Introduction
The clinical spectrum of COVID-19 varies, ranging from asymptomatic infection to critical illness. The most commonly used tool for the classification of disease severity is the National Institute of Health (NIH) severity scale, which has four categories: mild, moderate, severe, and critical (1). Patients with moderate and severe clinical courses are followed up in wards, while those with the critical disease require intensive care (2).
Although the inefficacy of antibiotics in COVID-19 is well known, their use is very high for various reasons. Among the possible reasons for antibiotic use in COVID-19 are difficulty in excluding a bacterial co-infection, high levels of laboratory inflammation parameters, and chest imaging findings that mimic bacterial infections (3). Due to the excessive use of antibiotics in COVID-19, there has recently been an increased awareness in antimicrobial stewardship programs (4-7). These programs aim to reduce the development of antibiotic resistance, the incidence of adverse effects, and related costs (8, 9).
Although a high rate of antibiotic prescription is a well-known practice in COVID-19, there is little information on the frequency and preferences of antibiotics and the duration of treatment in different clinics. Therefore, in this study, we aimed to determine and compare the rates and duration of empirical antibiotic use between different clinics among patients hospitalized due to COVID-19.
Materials and Methods
Patient Selection
This cross-sectional study was performed in three inpatient clinics of a tertiary referral center, including infectious diseases clinic (IDC), chest diseases clinic (CDC), and internal medicine clinic (IMC). The study included all consecutively hospitalized patients aged 18 years and older with a PCR-confirmed COVID-19 diagnosis between May 30, 2021, and August 30, 2021. A total of 683 patients hospitalized with the presumptive diagnosis of COVID-19 infection were evaluated in terms of eligibility for the study. Seventy patients were excluded due to a negative SARS-CoV-2 PCR test, 15 due to missing data, and seven because of transfer to another hospital. As a result, the data of 581 patients were included in the final analysis.
COVID-19 severity was defined according to the NIH Guidelines and categorized as follows: mild=1, moderate=2, severe=3, and critical=4 (1). Since mild cases were followed up without hospitalization, they were not included in the study. The study group included moderate and severe COVID-19 cases. Laboratory findings obtained on the first day of hospital admission were recorded. COVID-19 treatment was undertaken according to the Republic of Türkiye Ministry of Health Guidelines available during the study period (10). Co-infections treated with antibiotics were diagnosed as community-acquired if the diagnosis was made at the time of or in the first 48 hours of hospital admission, and hospital-acquired if diagnosed ≥48 hours after admission for COVID-19.
Kütahya Health Sciences University Ethics Committee approved the study on October 6, 2021 with the decision number “2021/14-17”.
Data Collection
The demographic data of the patients, admission date, discharge, date of transfer to the intensive care unit/death, and underlying diseases were recorded from the electronic medical records database of the hospital. In addition, disease severity was determined according to NIH, and all antibiotics initiated within the first three days of admission were recorded until the end of treatment. Initiation and subsequent discontinuation of antibiotic therapy were at the physicians’ discretion. The primary outcome measure was the rate of empirical antibiotic use in hospitalized patients with COVID-19. The secondary outcome measures were the comparison of the rates and duration of antibiotic use in these patients between IDC, CDC, and IMC.
Statistical Analysis
The Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) 20.0 (IBM Corp., Armonk, NY, USA). Frequency tables, descriptive statistics, cross-tables, bar graphs, and chi-square test (for independent groups), Levene test, post-hoc: Dunnett T3, and one-way analysis of variance were used in the analysis of the data.
Results
A total of 581 patients were eligible for the study. The median age was 58.40 years, and 310 were women (53.4%) (Table 1). The severity of COVID-19 infection was similar in the three clinics. Co-morbidities included hypertension (39% in CDC, 41% in IDC, and 43.4% in IMC), diabetes mellitus (28% in CDC, 32% in IDC, and 31.2% in IMC), chronic obstructive pulmonary disease (8.5% in CDC, 1.7% in IDC, and 13.2% in IMC), coronary artery disease CDC (4% in CDC, 5% in IDC, and 13.8% in IMC). 13.6% were admitted to the ICU, and in-hospital mortality was 6.3%.
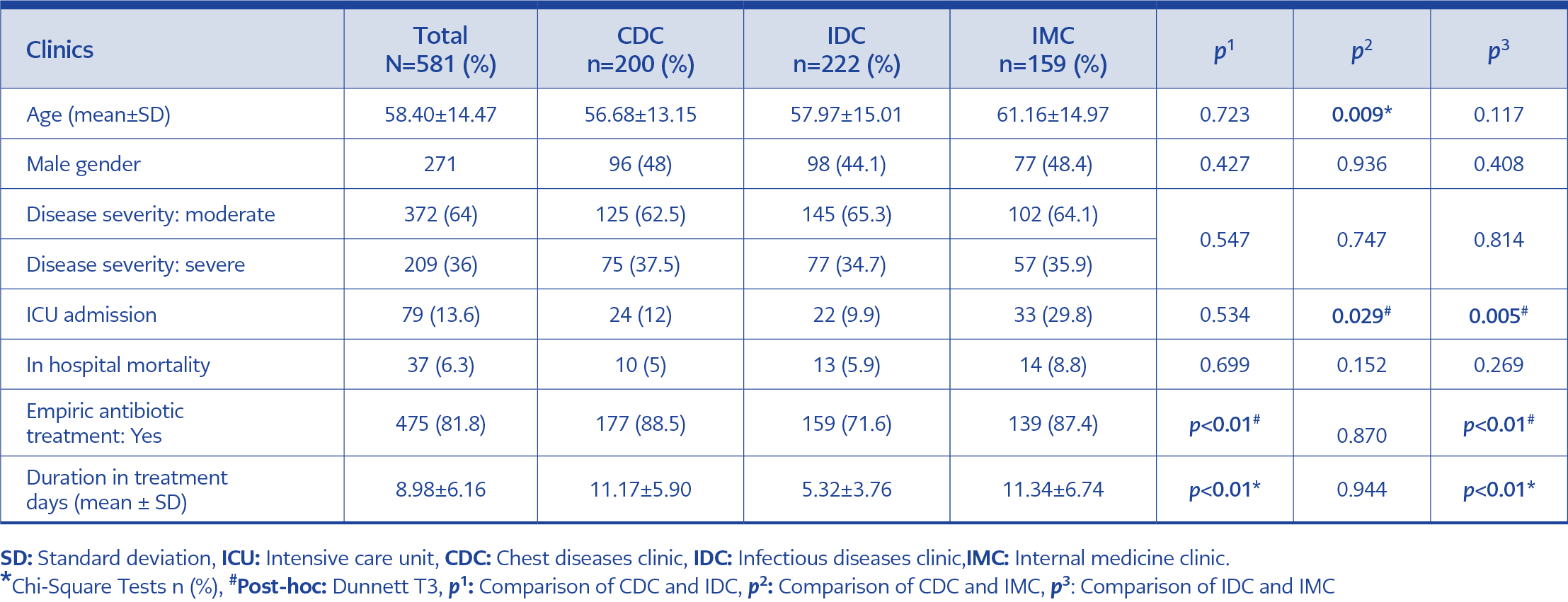
Table 1. Patient characteristics and empiric antibiotic treatment in patients with COVID-19 according to the clinic.
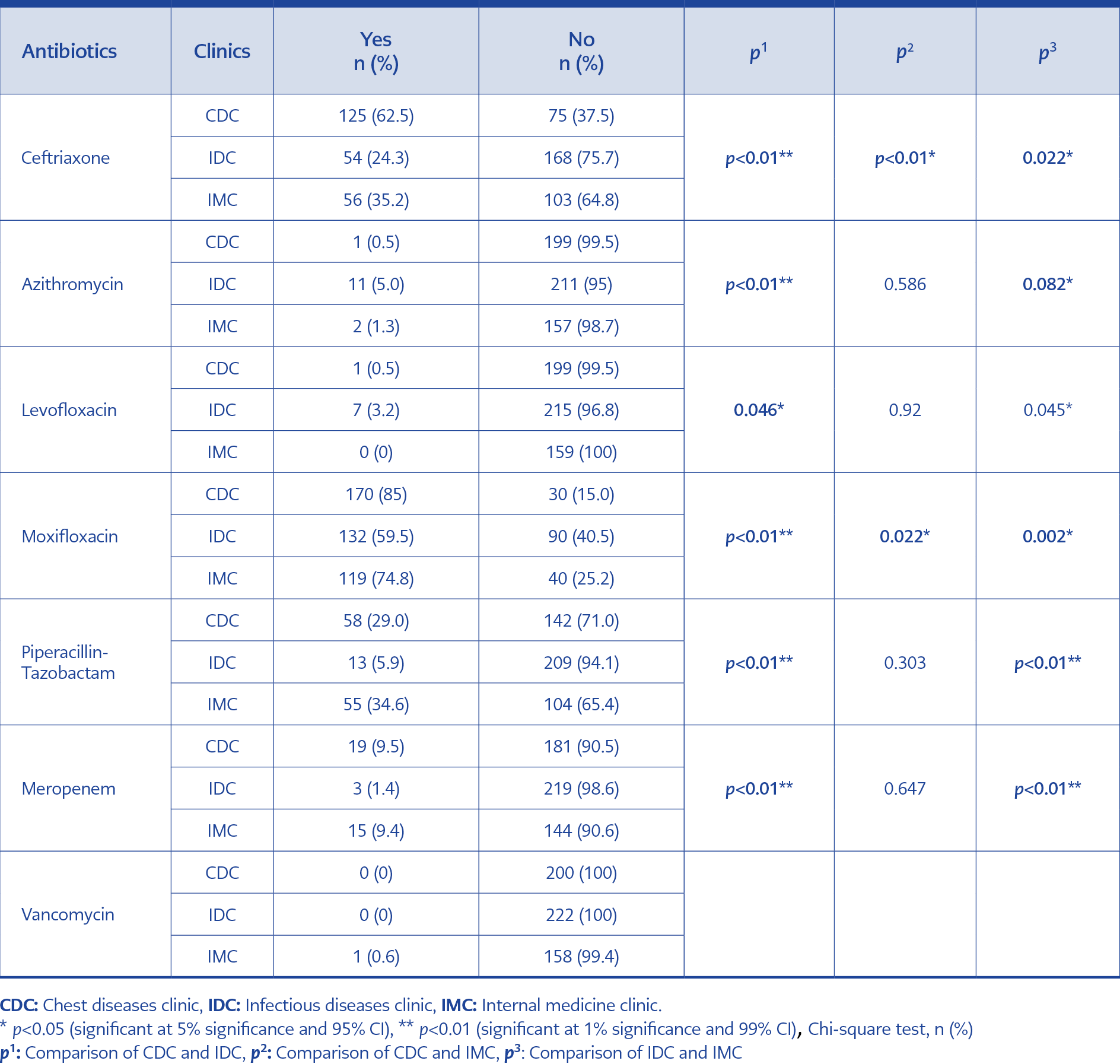
Overall, 475 (81.8%) patients received antibiotics. The number of patients prescribed antibiotics was 159 (71.6%) for IDC, 177 (88.5%) for CDC, and 139 (87.4%) for IMC. Antibiotic use was significantly lower in IDC compared to CDC and IMC (p<0.001 and p<0.001, respectively) (Table 1). The most commonly used antibiotic was moxifloxacin in all groups (72.4%), and the number of patients receiving this antibiotic prescription was 170 (85%) in CDC, 132 (59.5%) in IDC, and 119 (74.8%) in IMC. Moxifloxacin use was significantly lower in IDC compared to CDC and IMC (p<0.001 and p=0.002, respectively) (Table 2).
The number of patients who received ceftriaxone was 125 (62.5%), 54 (24.3%), and 56 (35.2%) in CDC, IDC, and IMC, respectively; this rate was lower in IDC compared to CDC and IMC (p<0.001 and p=0.022, respectively) (Table 2).
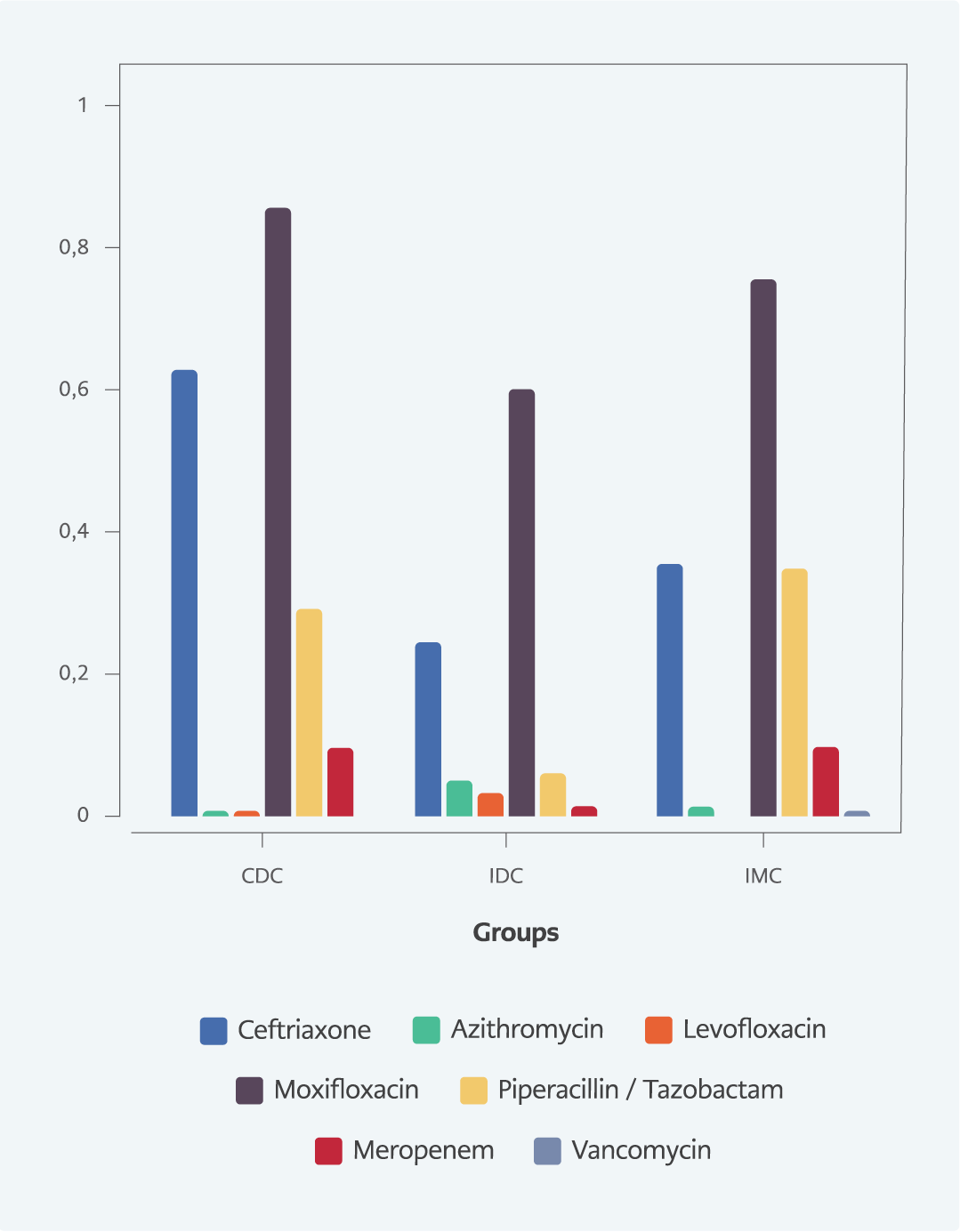
Broader spectrum antibiotics, such as piperacillin-tazobactam, were prescribed for 13 (5.9%) in IDC, 58 (29%) in CDC, and 55 (34.6%) in IMC. Meropenem was given to 3 (1.4%) patients in IDC, 19 (9.5%) in CDC, and 15 (9.4%) in IMC. Piperacillin-tazobactam and meropenem use was significantly lower in IDC compared to CDC and IMC (p<0.001 for all) (Table 2 and Figure 1).
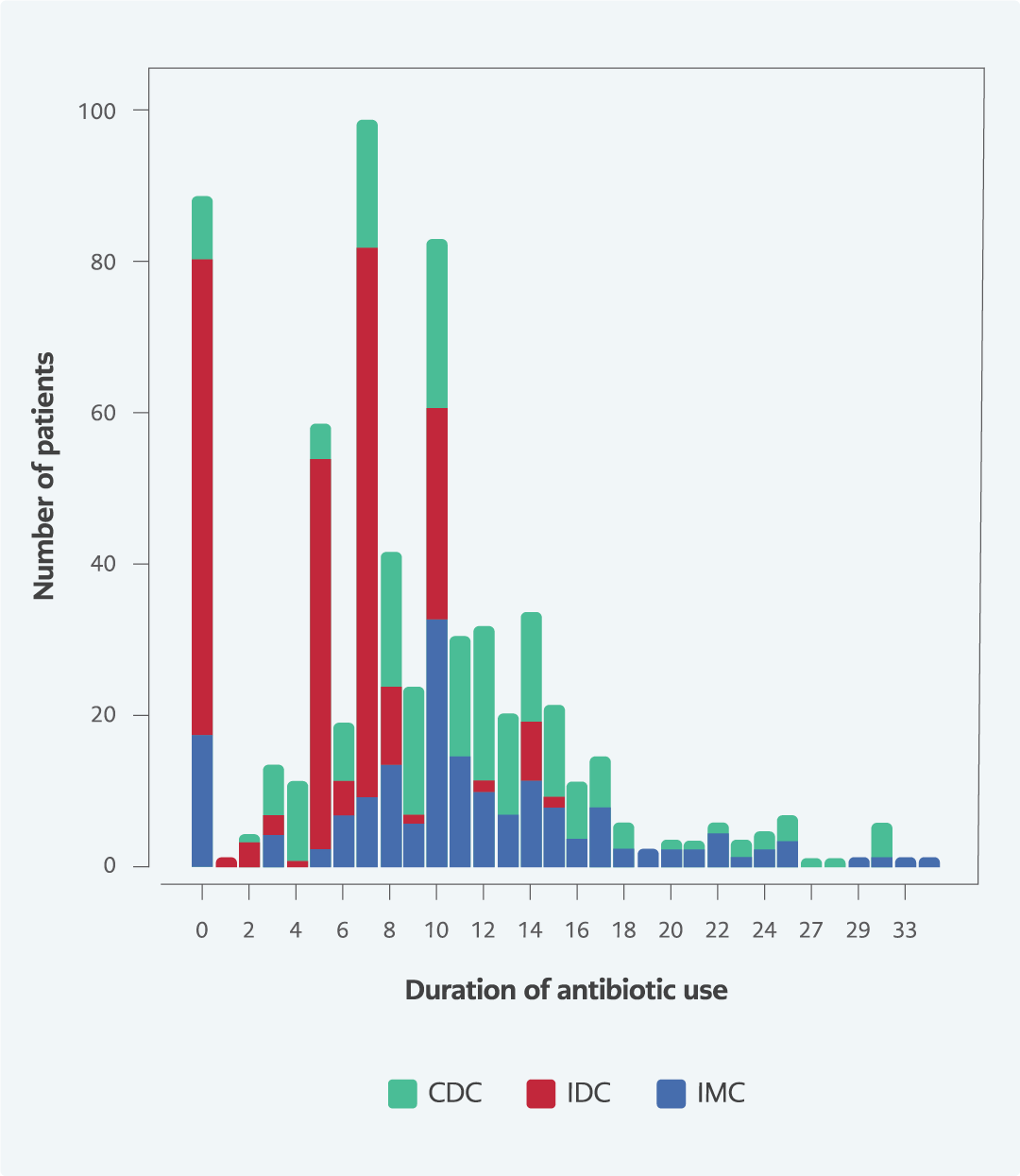
The mean duration of antibiotic treatment was 8.9 days in the whole cohort; 11.1 days for CDC, 11.3 days for IMC, and 5.3 days for IDC. The duration of antibiotic use was significantly lower in IDC compared to CDC and IMC (p<0.001 for both) (Table 2 and Figure 2).
Discussion
In our study, most patients hospitalized with COVID-19 received empirical antibiotics. In a study from Detroit, the frequency of antibiotic use was 69.2% in hospitalized patients with COVID-19 (11). A multicenter study involving 820 patients in Scotland reported a similar frequency of antibiotic use (62.4%) (12). Although the frequency of empirical antibiotic use may differ between studies, they are generally much lower compared to our results. This is compatible with the generally higher empiric antibiotic use in daily clinical practice in Turkey (3, 13, 14).
In the literature, there are limited studies comparing empirical antibiotic use between different pandemic clinics. In a multicenter study from Spain, Estrada et al., reported the rate of antibiotic use as 87.8% in 13,932 patients treated in IMC (15). In another study, the frequency of antibiotic use was reported to be lower in university hospitals than in state hospitals (16). In the current study, the lowest frequency and shortest duration of treatment were observed in IDC. Although the severity of COVID-19 infection in patients admitted to the clinics was similar, the rate of broad-spectrum antibiotic use, such as piperacillin-tazobactam and meropenem, was very high in CDC and IMC compared to IDC. These two antibiotics have required infectious diseases approval for reimbursement since 2003. Unfortunately, due to the excessive workload during the pandemic, case-by-case evaluation of the need for these antibiotics by infectious diseases physicians before approval was not feasible in most instances (17). The lower rate of antibiotic use in IDC may be associated with higher awareness of antimicrobial management among infectious disease specialists.
During the ongoing COVID-19 pandemic, various antibiotics have been used across the world. In a study conducted in Wuhan at the beginning of the pandemic, the most commonly used antibiotic class was fluoroquinolones (18). In an international survey study, the combination of beta-lactam and macrolide was the most commonly used antibiotics in patients with COVID-19 (3). In a multicenter study involving 820 patients in Scotland, the most commonly used antibiotics in hospitalized patients were amoxicillin, doxycycline, and amoxicillin-clavulanic acid. In that study, quinolones and cephalosporins were prescribed infrequently due to their higher adverse effects and Clostridioides difficile superinfection (12). Moxifloxacin, an antibiotic highly effective against community-acquired respiratory pathogens, was the most commonly used antibiotic in our study, which is similar to the results of other reports from Turkey (3).
The efficacy of quinolones against typical and atypical pathogens, their practical use, and possible immunomodulatory effects may have increased their use at the beginning of the pandemic in some countries, including Turkey (19). However, recent evidence suggests a lower frequency of bacterial co-infections (3.5%), especially those caused by atypical pathogens in COVID-19 (20). The Study Group for Antimicrobial Stewardship recently published a guideline that recommends against empiric antibiotic usage in COVID-19. It is also recommended to perform microbiological examinations, such as blood culture analysis and Legionella antigen test in selected patients, due to their limited benefits (4). Thus, antibiotic use has been restricted in recent months by infectious disease committees and specialists (6, 7, 21).
The Scottish Antimicrobial Management Group recommends using narrow-spectrum antibiotics only for short-term (5 days) in treating pneumonia and purulent bronchitis in COVID-19 (5). In a study by Towsend et al., which included 117 patients, the mean duration of antibiotic use was determined as seven days (22). In an international survey study involving clinicians following up patients with a diagnosis of COVID-19, the mean duration of antibiotic use was five days in North America, 5.4 days in England, 6.5 days in Spain, 7.3 days in Slovenia, and 7.6 days in Turkey (3). In our study, the mean duration of antibiotic use was similar to the results reported for Turkey but longer than in other countries.
The strengths of our cross-sectional study are the number of patients included and that it reflected real-life experience. This allowed us to obtain detailed information on how antibiotics were used during the COVID-19 pandemic in different clinics. However, our study has several limitations. First, the results may not be generalized because this is a single-center study with a relatively small sample size in different clinics. Second, there were no specified criteria for microbiological sampling and local antibiotic guidelines for empiric antibiotic treatment in COVID-19 patients. Thus, community-acquired and hospital-acquired co-infection rates could not be specified, and the appropriateness of antibiotic use was not evaluated.
In this study, antibiotic use was determined to be very high in all clinics caring for COVID-19 patients. On the other hand, the lowest frequency and shortest treatment duration of antibiotic use were observed in IDC. Therefore, to reduce unnecessary antibiotic use, antimicrobial management programs should be carried out under the leadership of IDC.