Introduction
Clinically COVID-19 can be observed with mild symptoms such as fever, malaise, cough, and also severe symptoms such as acute respiratory distress syndrome (ARDS) that may require respiratory support in up to 25% of the patients (1). COVID-19 has a high mortality rate, in hospitalized patients (2).
The virus-borne cytopathic effects and the escape from the host’s immune response are thought to be effective in the severity of the disease (3). The overproduction of pro-inflammatory cytokines has been observed as cytokine storms in COVID-19 patients, undergoing a severe clinical course (4). COVID-19-related multiple-organ failure and ARDS are accepted as the results of cytokine storms. Suppressing the cytokine storm is beneficial for COVID-19 patients. TCZ is a monoclonal antibody that blocks specifically Interleukin 6 (IL-6), which takes a major role in this inflammatory response (3). In recent studies, TCZ was shown to be effective in COVID-19 (5-7). In June 2021 the U.S. Food and Drug Administration (FDA) issued an emergency use authorization for TCZ for the treatment of hospitalized patients who are receiving systemic corticosteroids and require supplemental oxygen.
We aimed to describe the appropriate use of TCZ in severe to critical cases with COVID-19 pneumonia in the early phase of the pandemic.
Materials and Methods
This single center, retrospective, observational study was conducted among the patients with COVID-19 who were hospitalized and received TCZ between April 01, 2020, and June 30, 2020, in Health Sciences University Ümraniye Research and Training Hospital İstanbul, Turkey. All the patients were treated according to the national COVID-19 treatment guideline. During the study period, the standard treatment suggested by the Ministry of Health of Turkey included hydroxychloroquine and/or favipiravir (8).
TCZ was administered to the patient when the cytokine storm was suspected. The need for oxygen supply, radiological worsening, increased levels of C-reactive protein (CRP), lactate dehydrogenase (LDH), ferritin, D-dimer and lymphopenia were the predictors for cytokine storms according to Chinese guidelines. (9). The initial dose of TCZ was 8 mg/kg. If the first dose was ineffective, one more dose was given at least 12 h later (9).
We included patients who were 18 years of age or older with positive SARS-CoV-2 polymerase chain reaction (PCR) in nasopharyngeal swab samples. We obtained the demographic characteristics, comorbidities, clinical characteristics of the infection, and patients’ 28th day mortality data from the medical records of the hospitalised patients, and then through direct phone calls after their discharge.
Severe patients met any of the following criteria: 1) Respiratory rate ≥ 30 breaths/min; 2) SpO2 ≤ 93% while breathing room air; 3) PaO2/FiO2 ≤ 300 mmHg. A critical case additionally met at least one of the following criteria: 1) Respiratory failure requiring mechanical ventilation; (2) Shock; (3) Combined with other organ failures, need to be admitted to intensive care unit (ICU) (8).
Patients who needed to be referred to an external center at the time of admission, transferred from an external ICU to our hospital, individuals <18 years of age, COVID PCR negative patients and patients who died within the first 48 hours of hospitalization were excluded from the study.
The ethics committee of the Health Sciences University of Turkey, Ümraniye Research and Training Hospital approved the study on April 27, 2020, with the decision number 140.
Statistical Analysis
Categorical data were compared by using the chi-square test. For a continuous variable, the Kruskal-Wallis test was used. For the predictors of 28-day fatality, logistic regression was performed. Gender, age, number of comorbidities, lymphocyte count, CRP value, initiation of TCZ in the ICU, and an additional dose of TCZ were included in the multivariate model. STATA V.15.0 (Stata Corporation, College Station) was used for statistical analysis and statistical significance was set as p<0.05.
Results
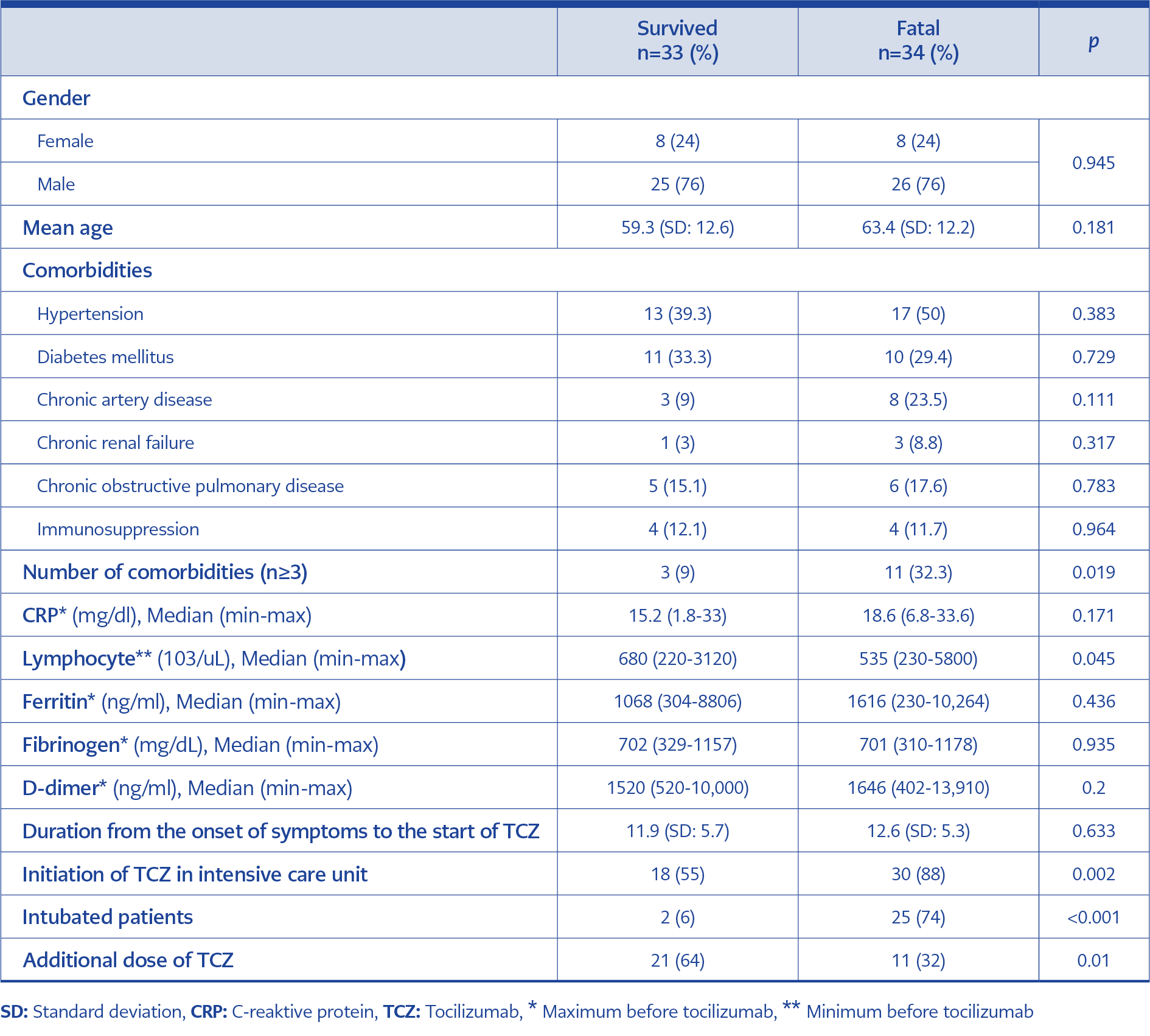
Among the 67 patients included in this study, 51 (76%) were male, 16 (24%) were female, and the mean age was 61.35±12.4. At the time of admission to the hospital, 17 (25%) patients were classified as mild, 27 (40%) moderate, and 23 (35%) severe clinical classification. The most common comorbidity was hypertension (30; 44%) followed by diabetes mellitus (22; 33%), chronic artery disease (11; 16%) and chronic obstructive pulmonary disease (11; 16%). TCZ was initiated in 48 (72%) of the patients in the ICU. Twenty-seven (40%) of the patients were intubated when they received TCZ. An additional dose of TCZ was received for 33 (49%) of the patients. Among all patients, 34 (%51) had died in 28 days follow up.
The survived and fatal cases were compared. There was no difference in the gender, age, incidence of hypertension (39.3% versus 50%; p<0.383), diabetes mellitus (33.3% versus 29.4%; p<0.729), chronic artery disease (9% versus 23.5%; p<0.111), chronic renal failure (3% versus 8.8%; p<0.317), chronic obstructive pulmonary disease (15.1% versus 17.6 %; p<0.783), and immunosuppressive disease (12.1% versus 11.7%; p<0.964) between the survived and fatal groups (Table 1).
In the fatal group, the percentage of the number of comorbidities ≥3 (32.3% versus 9%; p<0.019), minimum lymphocyte count before initiation of TCZ ( 535 versus 680; p<0.045), initiation of TCZ in ICU (30 versus 18; p<0.02), initiation of TCZ while intubated (25 versus 2; p<0.001), and not receiving an additional dose of TCZ (11 versus 2; p<0.01) were significantly more commonly compared to survived group (Table 1).
There was no statistical difference in the maximum CRP count before initiation of TCZ (18.6 versus 15.2 (p<0.17), maximum ferritin count before initiation of TCZ (1616 versus 1068 (p<0.43), maximum fibrinogen count before initiation of TCZ (701 versus 702 (p<0.93), maximum D-Dimer count before initiation of TCZ (1646 versus 1520 (p<0.2), and duration from the onset of symptoms to the start of TCZ (12.6 versus 11.9 days (p<0.63) between the survived and fatal group (Table 1).
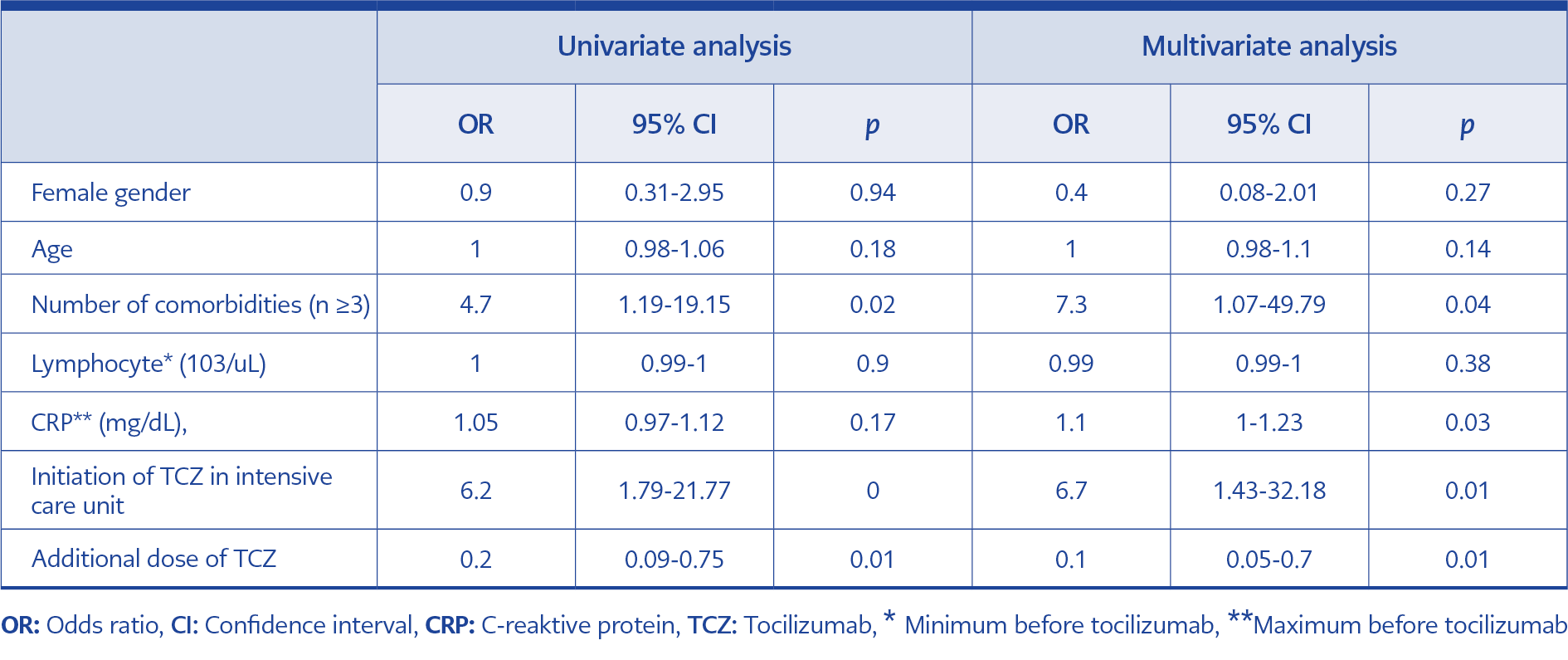
A logistic regression model identified four factors associated with 28-day fatality; ≥3 number of comorbidities (odds ratio [OR], 7.3; 95% confidence interval [CI], 1.07 to 49.79), high levels of CRP before initiation of TCZ (OR, 1.1; 95% CI, 1 to 1.23), initiation of TCZ in ICU (OR, 6.7; 95% CI, 1.43 to 32.18), and not receiving an additional dose of TCZ (OR, 0.1; 95% CI, 0.05 to 0.7) (Table 2).
Discussion
COVID-19 induced cytokine storms are thought to be a primary factor in the mortality of COVID-19 patients, and IL-6, the human body’s own cytokine plays a key role in the COVID-19 induced cytokine storms. Recent studies have shown the efficacy of TCZ as an IL-6 inhibitor and it has been approved for emergency use authorization by the FDA.
In this study, we retrospectively observed 67 patients with severe and critical COVID-19 who received TCZ, 76% of our patients were male. The mean age was 61. Most of the patient was severely ill.
It was observed that older adults, males and patients having chronic comorbidities have a higher risk of death (10, 11). In a systematic review, the fatality of patients showed an association with comorbidity (12). In a nationwide population-based COVID-19 study, older age and multimorbidity were strongly associated with hospitalization and death (13). Similarly, we found that the number of comorbid diseases increased the 28-day fatality in multivariate analysis, but we did not find an association between age and gender.
In a recent randomized controlled trial, death from any cause by day 28 occurred in 10.4% of the patients in the TCZ group (7). Our mortality rate was found 51%. In our study, TCZ was initiated in 72% of the patients in the ICU and 40% were intubated. In early of pandemic, insufficient data on the efficacy and safety of TCZ, it was commonly used in ICU, where most of the cases were intubated. Our high rate of fatality could be associated with the high rate of use of TCZ in the ICU.
Keske et al. reported that earlier use of TCZ in COVID-19 infection was beneficial for survival, length of hospitalization and duration of oxygen support (14). In our study, among all patients, 55% of survivors received TCZ on the ward, 6% intubated, 12% of fatal received TCZ on the ward, and 74% intubated. The current study observed that administration of TCZ to the patient in the ICU was associated with mortality. Early initiation of TCZ when cytokine storm is suspected is effective on survival.
CRP is used as a biomarker to measure the clinical state of COVID-19 patients. It is associated with a hyperinflammatory response. Campochiaro et al. found that patients who received TCZ with the CRP levels beginning at 128 mg/L had positive clinical outcomes, and patients having CRP levels over 186mg/L were nonresponsive as well (15). We found that high levels of CRP before initiation of TCZ were associated with 28-day fatality in multivariate analysis. This study may help to identify the parameters for the target population who could benefit from TCZ.
Due to progressive respiratory worsening 32 patients were given additional doses of TCZ. In the multivariate analysis, not receiving additional doses of TCZ to the patients was associated with death. Luo et al. found a significant correlation between additional dose and positive clinical response (16).
Our research was a single-center observational study with a limited number of patients, in the early period of the pandemic, before the efficacy of TCZ was demonstrated by controlled randomized studies. TCZ was administered to the patient when the cytokine storm was suspected. We could not measure IL-6 levels, one of the most important pro-inflammatory cytokines. We examined the real-life data of patients who received TCZ in our hospital.
Our findings show that giving TCZ while the patient is still in the ward and before the ICU had a good impact on clinical response. A second dose of TCZ given to patients who had progressive respiratory worsening proved to be effective. This study will contribute to the appropriate use of TCZ in severe to critical cases with COVID-19 pneumonia.