Introduction
Brucellosis, also known as “Mediterranean fever,” “Malta fever,” and “undulant fever,” is one of the most important zoonotic infections worldwide (1). The disease is caused by Brucella spp., which are small, non-motile, non-spore-forming, slow-growing, facultatively intracellular Gram-negative coccobacilli belonging to the family Brucellaceae (2). Brucellosis represents a significant public health problem and imposes a considerable socioeconomic burden, particularly in regions where food safety measures, hygiene standards, and veterinary control are insufficient (3).
Although the exact global incidence of human brucellosis remains uncertain, recent estimates suggest that approximately 1.6–2.1 million new cases occur annually worldwide (4). Brucellosis remains endemic in many parts of the world, including the Middle East, the Arabian Peninsula, the Eastern Mediterranean basin, Southern Europe, Latin America, the Indian subcontinent, Central Asia, and several African countries (5). Transmission to humans occurs primarily through consumption of contaminated unpasteurized dairy products, inhalation of infected aerosols, or occupational exposure to infected animals (6).
Clinically, brucellosis typically presents as an acute febrile illness; however, it may also progress to a persistent or chronic disease and lead to severe complications (7). The disease often has an insidious onset and may present with a wide spectrum of nonspecific or typical clinical manifestations, making diagnosis challenging in many patients. Accurate diagnosis, therefore, relies on the appropriate use and careful interpretation of laboratory tests together with clinical findings (1). Brucellosis is a multisystem disease that can involve almost any organ system, and hematologic, osteoarticular, gastrointestinal, genitourinary, respiratory, cardiovascular, cutaneous, and neurologic complications may occur during the course of infection (8).
In this retrospective study conducted at two hospitals, we aimed to analyze the demographic characteristics and clinical presentations, laboratory findings, complications, and treatment approaches of patients diagnosed with brucellosis.
Materials and Methods
Study Design and Patients
Patients aged ≥18 years who were diagnosed with and followed for brucellosis in the Infectious Diseases and Clinical Microbiology clinics of Osmaniye Kadirli State Hospital and Batman Training and Research Hospital between December 2018 and December 2023 were included in this retrospective study. Patient information was obtained from the hospital information management system. Sociodemographic characteristics (age, sex, occupation, history of animal husbandry, consumption of unpasteurized milk or dairy products), clinical symptoms and findings, laboratory test results, and diagnostic and therapeutic approaches were analyzed.
Diagnosis and Definitions
Diagnosis of Brucellosis
Brucellosis was diagnosed based on compatible clinical symptoms or signs together with at least one of the following criteria (7):
- Isolation of Brucella spp. from sterile body fluids, or
- Standard tube agglutination (STA) test titer ≥1/160, or
- Coombs-STA test titer ≥1/160.
Classification by Duration of Symptoms
According to the duration of symptoms (7,9), clinical manifestations lasting ≤2 months were classified as acute brucellosis, those lasting 2–12 months as subacute brucellosis, and those persisting for >12 months as chronic brucellosis.
Relapse was defined as new blood culture positivity or an increase in STA titer accompanied by recurrence of disease-specific symptoms or signs within six months after completion of treatment.
Definitions of Focal Involvement
Focal organ involvement was defined according to the following clinical, laboratory, and imaging criteria (7,9):
Hepatobiliary involvement: Alanine aminotransferase (ALT) or aspartate aminotransferase (AST) levels ≥ 5 times the upper limit of normal (ULN) were considered clinical hepatitis. Alkaline phosphatase (ALP) ≥3 times the ULN, gamma-glutamyl transferase (GGT) ≥1.5 times the ULN, or total bilirubin >2 mg/dL was considered cholestatic hepatitis.
Testicular involvement: Testicular involvement was defined as the presence of epididymitis, orchitis, epididymo-orchitis, or testicular abscess on scrotal color Doppler ultrasonography (US) together with genital symptoms such as testicular pain, swelling, or increased local temperature.
Osteoarticular involvement: In addition to the diagnosis of brucellosis, the presence of peripheral arthritis, sacroiliitis, spondylitis, tenosynovitis, bursitis, and osteomyelitis was considered osteoarticular involvement.
Hematopoietic involvement: After excluding other etiological causes, thrombocytopenia was defined as a platelet count <150,000 /μL, leukopenia as a leukocyte count <4000 /μL, leukocytosis as a leukocyte count >11,000 /μL, and anemia as a hemoglobin level <12 g/dL in women and <13 g/dL in men. The simultaneous presence of anemia, leukopenia, and thrombocytopenia was defined as pancytopenia.
Cardiac involvement: Cardiac involvement was defined as the presence of endocarditis, myocarditis, pericarditis, or pancarditis associated with brucellosis.
Statistical Analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) version 27.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics for continuous variables were expressed as mean ± standard deviation (SD) or median with interquartile range (IQR), minimum (min), and maximum (max). Categorical variables were expressed as numbers (n) and percentages (%).
The chi-square test was used to compare categorical variables. Since continuous variables were not normally distributed, the Mann-Whitney U test was used for comparisons between two groups, and the Kruskal-Wallis test was used for comparisons among three or more groups. When multiple pairwise comparisons were performed, the Bonferroni correction was applied. Statistical significance was set as p<0.05.
Results
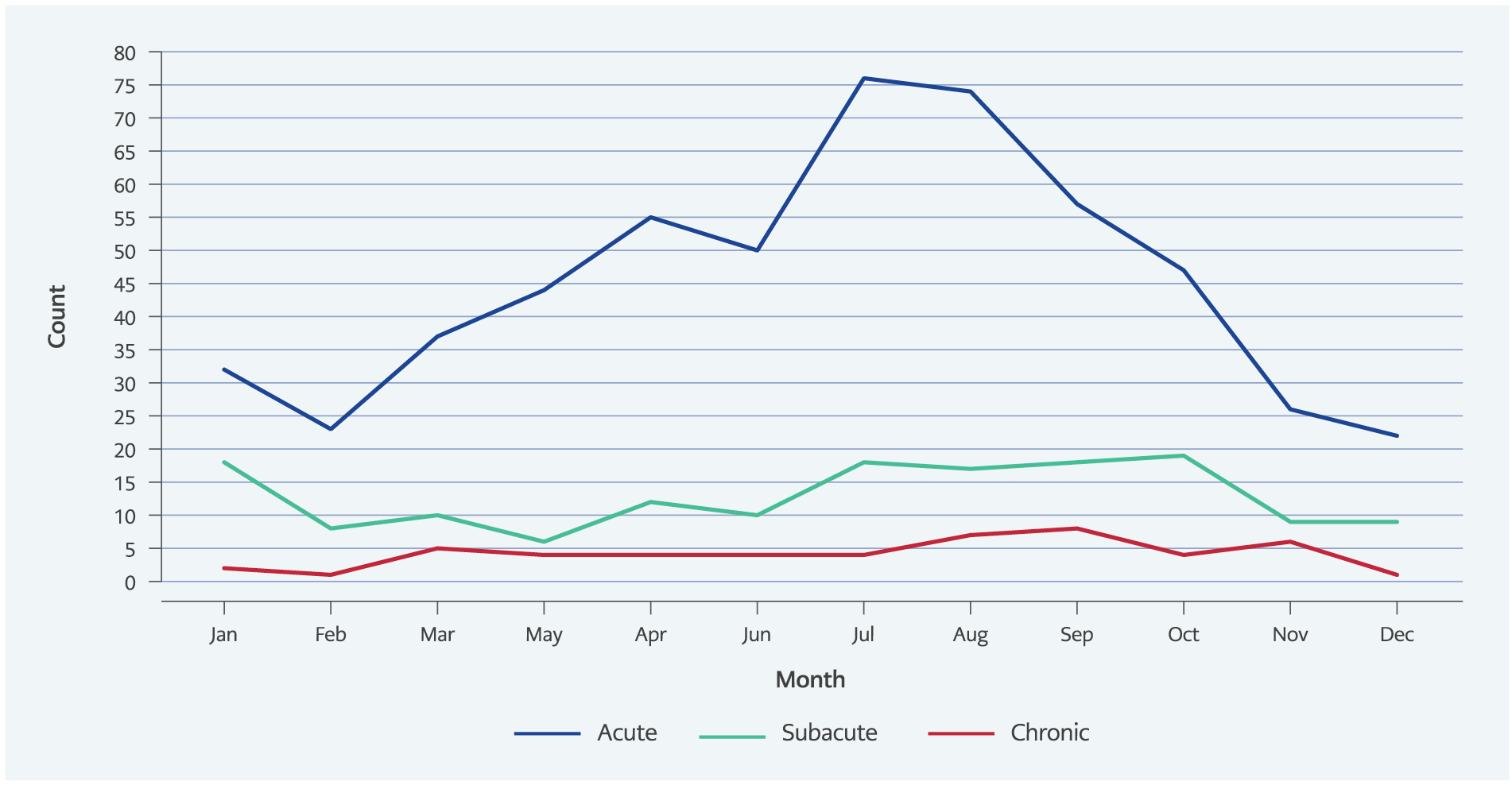
Figure Monthly distribution of brucellosis cases according to clinical stage (acute, subacute, chronic).
The study included 748 patients diagnosed with brucellosis. Of these patients, 484 (64.7%) were female, and the mean age was 39.2±15.0 (range: 18–96) years. Patients with acute brucellosis most frequently presented between June and August (Figure). The median duration of symptoms before presentation was 30 days (IQR: 46; range: 2–1460).
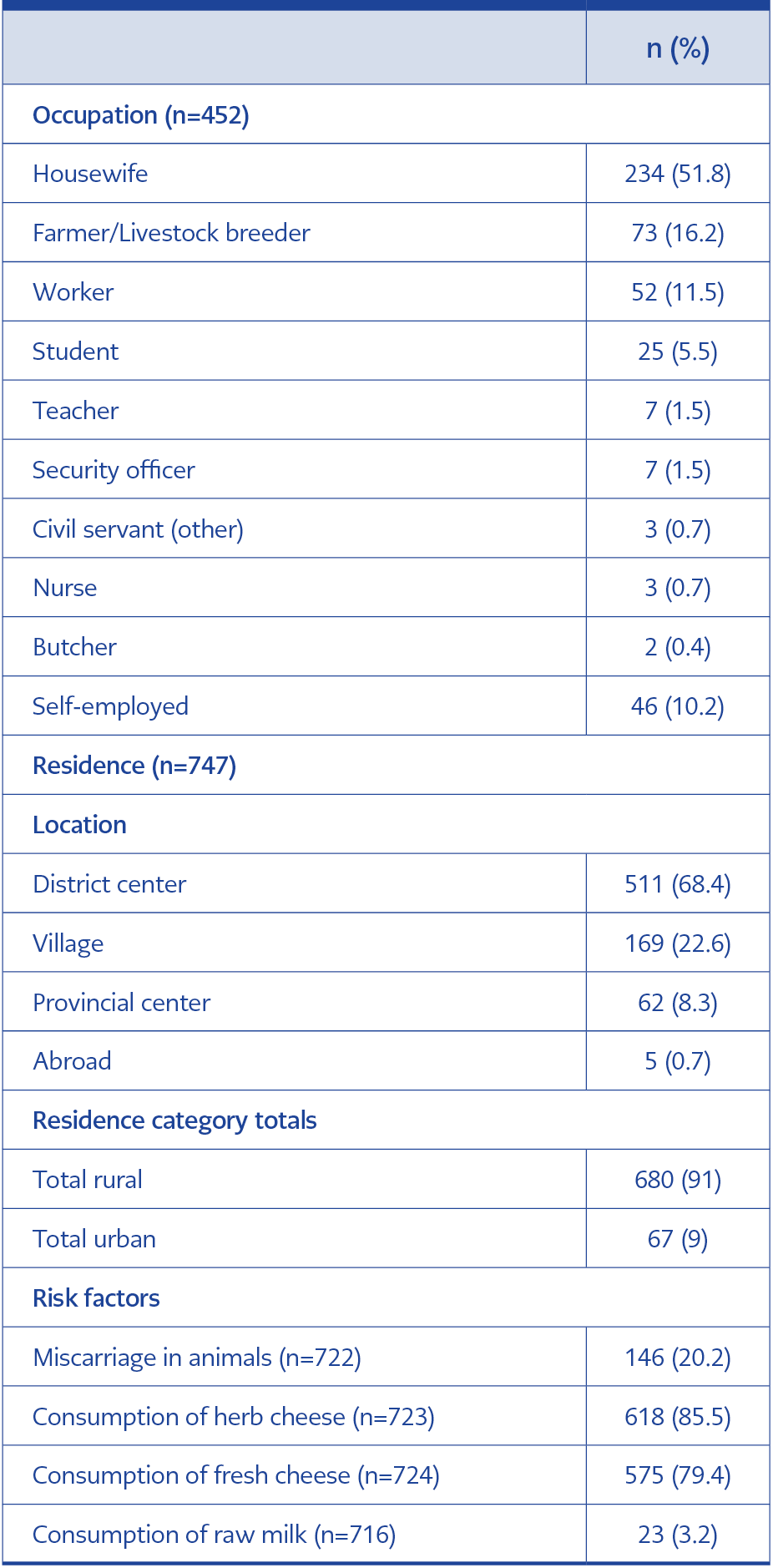
Table 1. Occupational characteristics, place of residence, and potential risk factors among patients.
The most common occupational groups were housewives (n=234, 51.8%) and farmers (n=73, 16.2%), and most patients lived in rural areas (n=680, 91%). A previous history of brucellosis was present in 263 patients (35.3%), and 151 patients (20.2%) reported a family history of brucellosis. Patients with a history of brucellosis had experienced the disease a mean of 45.6±54.5 months earlier. Regarding the mode of transmission, 575 patients (79.4%) reported consumption of unpasteurized fresh cheese (Table 1).
According to the duration of clinical symptoms, 544 patients (72.7%) had acute, 154 (20.6%) had subacute, and 50 (6.7%) had chronic brucellosis. Relapse occurred in 93 patients (12.4%); 71 (76.3%) presented during the acute phase and 22 (23.7%) during the subacute phase.
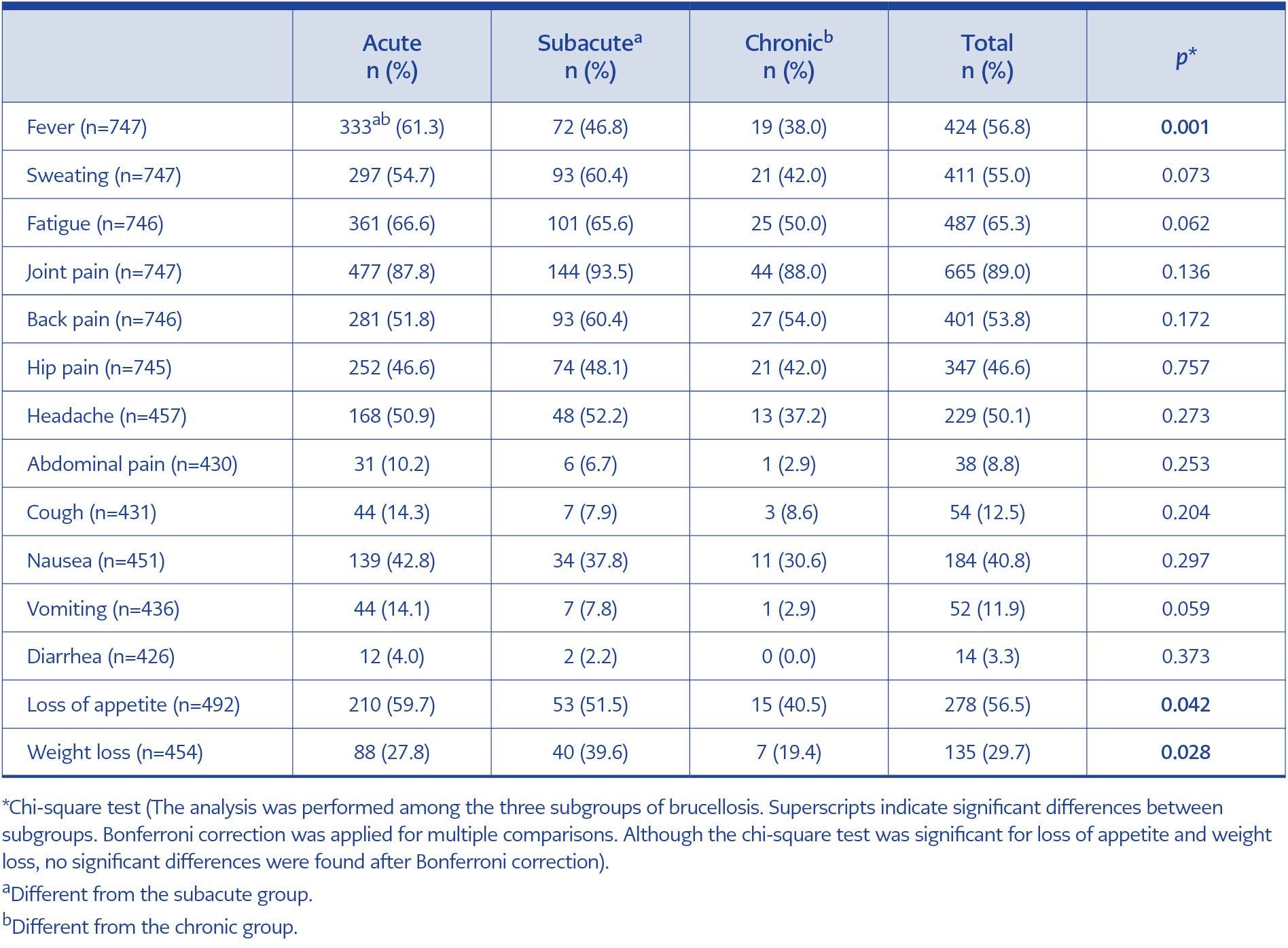
The most common symptoms at presentation were joint pain (n=665, 89%), malaise (n=487, 65.3%), and fever (n=424, 56.8%). Among patients with acute brucellosis, the most frequent symptoms were joint pain (n=477, 87.8%), malaise (n=361, 66.6%), and fever (n=333, 61.3%). In patients with subacute brucellosis, the most common symptoms were joint pain (n=144, 93.5%), malaise (n=101, 65.6%), sweating, and back pain (n=93, 60.4%). In patients with chronic brucellosis, joint pain (n=44, 88%), back pain (n=27, 54%), and malaise (n=25, 50%) were the most frequently reported symptoms.
In addition, fever and loss of appetite were significantly more common in patients with acute brucellosis (p<0.001 and p=0.042), whereas weight loss was significantly more common in patients with subacute brucellosis (p=0.028) (Table 2).
Abdominal or hepatobiliary USG was performed in 51 patients. No liver or spleen pathology was detected in 32 patients (62.7%). Hepatomegaly was reported in 12 patients (23.5%), splenomegaly in 9 (17.6%), and splenic infarction in 1 (1.9%). Most patients with hepatomegaly or splenomegaly were in the acute brucellosis group (81.2%).
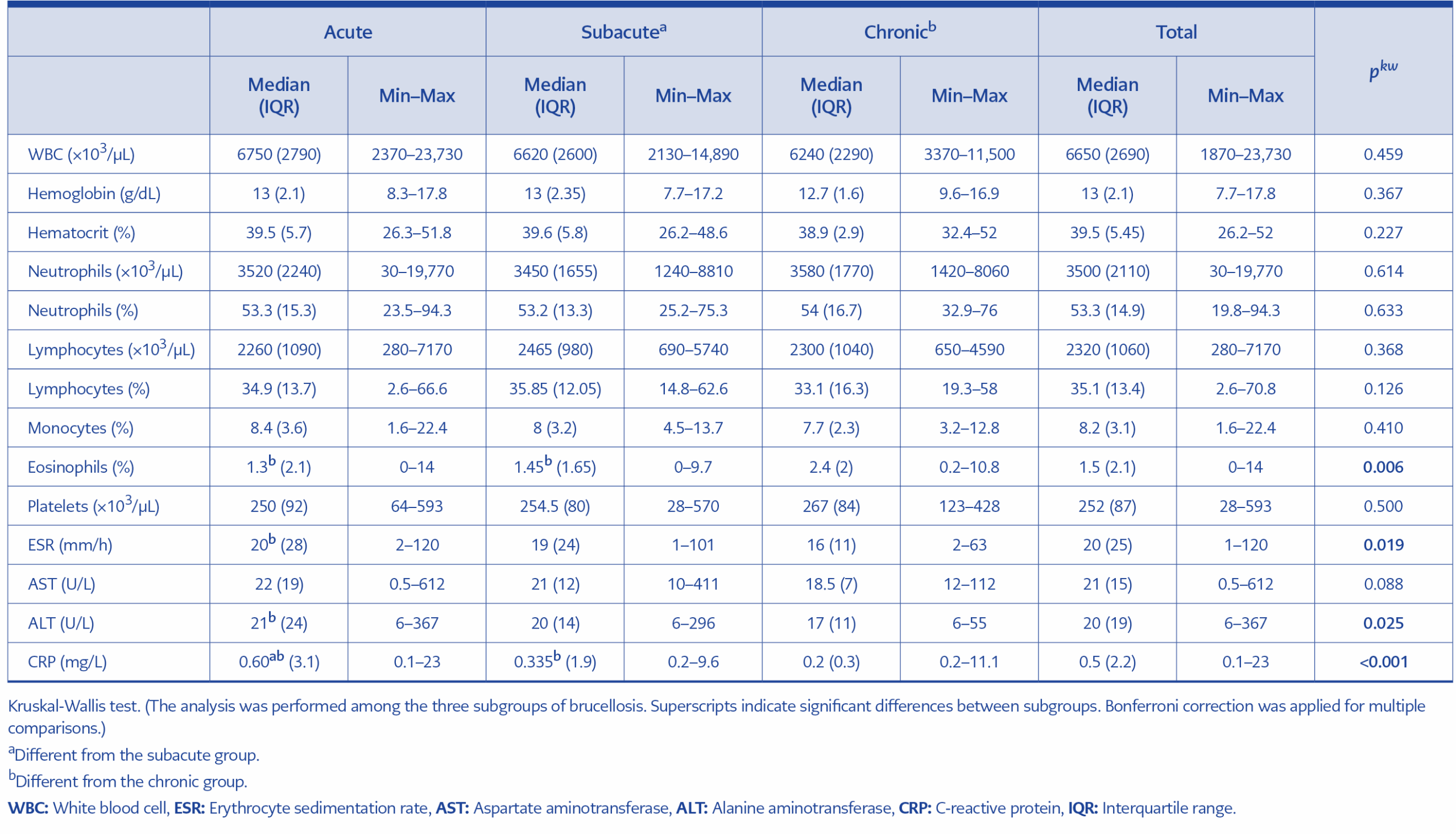
In laboratory analyses, white blood cell count, hemoglobin, hematocrit, neutrophil count, monocyte percentage, and AST levels were higher in the acute brucellosis group, whereas platelet counts were lower. However, no significant differences were observed between the other groups. Erythrocyte sedimentation rate (ESR), ALT, and C-reactive protein (CRP) levels were significantly higher in the acute brucellosis group, whereas eosinophil percentage was significantly higher in the chronic brucellosis group (Table 3).
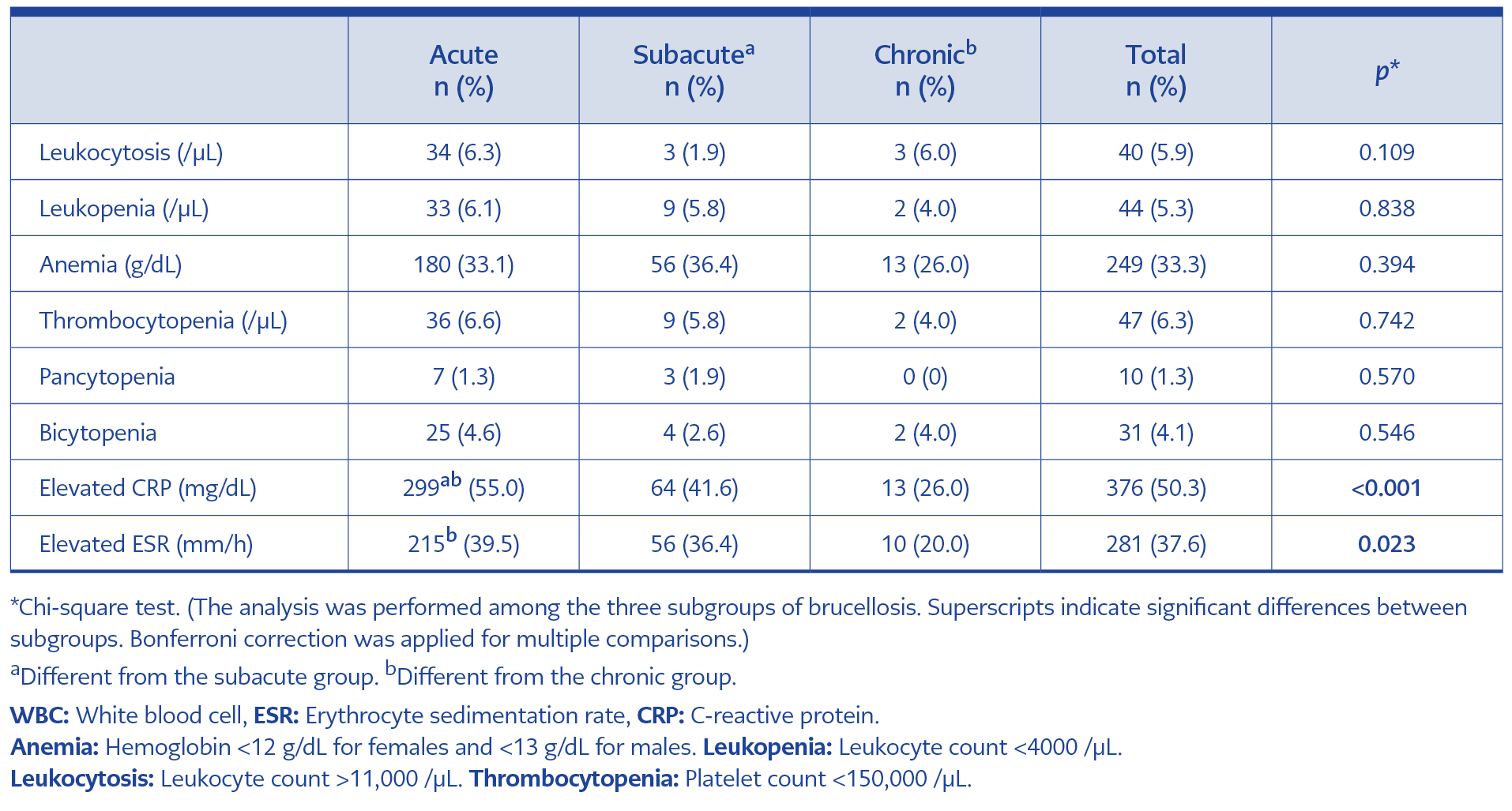
Among pathological laboratory findings, the most common abnormalities were elevated CRP (n=376, 50.3%), elevated ESR (n=281, 37.6%), and anemia (n=249, 33.3%). Elevated ESR and CRP levels were significantly more frequent in the acute brucellosis group than in the other groups (p=0.023 and p<0.001) (Table 4).
For diagnostic testing, blood cultures were obtained from 344 patients, and Brucella spp. were isolated in 109 (31.7%). Growth was detected in 35.5% of patients with acute brucellosis and 25% of those with subacute brucellosis; no growth was observed in patients with chronic brucellosis (p=0.005). Brucella spp. was also isolated from knee fluid in one patient. Brucella STA titers were ≥1/160 in all patients, and 746 patients (99.7%) had positive Rose Bengal slide agglutination test results.
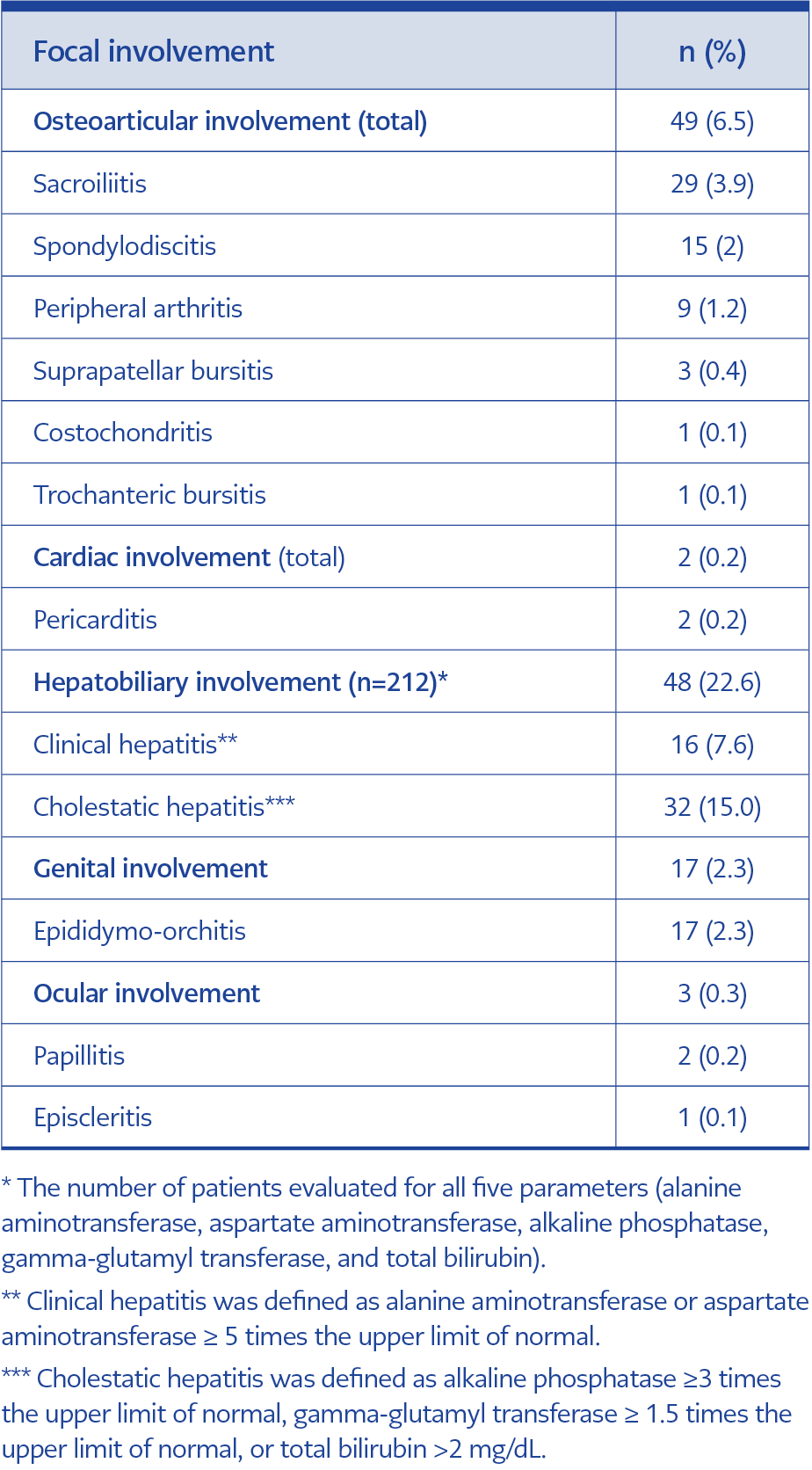
When focal involvements were evaluated, hepatobiliary involvement occurred in 22.6% of patients and osteoarticular involvement in 6.5%. The most common hepatobiliary involvement was cholestatic hepatitis (n=32, 15%). The most common osteoarticular involvements were sacroiliitis (n=29, 3.9%) and spondylodiscitis (n=15, 2%). Among patients with sacroiliitis, 26 (89.6%) had unilateral involvement (16 left, 10 right), and 3 (10.4%) had bilateral involvement. In patients with spondylodiscitis, lumbar vertebrae were involved in 12 cases (80%) and thoracic vertebrae in 3 (20%). Paravertebral or psoas abscesses were present in six patients (40%) (Table 5).
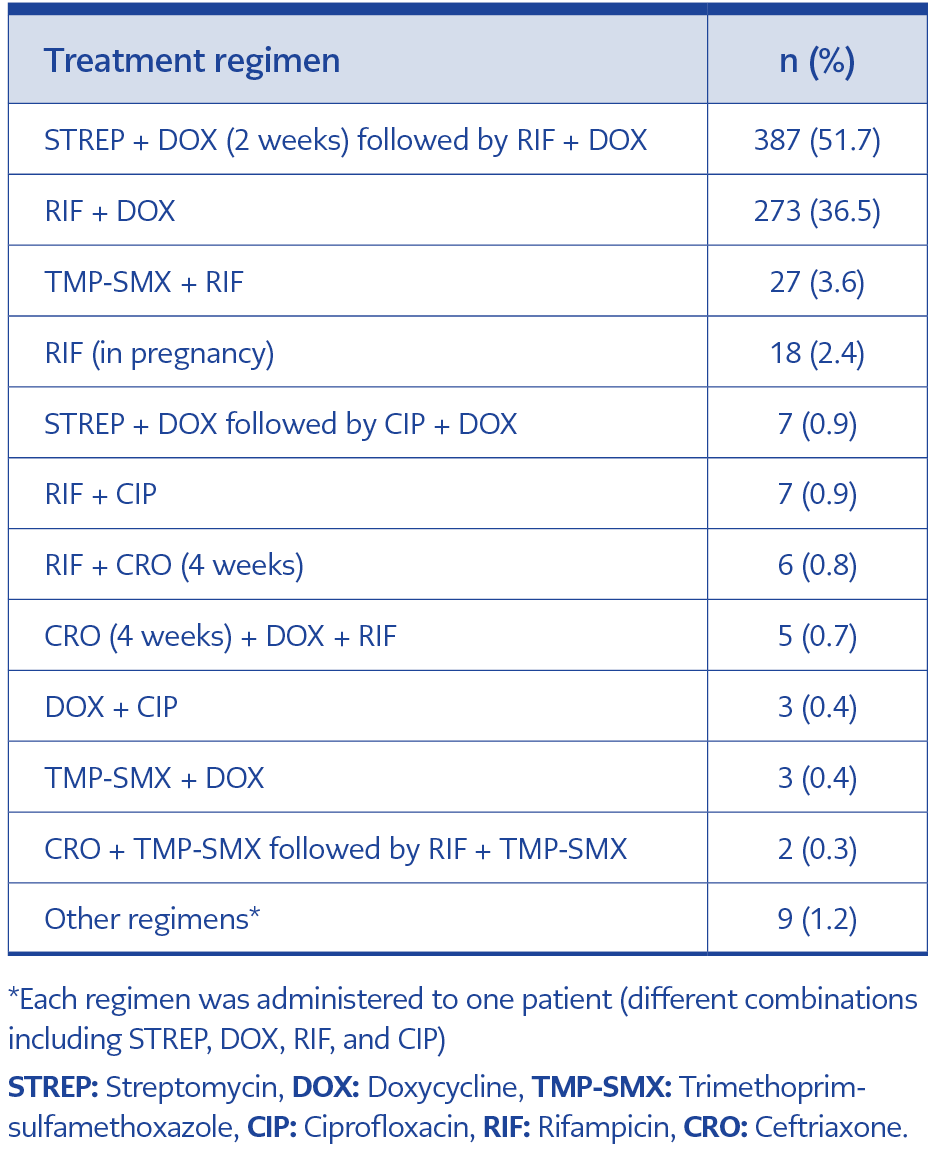
The most commonly used treatment regimen was streptomycin (STREP) plus doxycycline (DOX) for two weeks, followed by rifampicin (RIF) plus DOX for four weeks (n=387, 51.7%), followed by RIF plus DOX for six weeks (n=273, 36.5%). Other regimens included RIF + trimethoprim-sulfamethoxazole (TMP-SMX) (n=27, 3.6%) and RIF monotherapy (n=18, 2.4%). All patients treated with RIF alone were pregnant women.
Drug intolerance, adverse effects, or unavailability of certain drugs (e.g., RIF) in local pharmacies at the start of treatment led physicians to select alternative treatment regimens in some cases. For these reasons, treatment regimens were changed in 83 patients (11.1%) (Table 6).
The mean treatment duration was 7.8±3.2 weeks, and there was no statistically significant difference between brucellosis subgroups (p=0.173). In 43 patients with spondylodiscitis or sacroiliitis, the mean and median treatment durations were 14.2 and 12 weeks, respectively (range: 12–52 weeks). These durations were significantly longer than those observed in the other groups (p<0.001).
The most frequently used regimen in these cases was aminoglycoside plus DOX, followed by DOX plus RIF (n=32, 74.4%). In five cases, ceftriaxone (CRO) plus DOX plus RIF was administered. All of these patients developed abscesses; CRO was discontinued after 4 weeks, while the other agents were continued. In four cases, DOX plus RIF was used alone. No deaths related to brucellosis occurred during the study.
Discussion
Brucellosis remains a major medical problem in countries such as Türkiye, where the disease is endemic (10). In many endemic areas, factors such as uncontrolled animal movements and insufficient veterinary infrastructure hinder disease control (11). Our study was conducted in two provinces where brucellosis is endemic, and livestock farming is common. In the present study, 64.7% of patients diagnosed with brucellosis were female, and the mean age was 39.2 ±15.0 years. Most admissions occurred between June and August, and housewives (51.8%) and farmers or livestock keepers (16.2%) constituted the most affected occupational groups. Most patients lived in rural areas (91%), 35.3% had a history of brucellosis, and 20.2% reported a family history of the disease. Furthermore, a large proportion of the participants reported consuming unpasteurized milk or dairy products (79.4%).
In countries where Brucella melitensis is prevalent, the marketing and distribution of sheep and goat milk products is characterized by practices that limit the implementation of hygiene measures. As a result, the entire population may be exposed to the disease, although higher incidence rates are often observed among women and children. In regions with temperate or cold climates, the spring and summer months—when livestock abortions and births peak—represent periods of increased exposure and disease incidence for individuals involved in animal care and dairy production (1). Several studies have reported that 48%–85% of patients with brucellosis were male and that the mean age ranged between 25.6 and 45.4 years (12–17). Previous studies have also described the seasonal distribution of cases and associated risk factors. In the study by Copur et al. (14), 30.9% of patients reported a family history of brucellosis, and 64.7% consumed unpasteurized dairy products. In a large case series by Buzgan et al. (15), cases were most frequently reported during the spring and summer months, and 63.6% of patients reported consumption of unpasteurized dairy products. Similarly, Zhang et al. (18) demonstrated seasonal variation in brucellosis cases, with the number of cases peaking between April and July. In that study, 88.8% of patients reported contact with infected sheep or cattle, and 36.5% were farmers or herders. Although the seasonal, age, and occupational distribution observed in our study were consistent with previous reports, a notable finding was the higher proportion of female patients. This may be explained by the fact that many housewives in our region participate in activities such as animal care, milking, and cheese production.
In the acute stage of brucellosis, nonspecific symptoms or signs may occur, including fever, malaise, chills, sweating, weight loss, arthralgia or arthritis, lymphadenopathy, hepatosplenomegaly, and hearing loss (6,19). A meta-analysis reported that the most common symptoms of human brucellosis were fever (78%) and sweating (54%) (20). Another meta-analysis reported fever (87%) and fatigue (63%) as the most frequent symptoms (21). In the study by Zhang et al. (18), fever (82.7%) and joint pain (65.5%) were the most commonly reported symptoms. Studies conducted in Türkiye have reported fever, joint pain, and sweating at rates of 72%–89%, 63%–74%, and 59%–85%, respectively (14–16). In our study, the most common symptoms at presentation were joint pain (89%), malaise (65.3%), and fever (56.8%). In addition, fever and loss of appetite were significantly more common in patients with acute brucellosis (p<0.001 and p=0.042), whereas weight loss was more common in patients with subacute brucellosis (p=0.028).
Hematological abnormalities are common in brucellosis but usually resolve with appropriate treatment. Anemia, lymphomonocytosis, and leukopenia are common findings, while neutropenia, thrombocytopenia, leukocytosis, hemolytic anemia, and pancytopenia are rare (22). In a large case series conducted in China, lymphocytosis was reported in 34.7% of patients, anemia in 25.5%, leukopenia in 17.9%, thrombocytopenia in 9.2%, and pancytopenia in 2.7% (23). Studies from Türkiye have reported elevated CRP and ESR, anemia, thrombocytopenia, and leukopenia rates of 56%–68%, 30%–61%, 13%–54%, 8%–12%, and 7%–26%, respectively (13–16,24). Laboratory parameters may vary depending on the clinical stage of the disease. In our study, the most common laboratory abnormalities were elevated CRP (50.3%), elevated ESR (37.6%), and anemia (33.3%). Erythrocyte sedimentation rate and CRP levels were significantly higher, particularly in patients with acute brucellosis (p=0.023 and p<0.001).
Brucella species have a particular affinity for the reticuloendothelial system (RES), and hepatomegaly, splenomegaly, and peripheral lymphadenopathy may occur (25). Previous studies have reported hepatomegaly rates of 6%-26% and splenomegaly rates of 8%-35% (16,20,21,26). In our study, hepatomegaly and splenomegaly were detected in 23.5% and 17.6% of patients, respectively, and most of these patients (81.2%) had acute brucellosis. The liver, the largest organ of the RES, plays a key role in host defense against brucellosis. However, hepatic function may be affected by infection of hepatocytes and intracellular bacterial proliferation (27). In most cases, aminotransferase levels are moderately increased (less than three times the ULN), whereas clinical hepatitis is rare (27,28). The reported incidence of brucellosis-associated clinical hepatitis varies between 1%–3% (10,15,28). Cholestasis may be associated with increased ALP and GGT levels during the early stages and elevated direct bilirubin levels in advanced stages. Buzgan et al. (15) reported bilirubin levels above 2.5 mg/dL in 1.6% of patients with brucellosis. Ozturk-Engin et al. (27) found that two-thirds of the patients had findings compatible with cholestatic involvement. In our study, hepatobiliary complications developed in 22.6% of patients, and cholestatic hepatitis was the most common hepatobiliary manifestation (15%).
Since Brucella species can survive and multiply within mononuclear phagocytic cells, focal involvement, chronic disease, treatment failure, and relapse may develop over time (8). Osteoarticular involvement is the most common complication of brucellosis. Spondylitis, sacroiliitis, peripheral arthritis, and osteomyelitis are more prevalent manifestations, while bursitis and tenosynovitis are relatively rare. Lumbar vertebral involvement is the most common form of spondylitis, while sacroiliitis may occur unilaterally or bilaterally (29). Previous studies have reported osteoarticular involvement rates ranging from 22% to 47.3% (13,15,17,18,21). In our study, the rate of osteoarticular involvement was 6.5%, which is lower than that reported in the literature. The most common osteoarticular manifestations were sacroiliitis (3.9%) and spondylodiscitis (2%). Unilateral involvement was present in 89.6% of patients with sacroiliitis. Among patients with spondylodiscitis, lumbar vertebrae were involved in 80%, and 40% had associated paravertebral or psoas abscesses.
The genitourinary system is affected in approximately 2%–20% of brucellosis cases. The most common complications include epididymo-orchitis and orchitis (30). Previous studies have reported epididymo-orchitis rates between 3.4% and 9% (15,17,18,20,21). In our study, epididymo-orchitis was detected in 2.3% of patients, which is consistent with the literature.
Because brucellosis presents with variable and nonspecific clinical manifestations, microbiological confirmation is essential for diagnosis. The primary diagnostic methods include culture, serologic, and molecular polymerase chain reaction (PCR)-based tests (2). Each method has advantages and limitations related to sensitivity, specificity, and time required to obtain results (3). The definitive diagnosis of brucellosis is based on the isolation of the bacteria from blood, bone marrow, or other tissues or body fluids. Culture positivity varies depending on disease stage, prior antibiotic use, specimen type, and laboratory methods (8). Reported culture sensitivity ranges from 10% to 90% (3). Because culture methods are time-consuming and have limited sensitivity, serologic testing is widely used in clinical practice. The STA test is regarded as the reference method for serological diagnosis (8). Previous studies have reported culture positivity rates between 24% and 53% (10,12,15,17,31). In our study, Brucella spp. was isolated in 31.7% of patients overall, including 35.5% of patients with acute brucellosis and 25% of those with subacute brucellosis, whereas no growth was detected in patients with chronic brucellosis (p=0.005). Standard tube agglutination was positive in all patients, and the Rose Bengal test was positive in 99.7% of cases.
Brucellosis requires prolonged combination therapy to prevent relapse and chronicity because the pathogen can invade cells. Effective antimicrobial agents must therefore have good cellular penetration. Antibiotics commonly used in combination regimens include tetracyclines, DOX, STREP, RIF, gentamicin (GEN), TMP-SMX, CRO, and ciprofloxacin (7). The RIF plus DOX regimen is recommended by the World Health Organization (WHO) as the first-line treatment because it can be administered orally; however, some clinical studies have suggested that relapse rates may be higher with this regimen. The DOX plus aminoglycoside combination has been associated with lower relapse risk. For the disease without focal complications, the recommended treatment duration is six weeks but can be extended to 3–6 months in patients with focal involvement (7,9,32).
In our study, the most commonly used regimens were STREP plus DOX followed by RIF plus DOX, and RIF plus DOX. Although these regimens are broadly consistent with guidelines, several variations were used in clinical practice (7,9). For example, the regimen of STREP plus DOX for two weeks followed by RIF plus DOX for four weeks is not specifically described in current guidelines. Ceftriaxone therapy is recommended for special situations such as neurobrucellosis and brucellosis during pregnancy (9). In our study, CRO was occasionally used in combination with other agents, particularly in patients with sacroiliitis and spondylodiscitis. Although this approach is not explicitly recommended in major guidelines, some studies have reported the use of CRO-containing regimens in cases with osteoarticular involvement (15,16,33).
The combination most frequently administered in our study, STREP (2–3 weeks) + RIF (6 weeks) + DOX (6 weeks), is also not among the standard treatment recommendations. However, the three-month version of this triple combination is among the primary treatment options for spondylodiscitis in the guidelines (7). Although not included in the main guidelines, the literature reports lower treatment failure and relapse rates with triple regimens containing aminoglycosides. Therefore, the GEN or STREP (2–3 weeks) + DOX + RIF regimen is also recommended as the first step in standard treatment (34,35).
Relapse in brucellosis may occur for several reasons and reported relapse rates range between 5% and 15% (3). Buzgan et al. (15) found a relapse rate of 4.7%, which increased to 8.5% among patients with osteoarticular involvement. Copur et al. (14) reported a relapse rate of 12.2%, whereas Cicek et al. (16) found a higher rate of 26.2%. In our study, relapse rate occurred in 12.4% of patients; 76.3% presented during the acute stage and 23.7% during the subacute stage. Brucellosis-related mortality is low (<1%) (8), and no deaths occurred in our study.
Our study has several limitations. First, it has a retrospective design and includes data from only two centers. Second, although many patients reported a previous history of brucellosis, detailed information about their earlier treatment regimens and durations was unavailable. This limitation arose because many patients had received treatment in different healthcare facilities. Another limitation is the lack of follow-up data for pregnant patients, which would require prospective monitoring or a structured retrospective follow-up system. Despite these limitations, our study provides valuable data because of the large number of cases and the availability of detailed clinical and treatment information. We believe that our findings reflect the current epidemiological and clinical characteristics of brucellosis in two southeastern provinces where animal husbandry is widespread, and the disease remains endemic.
In conclusion, our study highlights several noteworthy findings. Brucellosis disproportionately affected women and individuals living in rural areas. Consumption of unpasteurized fresh cheese was common, and most patients presented during the acute phase of the disease. Strengthening public awareness about pasteurization, implementing effective preventive measures, and ensuring compliance with relevant regulations are essential to reduce the burden of brucellosis.