Introduction
Introduction
Antimicrobial resistance (AMR) is one of the most pressing global public health challenges, and has been classified by the World Health Organization (WHO) among the top ten threats facing humanity (1). Recent global estimates suggest that AMR directly contributes to over 1.27 million deaths annually and is associated with nearly 5 million deaths worldwide (1,2). The growing burden of AMR has important implications for human, animal, and environmental health, making it a central component of the One Health framework.
The inappropriate and excessive use of antibiotics remains the primary driver of AMR. Between 2000 and 2010, global antibiotic consumption increased by nearly 70%, highlighting widespread overuse across diverse settings (3). Non-prescription antibiotic availability, unnecessary prescribing, patient demand, and poor adherence to treatment regimens further accelerate the emergence and spread of resistant pathogens (4,5).
Given the pivotal role of healthcare professionals in antibiotic stewardship, physicians’ knowledge, attitudes, and practices are central to addressing this crisis. However, variability in stewardship adherence persists, particularly in resource-limited settings (6,7). This study aimed to evaluate physicians’ knowledge, attitudes, and practices regarding AMR at a state hospital, identify gaps, and guide educational interventions.
Materials and Methods
This cross-sectional study was conducted between June and October 2023 at a state hospital in Aydın, Western Türkiye. The study population consisted of all physicians employed at the hospital during the study period. Data were collected using a structured, self-
administered questionnaire designed to assess physicians’ demographic characteristics as well as their knowledge, attitudes, and practices regarding AMR.
Research Variables
Dependent Variables
Knowledge
Knowledge regarding AMR was assessed using 11 items. The first five items were measured using a 5-point Likert scale. To calculate a cumulative knowledge score with the remaining six dichotomous items (scored as 1=correct, 0=incorrect), Likert responses were also dichotomized. Responses of “Agree” and “Strongly Agree” were assigned 1 point, considering them as correct knowledge, while neutral or negative responses were assigned 0 points. Accordingly, the total knowledge score ranged from 0 to 11.
Attitudes
Attitudes toward AMR were evaluated using eight Likert-scale items. The mean of the responses was calculated, with the eighth item reverse-scored to control for response bias. Possible scores ranged from 1 to 5, where higher scores indicated more favorable attitudes.
Practices
Practices related to antibiotic use and AMR prevention were measured with 10 Likert-scale items (1–5). Mean scores were computed, with higher scores representing better adherence to rational antimicrobial practices.
For each dimension (knowledge, attitudes, and practices), scores were divided into quartiles (25th, 50th, 75th, and 100th percentiles). Participants with scores equal to or above the 50th percentile were classified as having “adequate” knowledge, attitudes, or practices, whereas those below the 50th percentile were categorized as “inadequate.”
Independent Variables
Descriptive Variables
Age, gender, specialist status, specialty field, department or unit of employment, and years of professional experience were recorded.
Antibiotic Prescription and Related Education Variables
Independent variables included demographic and professional characteristics such as age, gender, specialization status (specialist or general practitioner), field of specialty, department/unit of employment, and years of professional experience.
Variables related to antibiotic prescription and education were also assessed, including the primary unit of work, average daily patient load, average consultation time per patient, number of antibiotics prescribed in the preceding week, and the most frequently prescribed antibiotic (classified according to the WHO Access, Watch, and Reserve (AWaRe) Framework (8). Additionally, participants were asked about their exposure to rational antibiotic use training during residency, participation in antibiotic stewardship education within the past year, and sources of such training.
Questionnaire Development and Validation
The questionnaire was adapted from previously validated instruments used in studies by Cheoun et al. (9), Firouzabadi et al. (10), and Venugopalan et al. (11) among healthcare workers and physicians, and subsequently modified to reflect the local healthcare context. The final instrument included 29 items: 11 assessing knowledge, eight assessing attitudes, and 10 assessing practices. Before administration, the questionnaire underwent expert review by 10 physicians representing various specialties to ensure clarity, content validity, and cultural appropriateness.
Statistical Analysis
Data were analyzed using IBM SPSS Statistics version 26.0 (IBM Corp., Armonk, NY, USA). The normality of continuous variables was evaluated using the Kolmogorov-Smirnov and Shapiro-Wilk tests. Associations between continuous independent variables (e.g., age, years of experience, daily patient load, consultation time, and number of antibiotics prescribed in the previous week) were assessed using the independent-samples t-test or the Mann-Whitney U test, as appropriate. Relationships between categorical dependent variables (knowledge, attitude, and practice levels) and categorical independent variables (e.g., gender, specialization status, department, receipt of antibiotic training, and source of training) were analyzed using the chi-square (χ2) test. A p-value <0.05 was considered statistically significant.
Results
A total of 141 physicians were employed at the study hospital, of whom 99 participated, yielding a response rate of 70.2%. Among the participants, 74 (74.7%) were specialists and 25 (25.3%) were general practitioners, while 51 (51.5%) were male and 48 (48.5%) were female. The mean age was 40.29 ± 8.48 years (median=40), and the mean duration of professional experience was 15.36 ± 8.37 years (median=14). Most respondents (60.6%) worked in outpatient clinics, while 4.0% were assigned to inpatient wards and the remainder to laboratory units.
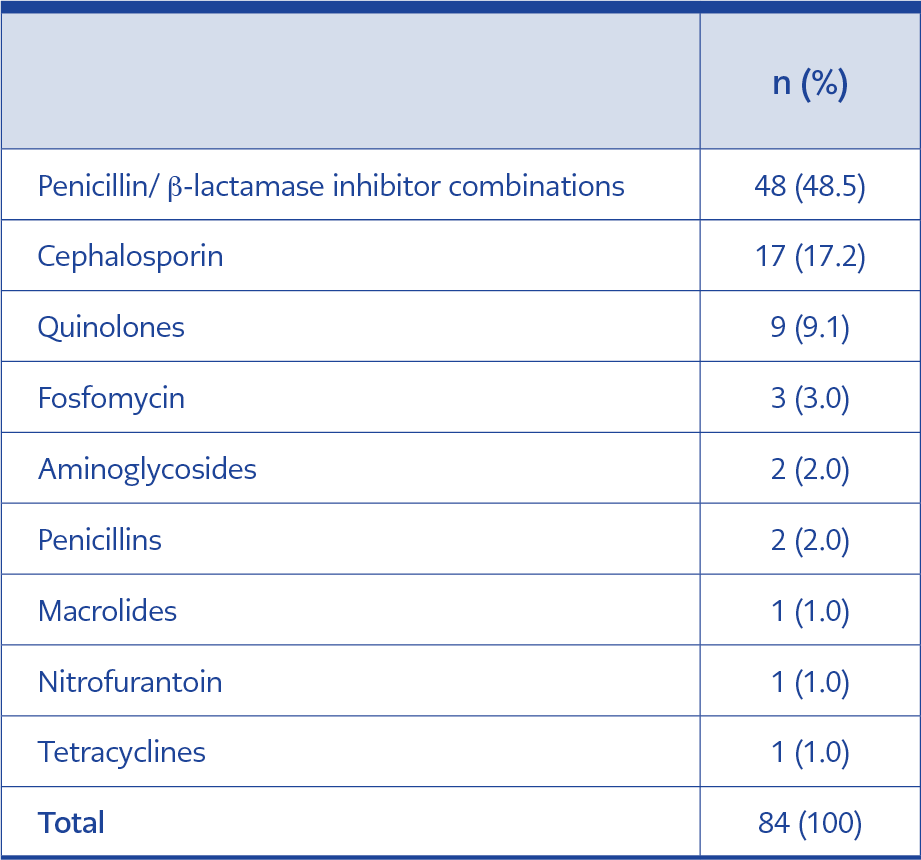
Participants reported seeing an average of 66.84 ± 50.73 patients per day (median=50) with a mean consultation time of 6.91 ± 4.42 minutes per patient (median=5.5). The mean number of antibiotics prescribed in the previous week was 17.64 ± 24.69 (median=10). As shown in Table 1, the most frequently prescribed antibiotic group was the penicillin/β-lactamase inhibitor combination (48.5%), followed by cephalosporins (17.2%) and quinolones (9.1%). Prescribing rates for macrolides, nitrofurantoin, and tetracyclines were each 1.0%.
Knowledge
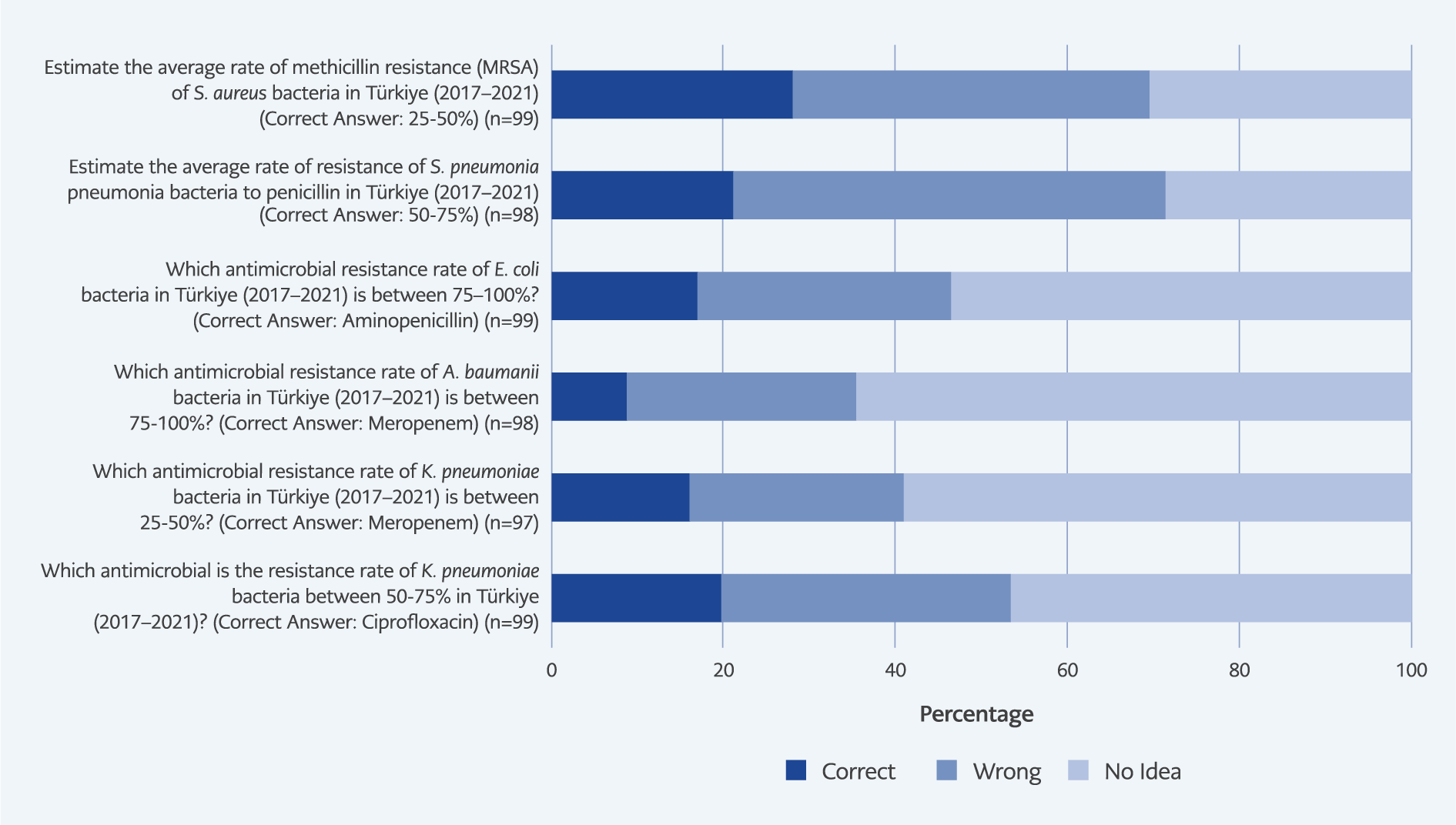
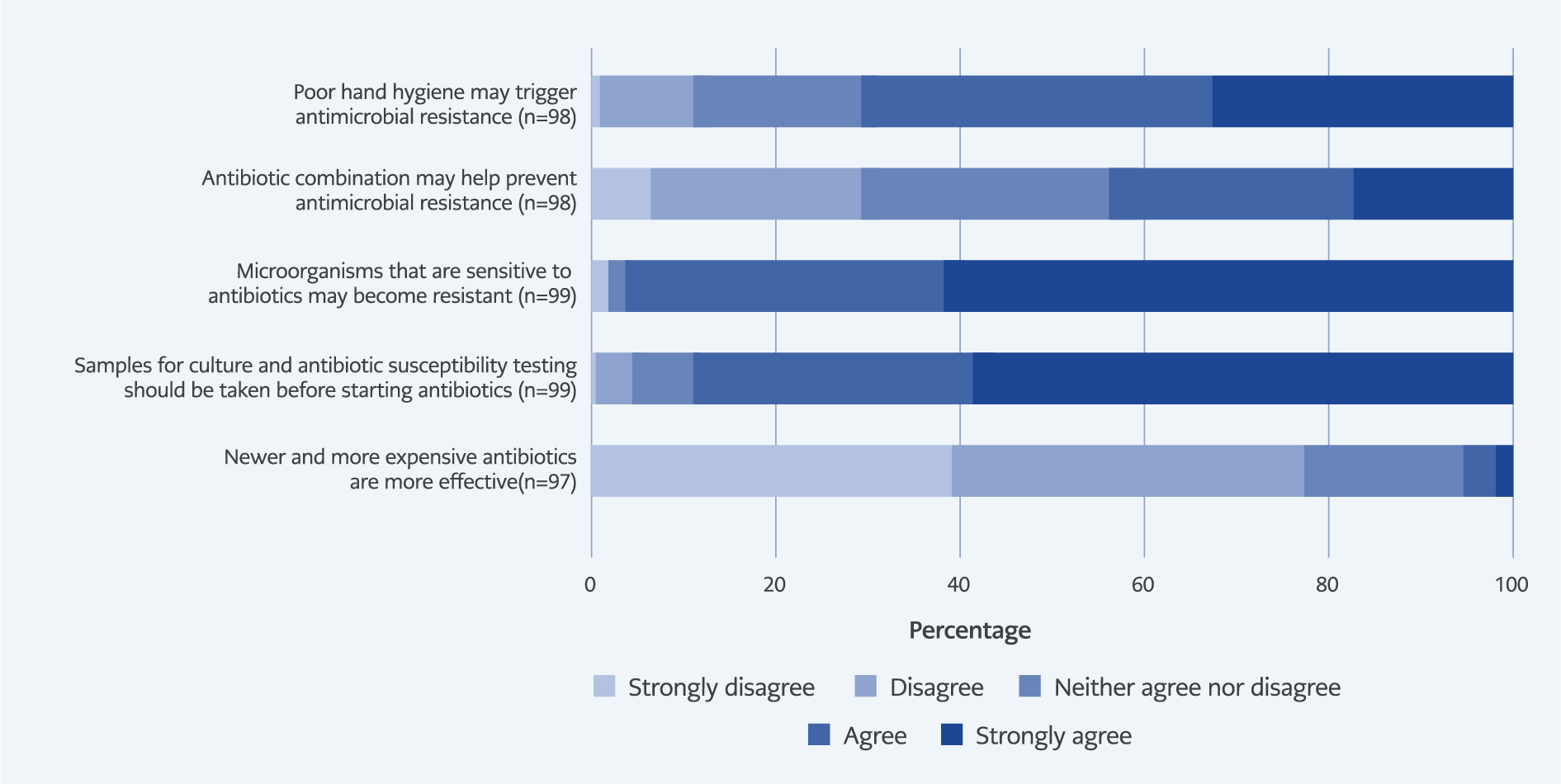
Figure 1B. Percentage of participants’ responses to questions measuring knowledge level of antibiotic resistance.
Knowledge-related responses are presented in Figure 1A–1B. Most physicians (77.3%) disagreed that newer or more expensive antibiotics are superior. A large majority (88.9%) agreed that culture and susceptibility testing should precede antibiotic initiation, and 95.9% acknowledged that antibiotics effective against pathogens could eventually lose their efficacy due to resistance. However, only 43.8% believed that combination therapy prevents resistance, and 70.5% recognized poor hand hygiene as a contributor to AMR. Knowledge of local resistance rates was notably limited, with correct response rates ranging from 9.2% to 28.3%, indicating important informational gaps among participants.
Attitudes
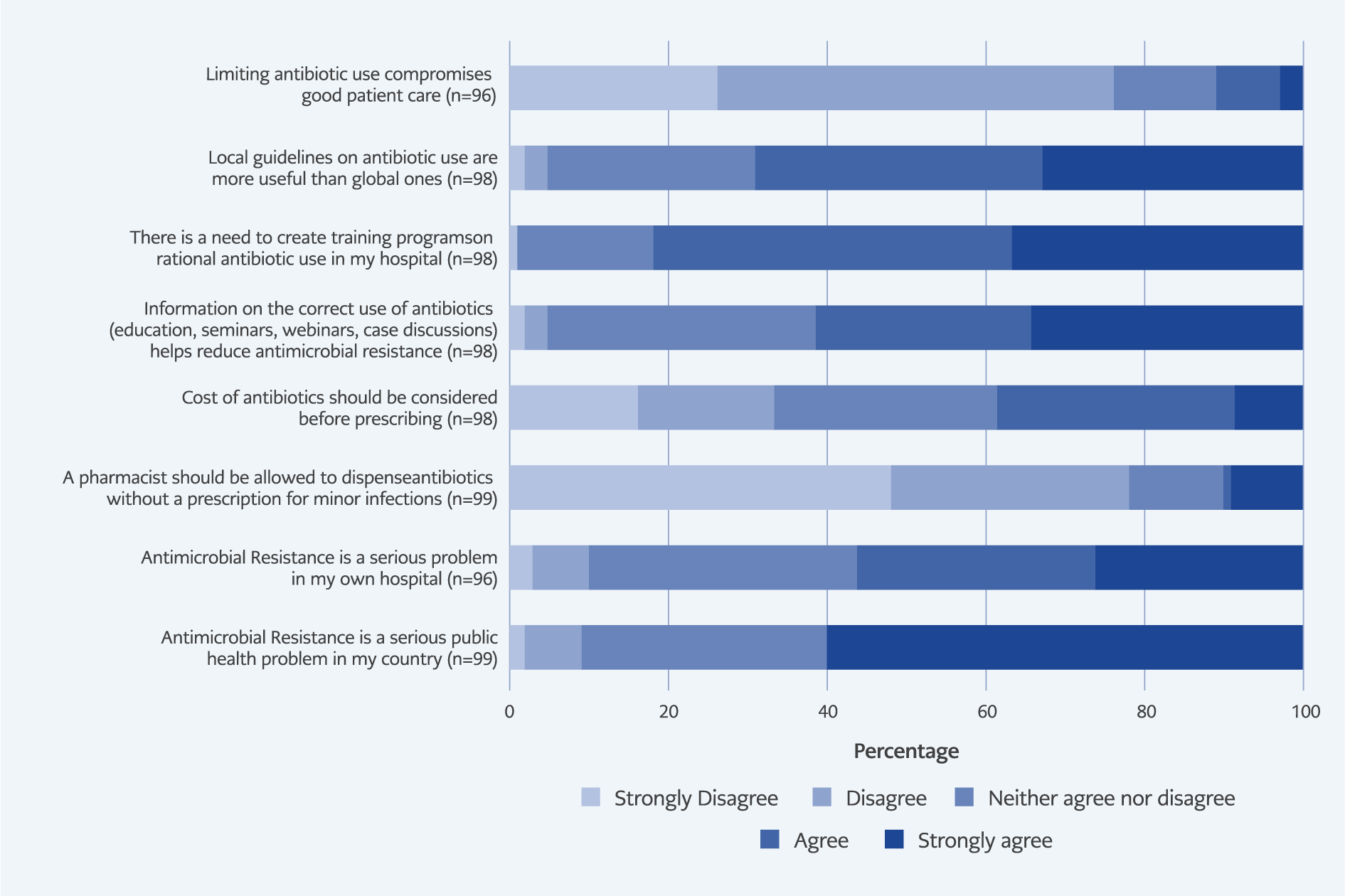
As shown in Figure 2, nearly all respondents (90.9%) regarded AMR as a serious national public health concern, but only 57.1% viewed it as a major issue within their own hospital. A small minority (10.1%) agreed that pharmacists should dispense antibiotics without a prescription for minor infections, and 39.8% believed cost considerations should influence prescribing decisions. The majority (82.7%) expressed a strong need for institutional training on rational antibiotic use, and 69.4% preferred local treatment guidelines over international ones.
Practices
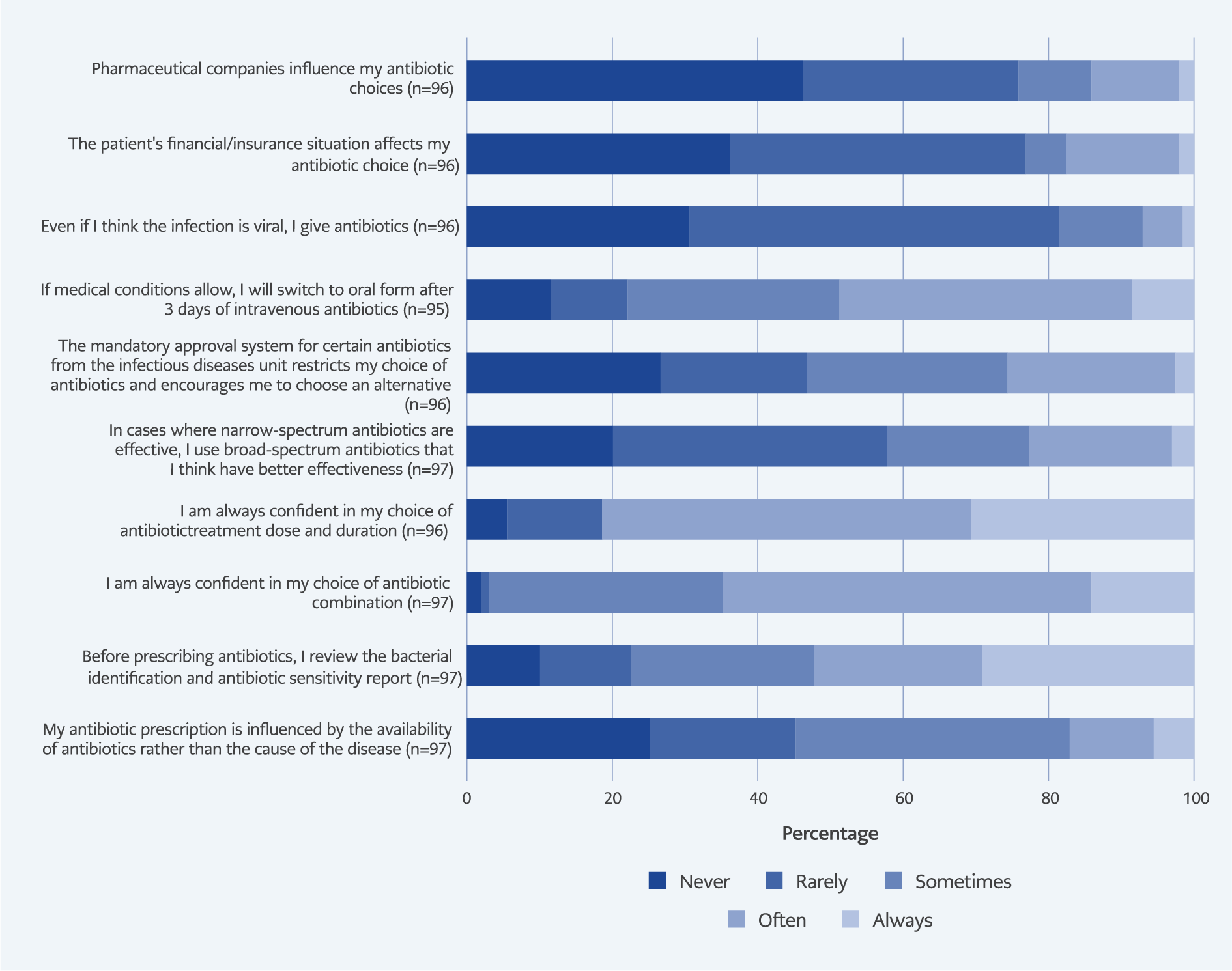
Physicians’ self-reported practices are summarized in Figure 3. Less than half (43.3%) consistently reviewed bacterial identification and susceptibility reports before prescribing, while 54.7% felt confident in selecting appropriate antibiotic combinations. Confidence was higher regarding dosage and treatment duration decisions (80.3%). Mandatory infectious disease approval policies influenced prescribing choices; 23.9% reported modifying prescriptions due to these restrictions. Nearly half (49.5%) stated that they switched from intravenous to oral therapy after three days when appropriate. Nonetheless, 7.3% admitted to prescribing antibiotics despite suspecting viral infections, and 16.7% acknowledged that patients’ financial or insurance conditions affected prescribing behavior. Reported influence from pharmaceutical companies was minimal.
Adequacy of Knowledge, Attitudes, and Practices
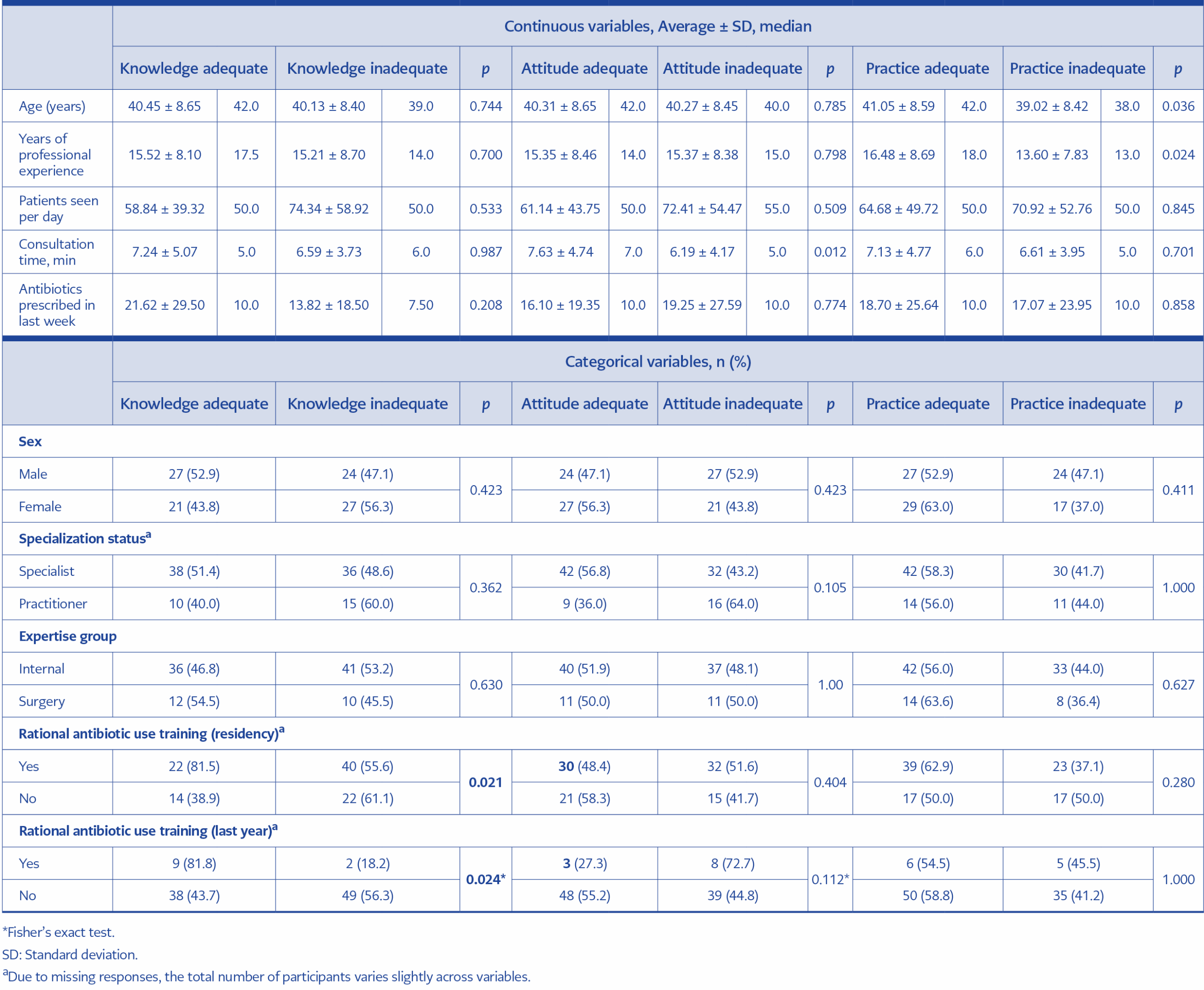
Overall, 48.5% of physicians demonstrated adequate knowledge, 51.5% exhibited adequate attitudes, and 57.7% showed adequate practices. Adequate knowledge was significantly associated with having received training on rational antibiotic use during residency (p=0.021) and within the past year (p=0.024). Adequate attitudes were associated with longer consultation times (p=0.012), while adequate practices were more frequent among older physicians and those with longer professional experience (p=0.036 and p=0.024, respectively) (Table 2).
Discussion
Antimicrobial resistance represents a major and escalating global public health threat, compromising the effectiveness of existing antimicrobial therapies and threatening the sustainability of modern medicine (12,13). Addressing AMR requires a comprehensive understanding of the multifactorial determinants underlying its emergence and spread. Among these, the overuse and misuse of antimicrobials in human medicine remain the predominant drivers. Excessive antibiotic prescribing, inappropriate patient adherence, and the commercial pressures exerted by the pharmaceutical industry continue to amplify this challenge. Consequently, assessing physicians’ knowledge, attitudes, and practices is critical for developing evidence-based interventions to curb AMR.
In the present study, participants’ knowledge levels were moderately adequate. Most physicians (77.3%) rejected the belief that newer or more expensive antibiotics are inherently more effective, and nearly all participants (95.9%) recognized that antimicrobials effective against microorganisms could eventually lose their efficacy due to resistance. Similarly, 88.9% endorsed the importance of performing culture and susceptibility testing prior to initiating therapy. These findings align with reports from Iran and Chinese county hospitals indicating good general awareness of AMR mechanisms among physicians (10,14).
However, participants’ knowledge of local resistance data was notably limited, with correct response rates ranging between 9.2% and 28.3%. Comparable deficits have been observed in studies from France, Scotland, and Egypt, where clinicians demonstrated insufficient familiarity with regional resistance profiles (15,16). This knowledge gap underscores the urgent need to expand access to up-to-date local antibiograms and resistance reports to inform empiric therapy decisions and improve antimicrobial stewardship. Currently, local resistance data in our setting are primarily available through passive surveillance reports in the hospital information system, which are not proactively disseminated to clinicians. This lack of active dissemination likely contributes to the low awareness levels observed.
Perceptions of AMR severity varied between national and institutional contexts. While most participants (90.9%) recognized AMR as a critical national issue, only 57.3% perceived it as a problem within their own hospital. Similar perceptual discrepancies have been documented among physicians in France, Scotland, Peru, and Australia (16–18). Such findings suggest a persistent “externalization bias,” wherein physicians attribute AMR to broader systemic factors rather than local practices. Interventions emphasizing individual and institutional accountability may therefore strengthen the effectiveness of stewardship initiatives. To mitigate this bias, we recommend implementing feedback mechanisms, such as providing physicians with regular, anonymous reports comparing their individual antibiotic prescribing rates against hospital averages. Such comparative data can help internalize the issue by highlighting personal practice gaps.
Poor hand hygiene is a well-documented contributor to the spread of AMR (19). In our study, 70.5% of physicians recognized its role, a finding similar to that reported in Poland, where 74.4% of physicians acknowledged the association between hand hygiene and antimicrobial resistance; however, its importance was somewhat underestimated (20). This reinforces the necessity of continuous infection control training to sustain high compliance with hand hygiene standards. Moreover, socioeconomic factors were shown to influence prescribing behavior: 39.8% of physicians reported considering cost before prescribing, and 16.7% acknowledged that patients’ financial or insurance status affected their prescribing decisions. Comparable associations between economic constraints and antibiotic misuse have been observed in Egypt (15).
The study revealed both strengths and weaknesses in prescribing behavior. Nearly half of the participants (43.3%) routinely reviewed bacterial identification and susceptibility results before prescribing, and 54.7% expressed confidence in selecting appropriate antibiotic combinations. A larger proportion (80.3%) felt confident in determining the correct dosage and duration, reflecting a relatively strong understanding of pharmacologic principles. Nevertheless, 7.3% admitted to prescribing antibiotics despite suspecting viral etiology, highlighting persistent diagnostic uncertainty or patient-driven pressure. However, this relatively low self-reported rate contrasts with broader national trends, suggesting a disconnect between knowledge and practice. A multicenter study by Polat Yuluğ et al. (21) showed that in real-world, high-anxiety scenarios such as COVID-19, irrational prescribing rates exceeded 70%, driven by clinical signs rather than microbiological evidence. This gap is further explained by Kose et al. (22), who found that although experienced physicians were more confident than students, they suffered from a “decay of theoretical knowledge” over time and prioritized defensive medicine out of fear of missing an infection. Similarly, Korkmaz et al. (23) reported that, even when knowledge levels were adequate, external factors, such as patient pressure, forced physicians to compromise on stewardship principles.
The most frequently prescribed antibiotic group was the penicillin/β-lactamase inhibitor combination (48.5%), followed by cephalosporins (17.2%) (Table 1). This preference for first-line agents is encouraging, as such antibiotics are generally less expensive, have narrower spectra, and are associated with fewer adverse effects. Their rational use can help preserve antimicrobial efficacy and limit the spread of resistance. However, 49.5% of respondents reported switching from intravenous to oral therapy after three days, suggesting room for improvement in implementing step-down therapy protocols. To improve adherence to step-down therapy, the formal development of hospital-specific protocols and the integration of electronic clinical decision support systems are recommended to guide physicians during the transition period.
Mandatory infectious disease approval systems affected prescribing decisions for 23.9% of physicians, a lower proportion compared with findings from the United States (11). Although such regulations may be perceived as restrictive, they remain essential for optimizing antibiotic selection and ensuring compliance with stewardship guidelines. Given that only a minority of physicians reported being influenced by these regulations, we suggest expanding the scope of restricted antimicrobials and ensuring stricter enforcement of the mandatory approval process to enhance its impact on prescribing practices.
Importantly, older age and greater professional experience were significantly associated with better antimicrobial practices. This finding supports the hypothesis that, in the absence of regular formal training, physicians rely heavily on practical clinical experience and informal mentorship from senior colleagues to develop appropriate antimicrobial stewardship behaviors. Similar trends have been observed in studies from China and Nepal, where more experienced physicians demonstrated stronger stewardship behaviors, potentially serving as role models for younger colleagues (9,14). Conversely, the narrow age range in the current study may have limited the detection of stronger associations with knowledge and attitude outcomes.
Educational exposure emerged as a critical determinant of AMR-related competencies. Adequate knowledge was significantly associated with having received rational antibiotic use training during both residency and within the past year (p=0.021 and p=0.024, respectively) (Table 2). However, only 11.1% of participants reported receiving such training within the previous year, despite 82.7% expressing a desire for structured educational programs. This stark contrast between the high demand for education and the low rate of actual delivery suggests that hospital management has not sufficiently prioritized or operationalized continuous antimicrobial stewardship training. Bridging this gap requires an institutional shift from voluntary to mandatory and structured educational modules. These findings align with evidence from Norway and Brazil, where antimicrobial stewardship initiatives integrating guideline dissemination, specialist feedback, and continuing medical education improved prescribing behavior and compliance (24,25).
The results emphasize the necessity for ongoing postgraduate training that bridges theoretical and clinical aspects of antimicrobial use. Studies from India, Nepal, and China have similarly recommended periodic refresher programs and institutional stewardship teams to sustain rational prescribing practices (9,14,15,20).
This study’s primary limitation is its single-center design, which may limit generalizability to other healthcare settings. Additionally, self-reported data may be subject to recall and social desirability bias. Nonetheless, the findings provide valuable insight into physicians’ current knowledge, attitudes, and practices regarding AMR in a representative state hospital context.
In conclusion, while physicians in this study exhibited moderate knowledge and generally positive attitudes toward AMR, gaps remain in local resistance awareness, adherence to susceptibility testing, and consistent step-down prescribing practices. Strengthening education, reinforcing stewardship oversight, and promoting local antibiogram use are key strategies to enhance rational antibiotic prescribing and mitigate the growing threat of antimicrobial resistance.