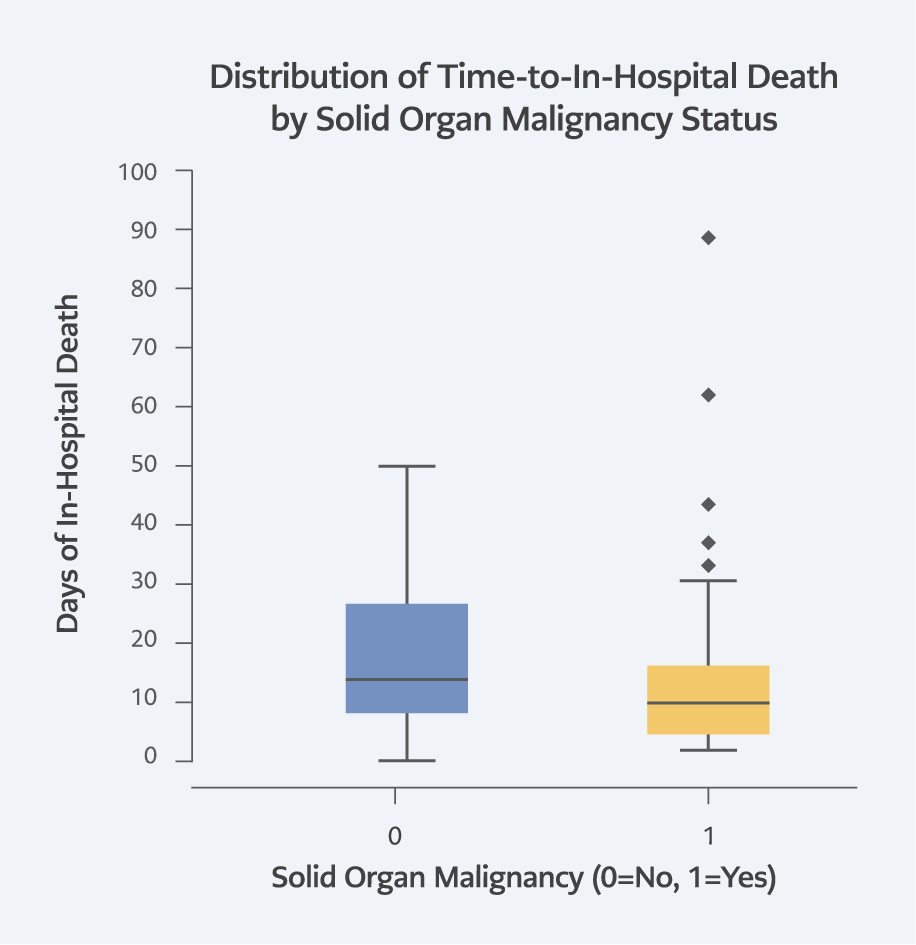
No significant difference was found between SOM and non-SOM patients in terms of total length of stay, intensive care unit (ICU) length of stay, and ward length of stay (p>0.05). However, the mortality rate was significantly higher in SOM patients (39.6% vs. 12.6%, p<0.001) (Table 2). The mean mortality day from hospitalization was 14.5 days for SOM patients and 18.3 days for non-SOM patients (Figure 1).
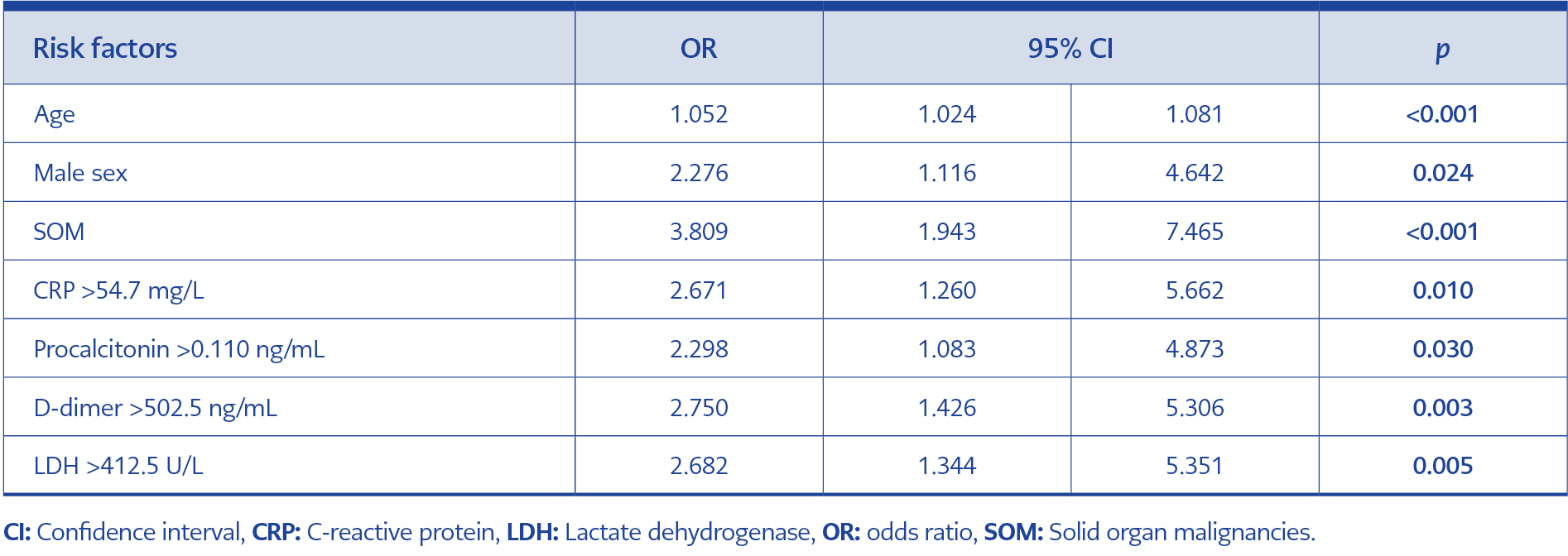
Table 3. Independent risk factors associated with mortality in COVID-19 patients: multivariate logistic regression analysis.
Multivariate logistic regression analysis identified the following independent risk factors for mortality in COVID-19 patients: age (odds ratio [OR]: 1.052, p<0.001), male sex (OR: 2.276, p=0.024), presence of SOM (OR: 3.809, p<0.001), elevated CRP (>54.7 mg/L, OR: 2.671, p=0.010), elevated procalcitonin (>0.110 ng/mL, OR: 2.298, p=0.030), elevated D-dimer (>502.5 ng/mL, OR: 2.750, p=0.003), and elevated LDH (>412.5 U/L, OR: 2.682, p=0.005). The presence of SOM was associated with a 3.8-fold higher risk of mortality (Table 3).
Among patients with SOM who died, metastatic disease and ECOG ≥2 were significantly more common (p<0.05), whereas no significant differences were observed in cancer type, site of metastasis, or number of metastatic sites (p>0.05). Furthermore, patients who died were less likely to have received antitumor therapy within the last 40 days (p<0.05). In univariate analysis, older age (p=0.036), male sex (p=0.006), metastatic disease (p=0.035), ECOG ≥2 (p=0.026), elevated CRP (p<0.001), procalcitonin (p=0.001), D-dimer (p=0.001), LDH (p<0.001), and ferritin (p<0.001) levels, as well as decreased lymphocyte count (p=0.013), were associated with increased mortality risk. In multivariate analysis, elevated CRP >54.7 mg/L (p=0.006, OR: 4.259, 95% confidence interval [CI]: 1.5–11.8) and elevated D-dimer >502.5 ng/mL (p=0.014, OR: 3.871, 95% CI: 1.3–11.3) remained significant predictors of mortality.
Discussion
Solid organ malignancies represent a major public health problem worldwide and rank among the leading causes of death. According to 2020 data, SOMs accounted for approximately 10 million deaths, corresponding to nearly one-sixth of all deaths globally (17). In individuals with SOM, both the disease itself and the immune dysfunction caused by antitumor therapies significantly increase the risk of developing severe COVID-19 infection (7,18,19).
In the present study, patients with SOM were compared with a control group without malignancy but with similar characteristics in terms of age, sex, comorbidities, and vaccination status. Our analysis revealed that patients with SOM had significantly higher mortality rates (p<0.001). Logistic regression analysis demonstrated that the presence of SOM was an independent predictor of mortality, conferring a 3.8-fold increased risk. The number of studies directly comparing COVID-19 outcomes between patients with and without SOM is limited (7–9,18,19). However, in agreement with our findings, previous studies—most of which also included hematologic malignancies—have consistently shown higher mortality among cancer patients (8,19,20). For instance, one study reported mortality rates of 31.7% in SOM patients compared to 20% in controls (p<0.001), with SOM being associated with a 1.4-fold increased risk of death (95% CI: 1.13–1.58) (20).
Although patients with SOM had a higher rate of critical illness, need for intensive care, and intubation, the length of stay was similar in both groups. Patient mortality dates were examined, and patients with SOM were found to die earlier. The similar length of stay between patients with and without SOM may reflect the increased risk of early in-hospital mortality in patients with malignancies due to fear of exposure and delayed access to care during the COVID-19 pandemic (21). In addition, COVID-19-associated thrombo-inflammatory processes, which disproportionately affect cancer patients may have contributed to rapid deterioration and shortened survival (22). Moreover, institutional pathways requiring transfer of post-COVID oncology patients back to cancer wards may also have similar hospitalization duration across groups. Collectively, these factors may explain the similar length of stay despite differences in baseline risk.
With respect to disease severity, the proportion of patients presenting with critical illness was significantly higher among those with SOM (p<0.001). In this cohort, ARDS and septic shock—two of the leading causes of COVID-19–related death—were also found to occur more frequently in patients with SOM (p<0.001). Consistent with prior studies, severe and critical disease have been described as major adverse clinical outcomes, reported to occur more often in patients with malignancies and to be strongly associated with increased mortality (6,20).
Regarding COVID-19–specific therapies, corticosteroid use was significantly more frequent in the SOM group (p<0.05), whereas no significant differences were observed between groups in the use of tocilizumab or anakinra (p>0.05). Given the poorer clinical course of patients with malignancy, higher use of immunomodulatory therapies might be expected in this group. A previous study similarly reported higher corticosteroid use among cancer patients, but no significant differences in the use of tocilizumab or other immunomodulatory agents (20). A plausible explanation may lie in treatment patterns during the early pandemic, when immunomodulatory agents such as tocilizumab and anakinra were typically reserved as rescue therapy in cases of COVID-19-related macrophage activation syndrome (MAS) not adequately controlled with glucocorticoids. The more frequent occurrence of severe clinical presentations and associated early mortality among SOM patients may have limited the opportunity to administer these therapies, potentially explaining the lack of significant difference in immunomodulatory use between groups.
In this study, cancer types, metastatic status, and functional performance of patients with SOM were evaluated in relation to mortality. Although no significant difference in cancer type was observed between survivors and non-survivors, the relatively small number of patients in each malignancy subgroup and the heterogeneity between groups may have influenced this finding. In the literature, hematologic malignancies have frequently been associated with higher mortality, and lung cancer has also been reported to carry particularly high mortality risk (6,8,23).
In our study, the presence of metastatic disease was significantly associated with mortality; however, no difference in mortality was detected between single-organ versus multiple-organ metastases, or according to the site of metastasis. Previous studies have similarly demonstrated higher mortality among patients with metastatic disease, with brain and lung metastases being particularly associated with poor short-term prognosis (6,13,24). Moreover, functional status, assessed by the ECOG performance scale, was shown to have prognostic value: patients with ECOG ≥2 had a significantly higher risk of mortality. Consistent with our findings, other studies have also reported that higher ECOG scores are associated with unfavorable clinical outcomes and increased mortality (7,25).
This study also identified several laboratory biomarkers associated with increased mortality, including CRP >54.7 mg/L, procalcitonin >0.11 ng/mL, D-dimer >502.5 ng/mL, and LDH >412.5 U/L. In the general population, one study investigating predictors of severe COVID-19 pneumonia found CRP ≥37.9 mg/L and LDH ≥335.5 U/L to be significant indicators of severe disease (26). Similarly, another study reported CRP ≥40 mg/L to be predictive of both severe disease and mortality (27). Such differences across studies may be attributable to variations in patient populations, disease course, viral variants, and regional characteristics. During the early period of the pandemic, the malignancy-related follow-up of patients with SOM was not interrupted due to quarantine restrictions. The oncology team continued to work without interruption and patients receiving treatment related to their malignancy were actively followed up.
This study has several limitations. First, due to its retrospective design, data were derived from existing medical records, raising the possibility of missing information for some clinical parameters. Second, being a single-center study may limit the generalizability of the findings. Furthermore, patients with SOM represent a heterogeneous group, with substantial variability in tumor type, disease stage, and response to antitumor therapies. These differences may have limited the ability to fully capture the impact of some variables on mortality in our analyses.
In conclusion, patients with SOM carry a significantly increased risk of mortality during COVID-19 infection. In this population, older age, male sex, and elevated inflammatory markers and biomarkers at presentation were significant predictors of poor clinical outcomes. These findings underscore the importance of early risk stratification and the implementation of individualized treatment strategies in the management of patients with cancer and COVID-19.