Introduction
Leuconostoc mesenteroides is a Gram-positive, catalase-negative, facultatively anaerobic coccus belonging to the family Leuconostocaceae (formerly classified within Lactobacillaceae in some taxonomies). It is not a common constituent of the human flora and is predominantly found in natural environments such as vegetables, fruits, and dairy products. Although generally regarded as non-pathogenic, Leuconostoc spp. have emerged as opportunistic pathogens in immunocompromised hosts, particularly in individuals with hematological disorders, post-transplantation, or prolonged antibiotic exposure (1–3).
The genus Leuconostoc is intrinsically resistant to vancomycin, a property that distinguishes it from most Gram-positive cocci. This feature poses a diagnostic and therapeutic challenge, as the organism can be misidentified as Streptococcus or Enterococcus, leading to inappropriate empirical therapy (4,5). Reported infections caused by Leuconostoc spp. include bacteremia, endocarditis, meningitis, peritonitis, liver abscess, and pneumonia. Infections have been reported across diverse clinical settings, including immunocompetent adults, neonates, and oncology patients, reflecting their expanding clinical relevance. However, it remains underdiagnosed, particularly in regions where automated systems are not routinely available (4).
In hematology and oncology units, disruption of mucosal integrity, central venous catheterization, and prolonged neutropenia facilitate the translocation of gastrointestinal flora into the bloodstream, predisposing patients to opportunistic infections. Furthermore, the widespread use of glycopeptides and broad-spectrum antibiotics may select for intrinsically vancomycin-resistant organisms such as Leuconostoc spp., Lactobacillus spp., and Pediococcus spp. We report a case of L. mesenteroides bacteremia and liver abscess in a child with very severe aplastic anemia, highlighting the diagnostic challenges and therapeutic implications of this rare but clinically significant pathogen.
Case
A 10-year-old boy with a known diagnosis of very severe aplastic anemia (absolute neutrophil count [ANC] <200 cells/µL) was started on posaconazole prophylaxis along with transfusion support and antibiotics for febrile neutropenia. He was being evaluated for hematopoietic stem cell transplantation (HSCT) as the definitive treatment option. He was admitted with fever and nausea but without localizing signs. There was no history of consuming uncooked food, including milk. As the patient was neutropenic, probiotics were also withheld from the diet. On examination, he had poor oral hygiene and pallor. There was no central venous catheter at that stage. Complete blood count showed anemia, neutropenia, and thrombocytopenia (hemoglobin 6.5 g/dL, ANC
64/µL, and platelet count 21,000 /µL).
After obtaining blood cultures (2 mL peripheral blood collected under adequate aseptic conditions), cefoperazone-sulbactam was initiated empirically (day 1) as per the departmental febrile neutropenia protocol. As the fever persisted beyond five days, amikacin (15 mg/kg/day) and teicoplanin (10 mg/kg/day) were added on day three, along with micafungin (2 mg/kg/day) for antifungal coverage on day five. The BACT/ALERT® automated microbial detection system (bioMérieux, Marcy l’Étoile, France) showed the growth of L. mesenteroides. Once the blood culture flagged positive, subcultures were performed on blood and MacConkey agar, and the cultures incubated at 37°C for 24 hours under aerobic and 5% CO2 conditions. Small, gray, nonhemolytic colonies were observed on blood agar, and Gram staining revealed Gram-positive cocci in pairs and short chains.
Antibiotic susceptibility testing was carried out using the VITEK® 2 Compact system (bioMérieux, Marcy l’Étoile, France) according to the Clinical and Laboratory Standards Institute (CLSI) 2023 criteria. The organism was resistant to vancomycin, clindamycin, ciprofloxacin, and trimethoprim-sulfamethoxazole, but susceptible to amikacin, chloramphenicol, and erythromycin. Given its intrinsic vancomycin resistance, therapy was continued with amikacin as the primary active antibiotic. The patient became afebrile within 72 hours of targeted therapy. Antibiotics were discontinued after 14 days; however, fever recurred within 48 hours of stopping amikacin. Repeat blood cultures at that time were sterile. Amikacin was reintroduced on day 18 and maintained for a further four weeks until evidence of neutrophil recovery, during which the patient remained afebrile, and subsequent cultures were negative. Surveillance chest computed tomography (CT) and echocardiography (ECHO) showed no evidence of infective endocarditis or invasive fungal infection
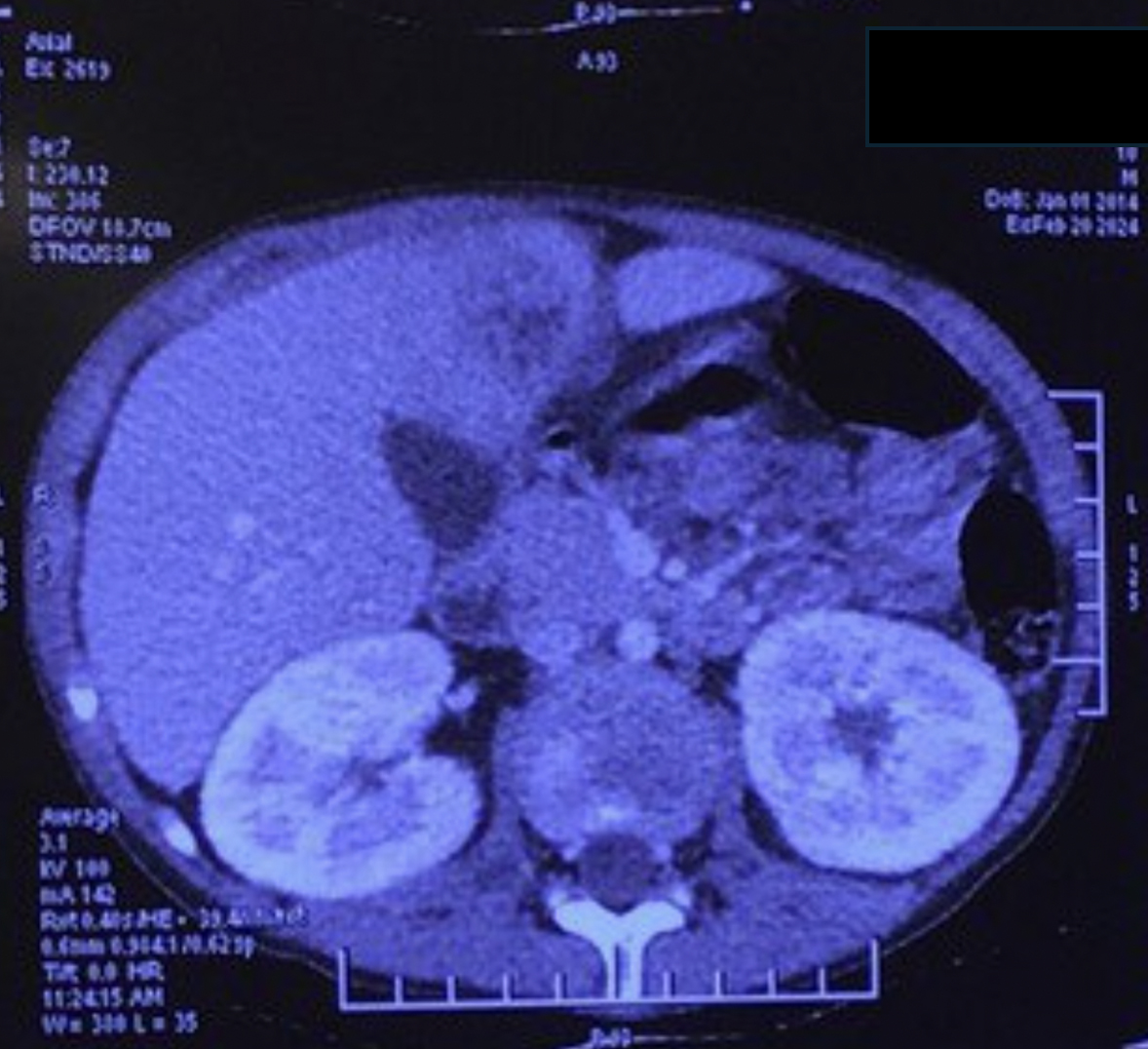
Four weeks later, he developed right upper quadrant abdominal pain associated with hepatomegaly and hepatic tenderness. Contrast-enhanced abdominal CT showed two well-circumscribed space-occupying lesions in segments 7 and 4B of the liver with estimated volumes of 50 mL and 35 mL, respectively (Figure 1). Post-contrast enhancement of the edematous peripheral rim was observed. A partially liquefied liver abscess was suspected; aspiration was not attempted due to refractory thrombocytopenia.
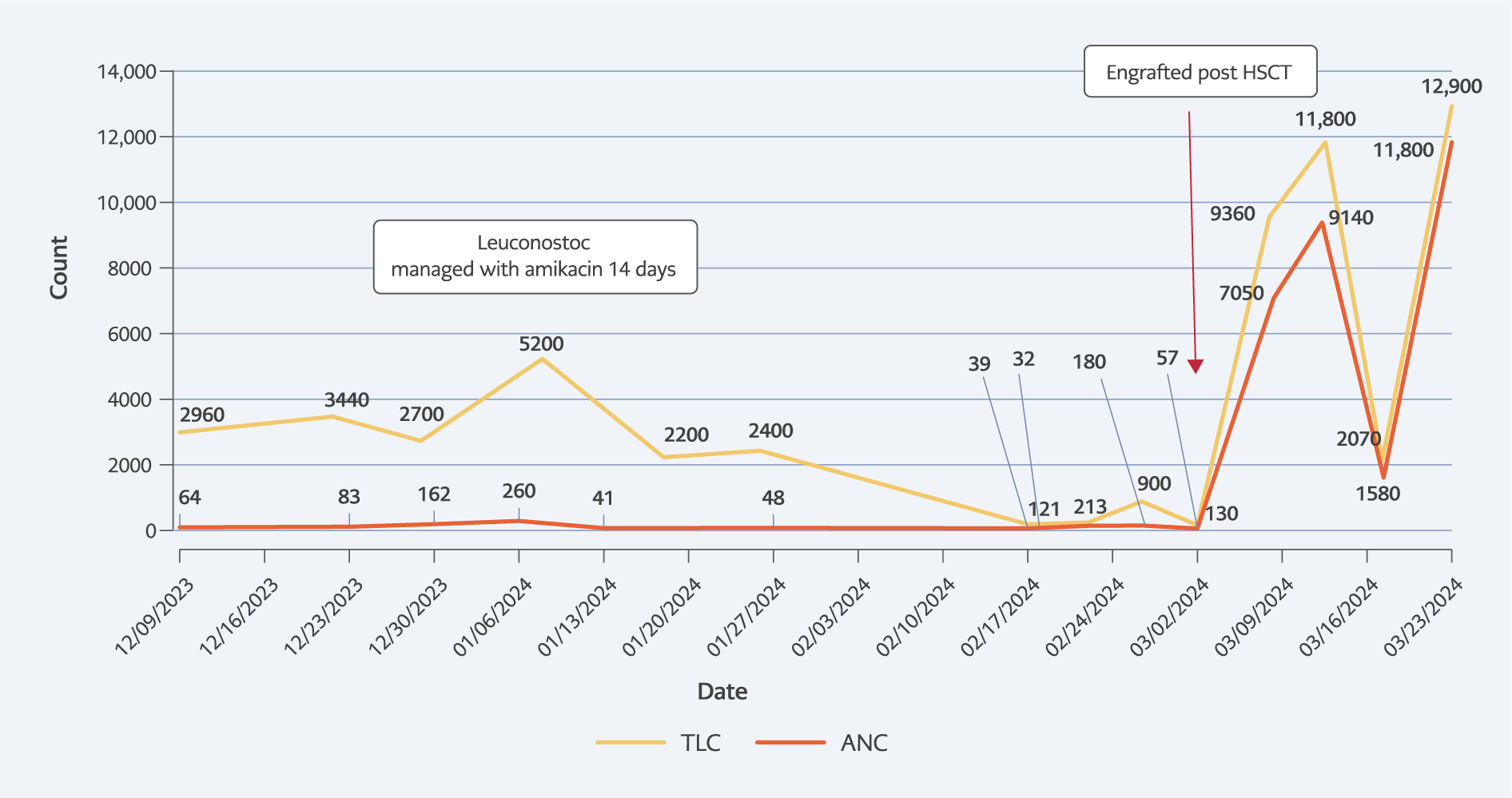
He was started on conditioning with fludarabine, cyclophosphamide, and anti-thymocyte globulin with 4 Gy total body irradiation for haploidentical HSCT (father as the donor) under coverage with the same antibiotics. Neutrophils engrafted on day 12, following which his fever improved. The timeline of infection and absolute neutrophil count is shown in Figure 2. The liver abscess showed clinical resolution with regression of fever and hepatomegaly. On repeat imaging, there was resolution of the abscess in segment 4B and regression in segment 7. The patient, however, developed steroid-resistant acute graft-versus-host disease with refractory diarrhea and died on day 74 post-HSCT.
Discussion
Leuconostoc spp. are uncommon but increasingly recognized causes of invasive infections in vulnerable patients. Our case of L. mesenteroides bacteremia complicated by liver abscess in a child with very severe aplastic anemia illustrates several important features noted in previous reports: the organism’s intrinsic resistance to glycopeptides leading to potential therapeutic failure if misidentified, its predilection for immunocompromised hosts with disrupted mucosal barriers or profound neutropenia, and the central role of host immune recovery in achieving durable clearance of infection.
Intrinsic vancomycin resistance is the defining microbiological characteristic of Leuconostoc spp. and has major therapeutic implications. Empirical regimens for Gram-positive bacteremia in neutropenic or critically ill patients frequently include vancomycin. When Leuconostoc spp. are misidentified as vancomycin-susceptible streptococci or enterococci, patients may receive ineffective therapy, prolonging bacteremia and increasing the risk of metastatic foci such as endocarditis or abscesses, as described in several case reports and outbreaks (1,2). In our patient, initial escalation of therapy included teicoplanin, but definitive identification by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF) and antimicrobial susceptibility testing (AST) guided continuation of amikacin and discontinuation of vancomycin, an approach consistent with recent recommendations that emphasize vancomycin-sparing regimens once Leuconostoc spp. are identified (1,3).
Antimicrobial susceptibility profiles reported in the literature are heterogeneous but frequently demonstrate susceptibility to aminoglycosides, macrolides, and chloramphenicol, with variable susceptibility to β−lactams and carbapenems (4–6). Our isolate’s susceptibility to amikacin, chloramphenicol, and erythromycin, and resistance to vancomycin, clindamycin, ciprofloxacin, and trimethoprim-sulfamethoxazole mirror patterns observed in other recent case reports (1,3,6). These findings underscore the importance of organism-level identification and formal AST rather than empirical inference from Gram stain morphology.
Human infection with Leuconostoc was first reported in 1985 in a patient with systemic lupus erythematosus (5). Since then, numerous cases of bacteremia and focal infections, including meningitis, brain and liver abscesses, pulmonary infections, and nosocomial infections, have been described (5–9). Hospital outbreaks associated with Leuconostoc have also been reported, particularly in relation to improper handling or preparation of parenteral nutrition (10). Infections have additionally been documented in patients with hematological malignancies and neutropenia, including lymphoblastic and myeloid leukemias in adults (6–8).
Host factors strongly influence outcomes. Most severe Leuconostoc infections occur in patients with hematological malignancies, profound neutropenia, central venous catheters, or prolonged exposure to broad-spectrum antibiotics (4,6). Our patient’s persistent neutropenia likely enabled gastrointestinal translocation and hematogenous seeding of the liver—similar to mechanisms proposed in patients with short bowel syndrome and leukemia (4,7). Notably, multiple reports have shown that recovery of neutrophil counts, rather than antibiotic therapy alone, correlates with infection resolution (3,4,7). In our case, liver abscesses regressed only after neutrophil engraftment following HSCT, reaffirming the critical role of host immune reconstitution.
Diagnostic advances have improved recognition of Leuconostoc. Conventional phenotypic methods may misidentify these organisms as streptococci or enterococci because of overlapping biochemical characteristics. The introduction of MALDI-TOF mass spectrometry and molecular assays has markedly improved accuracy and speed of identification, enabling timely antimicrobial adjustments (11,12). Recent studies suggest that the apparent increase in Leuconostoc detection likely reflects improved laboratory recognition rather than a true rise in incidence (12). In centers lacking MALDI-TOF, reference laboratory confirmation by molecular testing is advisable whenever vancomycin-resistant Gram-positive cocci are isolated.
Management of focal complications such as liver abscesses presents further challenges in neutropenic patients. Although percutaneous drainage is standard for pyogenic abscesses, thrombocytopenia or coagulopathy often preclude invasive procedures. Several pediatric and adult case reports describe successful conservative management using prolonged targeted antibiotic therapy alongside immune recovery (4,6). In our case, aspiration was deferred due to refractory thrombocytopenia, and the abscesses regressed following neutrophil recovery—supporting a conservative, individualized approach.
Reported mortality rates vary widely and are largely influenced by the severity of underlying disease and delays in appropriate antimicrobial therapy. In hematologic cohorts, mortality remains high when diagnosis is delayed, or vancomycin is continued empirically despite resistance (6,10). This highlights two key management principles: i) rapid, species-level identification and AST are essential to avoid ineffective glycopeptide therapy; and ii) restoration of neutrophil counts is pivotal to clinical recovery.
Current evidence remains limited to single-case reports and small case series, precluding standardized treatment recommendations. Prospective multicenter data are needed to define optimal therapy duration, the role of combination regimens (e.g., aminoglycoside plus β-lactam), and the timing of drainage procedures for focal infections.
In summary, L. mesenteroides should be considered in immunocompromised patients with vancomycin-resistant Gram-positive bacteremia unresponsive to standard therapy. Early recognition through MALDI-TOF or molecular diagnostics, targeted antibiotic therapy, and timely immune recovery remain central to achieving favorable outcomes.
Conclusion
Leuconostoc mesenteroides is an uncommon but clinically significant opportunistic pathogen, particularly in immunocompromised hosts such as those with aplastic anemia. Its intrinsic resistance to vancomycin necessitates accurate and timely genus- or species-level identification to avoid ineffective empirical therapy. This case represents, to our knowledge, the first reported instance of L. mesenteroides sepsis with liver abscess in a child with very severe aplastic anemia undergoing HSCT, underscores three critical principles:
- the need for early microbiological confirmation using automated identification systems
- the importance of individualized, susceptibility-guided antimicrobial therapy
- the pivotal role of neutrophil recovery in infection resolution.
Clinicians should maintain a high index of suspicion for Leuconostoc spp. in vancomycin-resistant Gram-positive infections that fail to respond to standard therapy. Continued documentation of such rare infections will help refine treatment strategies and guide future evidence-based recommendations.