Introduction
International collaborative consultations are significantly beneficial in combatting pandemics, although such opportunities were quite limited during the COVID-19 pandemic. We had the chance to convene with the experts to share experiences and review the practices in Kosova. Two days of expert consultation hybrid meeting were performed in Prishtina on 15-16 September of 2021 with the participation of all associated physicians in Kosova from the hospitals of Prishtina, Gjilan, Mitrovice, Gjakove, Peje, Vushtri, and Prizren. The face-to-face meeting was performed in Prishtina, whereas the physicians from all other provinces joined via telemedicine.
In the meeting, epidemiological features, the progress of the COVID-19 pandemic in the world, and the management of the cases were discussed. Antiviral, immunomodulatory drugs, and anticoagulation treatments among outpatients and inpatients were presented, and management of COVID-19 cases in the intensive care unit (ICU) was discussed.
Case management in Kosova
Antivirals
Hydroxychloroquine was used at the beginning of the pandemic, and its use was stopped soon after the declaration of the World Health Organization (WHO) in the December of 2020 (1).
Favipiravir is being used in Kosova for mild COVID-19 cases. The effectiveness of favipiravir has not been shown yet (2), and its place in COVID-19 should be reconsidered based on the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) therapeutic guideline (3) and Japanese guideline (4).
Remdesivir is accepted as an effective antiviral drug by the Infectious Diseases Society of America (IDSA) (5), but the living guideline of WHO was against its use in “severe” cases. It has been recommended to be used among “severe” cases by IDSA; however, based on the recent studies, it seems to be effective among mild to moderate cases with high-risk factors (6). If it becomes available, it might be used in mild to moderate cases with high-risk factors (such as older ages, obesity, diabetes mellitus, malignancy).
Molnupiravir was approved by US Food and Drug Administration (FDA), and European Medicines Agency (EMA) based on the ongoing trial showed that it decreases hospital admission or death among high-risk and unvaccinated patients compared to placebo (7). It seems to be a promising drug; however future studies should be waited (8). If it becomes available, it might be another option in the early stages of cases with COVID-19.
Nirmatrelvir/ritonavir (Paxlovid™) is a promising drug that was approved by UK Medicines and Healthcare Products Regulatory Agency (MHRA) based on a study that showed hospital admission or death is 87% lower in the nirmatrelvir/ritonavir group than in the placebo (9).
Neutralizing monoclonal antibodies such as casirivimab and imdevimab were suggested by WHO (10), IDSA (5), and ESCMID (3) guidelines, but they were not in use in Kosova yet. However, the use of monoclonal antibodies became limited in the omicron variant by the end of 2021.
Immunomodulator Drugs
The effectiveness of corticosteroids was demonstrated in July 2020 and are being used widely in severe and critical cases (11). Dexamethasone 6 mg/day is the usual dose, and equivalent corticosteroid agents such as methylprednisolone or prednisolone might also be used instead of dexamethasone (3). It was underlined that corticosteroids should never be used if the patient is not hypoxic.
Tocilizumab, which is an interleukin-6 monoclonal receptor blocker is used as an effective option for severe and critical cases (12). Anakinra, an interleukin-1 blocker was not available in Kosova. If the risk factors were considered to be at a high level, tocilizumab should be used with dexamethasone concomitantly, whereas if the risk factors were evaluated at a lower level, dexamethasone could be started initially, and depending on the progress of the patient, tocilizumab might be added within 48 hours. Tocilizumab has been used both among severe and critical cases in Kosova.
Overuse of Antibiotics
We observed that antibiotics are widely used in moderate and severe COVID-19 patients. The most commonly used antibiotics were ceftriaxone, piperacillin-tazobactam, imipenem, levofloxacin, and vancomycin. The administration of corticosteroids and/or IL-6 blockers does not require routine antibiotic use. The use of antibiotics should be evaluated based on suspicion of bacterial infection. Procalcitonin, a good indicator of bacterial infection, might be used as a diagnostic tool. However, it should be noted that secondary bacterial infections and fungi infections such as aspergillosis and mucormycosis in ICU have been reported from many countries (13). An antimicrobial stewardship program should be implemented.
Infection Control Measures
The hospital infection control committee (ICC) has been monitoring cases of COVID-19 in the hospitals. The ICC not only monitored the disinfection process of the COVID-19 but also followed up main infection control measures. The hospital staff completed their training program about infection control measures for COVID-19 before the outbreak started in the country. The current guidelines about infection control practices might be helpful to prevent nosocomial COVID-19. The vaccination of healthcare workers was found to be sufficient that was started in March 2021. Pfizer/Biontech and Astra Zeneca vaccines have been used mainly.
Management of Critical Patients
Ninety-eight cases were treated in the central ICU during the period August-September 2020. Initially, they were connected to the respirator with the continuous positive airway pressure / non-invasive ventilation (CPAP/NIV) mode. The invasive mechanical ventilation was implemented based on the response to the non-invasive mechanical ventilation, imaging and laboratory data. The most commonly used anticoagulant was enoxaparin (80%) that is followed by heparin (20%). Close monitoring of the patients in ICU, decreasing patient/nurse ratio, and implementation of prone position if PaO2/FiO2 is less than 160 mmHg were recommended to improve outcomes.
Future suggestions
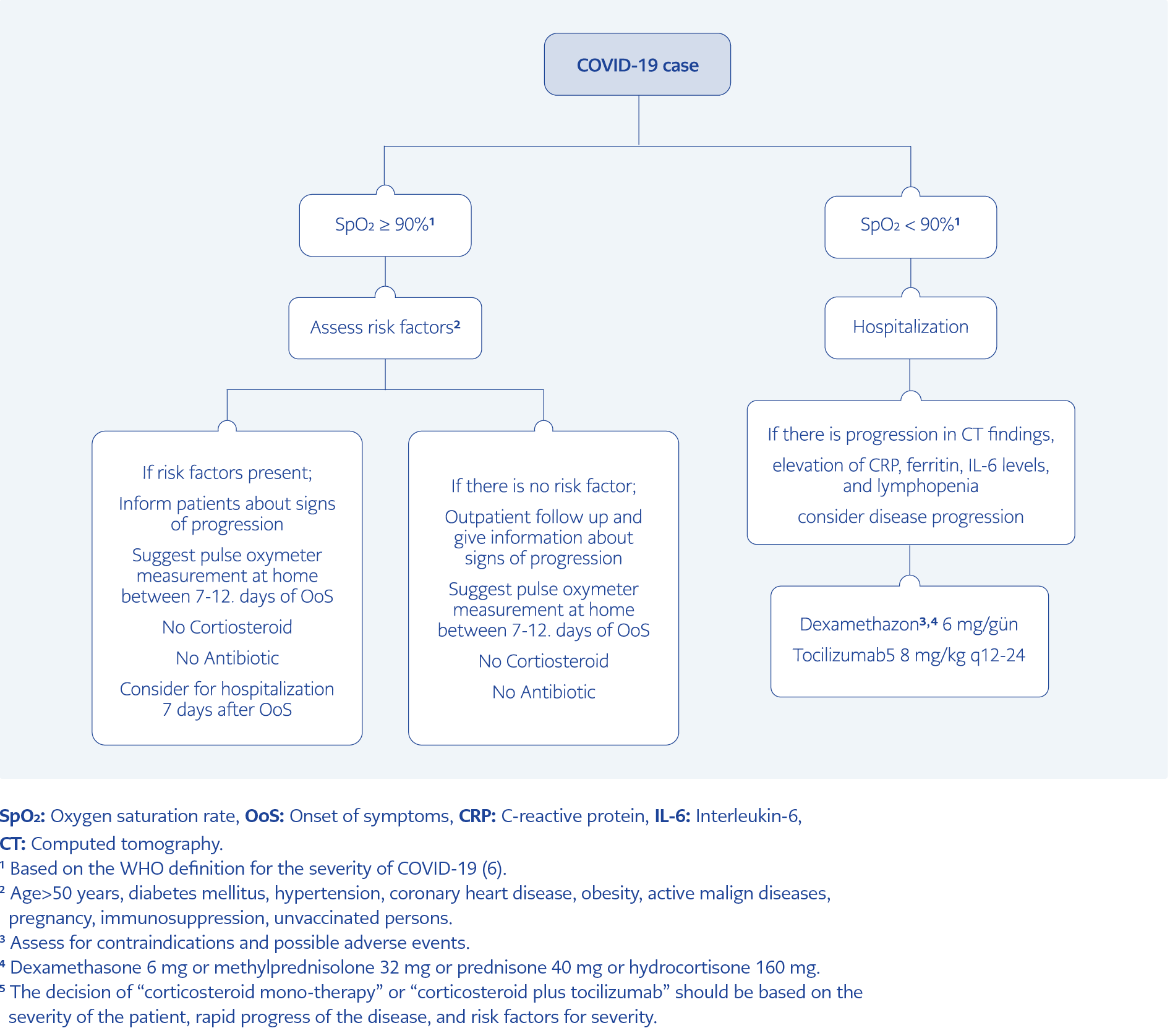
We suggested an algorithm for case management in Kosova (Figure 1). The availability of antiviral agents against SARS CoV-2, including monoclonal antibodies, and promising drugs such as molnupiravir, nirmatrelvir/ritonavir, and remdesivir could be beneficial in decreasing, hospital admission and adverse outcomes. The overuse of radiologic imaging should be controlled. Antimicrobial stewardship should be implemented to decrease unnecessary antibiotic use.