Introduction
Stenotrophomonas maltophilia is an aerobic, non-fermentative, Gram-negative bacterium widespread in aquatic environments and soil. It has an inherent ability to adhere to abiotic surfaces and forms biofilms (1). It is usually considered to be a commensal and a low-grade pathogen. However, it can cause severe infections in the immunocompromised and debilitated hosts (2, 3). S. maltophilia is associated with various clinical syndromes, including bacteremia, pneumonia, and urinary tract infections. S. maltophilia frequently colonizes respiratory secretions, wound exudates, urine, fluids, and invasive medical devices used in the hospital setting, including disposable nebulizers, tracheal suction catheters, and respirator circuits. Pseudobacteremia outbreaks of S. maltophilia due to contamination of medical instruments and blood collection tubes were reported in the literature (4-6).
Bacteremia is a frequent and significant presentation of S. maltophilia in hospitalized patients, with a high mortality rate ranging from 11% to 51% (7, 8). Management of S. maltophilia bacteremia in clinical practice has several difficulties. Firstly, diagnosis of true bacteremia is challenging because S. maltophilia-positive blood culture may not always be clinically significant. Blood culture contamination, pseudobacteremia, or transient and self-limited bacteremia can lead to confusion in the interpretation of the culture results, particularly in the critical, immunosuppressive, and debilitated hosts (4, 9, 10). Contamination of blood cultures leads to inappropriate antibiotic use, extended hospital stays, and an increased risk of other nosocomial infections (11).
On the other hand, S. maltophilia bacteremia is a serious condition associated with relatively high mortality. Blood culture contamination of S. maltophilia is an underrated phenomenon in the literature, although there are reports on the colonization of central venous catheters (CVCs) and pseudobacteremia outbreaks (4, 12). Our clinical experience from the outbreaks in our hospital also shows that isolation of S. maltophilia from blood cultures is not always consistent with bloodstream infections. Therefore, we think that discrimination of true bacteremia from contamination is the first step of patient management.
S. maltophilia is intrinsically resistant to many antimicrobials, including most beta-lactams. Therefore, treatment options are limited and clinical data on optimal treatment regimens are lacking. Trimethoprim-sulfamethoxazole (SXT) is accepted as the first-line treatment option for S. maltophilia; however, adverse effects and antimicrobial resistance can limit its use in some patients (13). Fluoroquinolones (FQs) are suggested as alternative regimens for the treatment of bacteremia. However, rising resistance to FQs is also of concern, and there are toxicity issues (14).
In this study, we comprehensively evaluated S. maltophilia bacteremia. We aimed to determine the rates of true and false-positive S. maltophilia, reveal factors discriminating true from false-positive bacteremia, and assess the risk factors for mortality and the impact of different treatment regimens.
Materials and Methods
This retrospective study was conducted in Gazi University Hospital, a 1007-bed teaching hospital, between January 2015 and October 2021. Gazi University Ethics Committee approved the study with decision number 14 on September 7, 2021.
Study Design and Data Collection
The medical records of all adult (≥18 years) patients with at least one positive S. maltophilia blood culture were evaluated retrospectively by two infectious disease specialists. Patients with insufficient clinical data were excluded from the analysis. Data on patients’ demographics, comorbidities, previous antibiotic use, possible risk factors for bacteremia, clinical and laboratory findings, treatment regimens, outcome, and antimicrobial susceptibility results were retrieved from electronic medical records. The Pitt bacteremia score (PBS) was calculated on the day of blood culture collection. The first episode of bacteremia was included in the study if the patient had more than one episode.
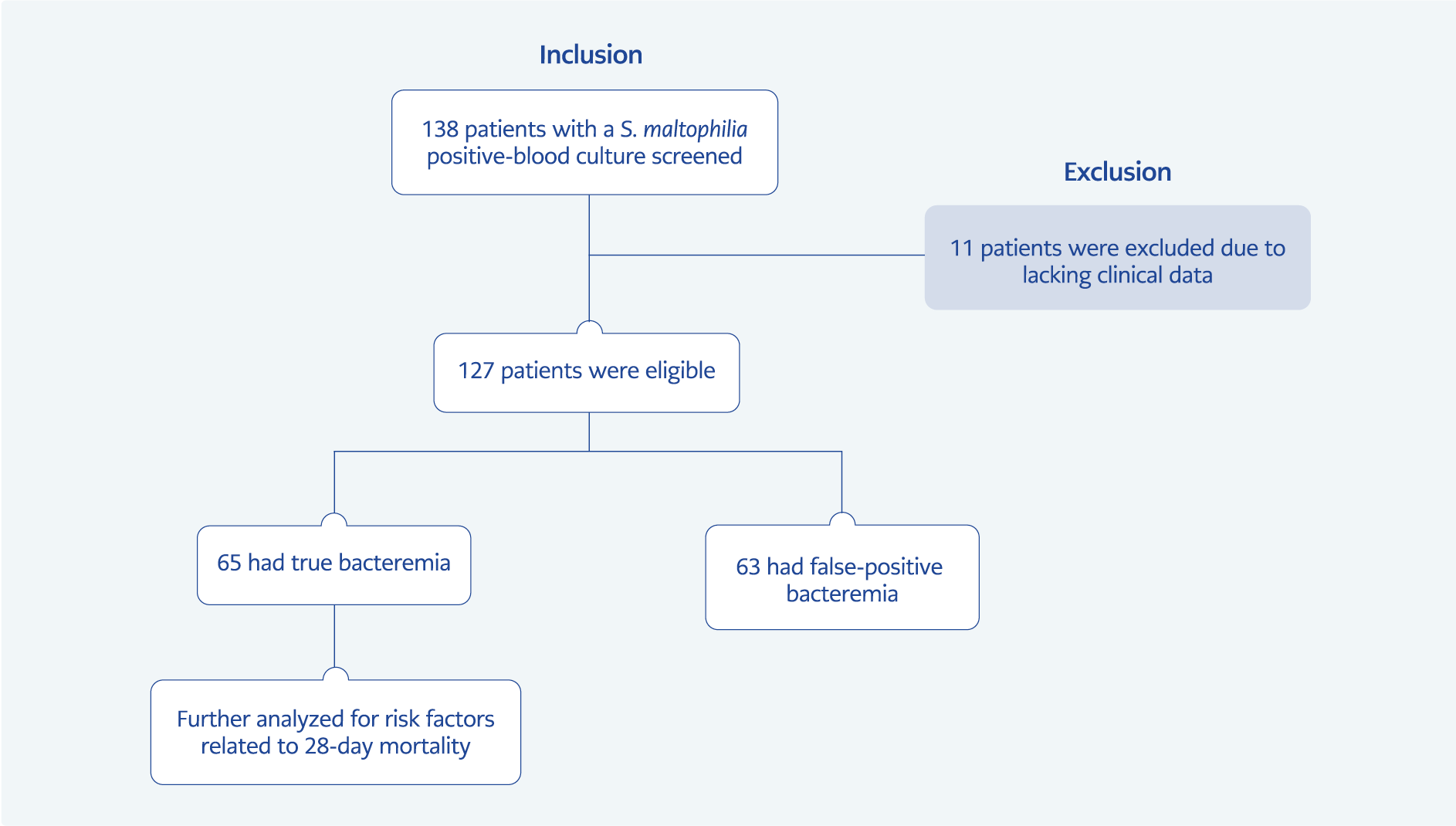
We did a two-stage analysis. First, the clinical significance of blood cultures was assessed, and patients were divided into “true bacteremia” and “false-positive bacteremia” groups. These assessments were mainly based on clinical signs and symptoms compatible with bacteremia, the presence of an infection source at the time of bacteremia, and the number of positive blood cultures. Laboratory markers, all bacterial cultures, and other possible infection sources were also evaluated. After this analysis, we excluded false-positive cases and further analyzed the factors associated with 28-day mortality in true bacteremia cases (Figure 1). We also evaluated the antimicrobial regimens used for the treatment of true bacteremia cases.
Microbiological Identification
Bacterial identification of S. maltophilia derived from blood cultures and other clinical specimens was performed with MALDI-TOF MS (Bruker, Germany) and VITEK-2 (bioMerieux, France) automated systems. Antimicrobial susceptibility testing was performed manually using the disk diffusion method on Muller Hinton agar. The susceptibility of SXT was evaluated according to European Committee on Antimicrobial Susceptibility Testing (EUCAST) Guideline, while the susceptibility of levofloxacin was according to Clinical and Laboratory Standards Institute (CLSI) standards.
Definitions
True bacteremia was defined as the presence of compatible clinical signs and symptoms with or without the presence of an infection source at the time of bacteremia or the presence of multiple positive blood cultures. Patients without compatible clinical signs and symptoms were grouped as “false-positive bacteremia”. Patients who had compatible signs and symptoms at the time of culture collection and also met the following criteria were also grouped in “false-positive bacteremia”:
- Clinical response to an empirical antibiotic that did not cover S. maltophilia,
- Any other clear infection source consistent with the clinical situation,
- Spontaneous resolution of the clinical sign and symptoms until the definitive culture results are available.
Hypotension was defined as a mean arterial pressure ≤65mmHg. Nosocomial bacteremia was defined as bacteremia that occurred at least 48 hours after hospital admission. Polymicrobial bacteremia was defined as the presence of any organism other than S. maltophilia in the same blood culture. The primary source of bacteremia was defined as the isolation of S. maltophilia from clinical specimens prior to or on the same day as the onset of bacteremia. A concomitant infection was defined as any other bacterial growth in any culture. Neutropenia was defined as an absolute neutrophil count (ANC) <500/mm3. Previous antibiotic use was defined as the administration of antibiotics longer than 48 hours within 30 days. Corticosteroid use was defined as taking 20 mg of prednisolone or an equivalent dose for at least two weeks within 90 days. Sepsis is defined as organ dysfunction, which can be identified as an acute change in total sequential organ failure (SOFA) score ≥2 points consequent to the infection (15).
Statistical Analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS) 22.0 program (IBM Corp., Armonk, NY, USA). The variables were investigated using visual (histograms, probability plots) and analytical methods (Kolmogorov-Smirnov and Shapiro-Wilk’s test) to determine whether they are normally distributed. The categorical variables were expressed as a number and a percentage, and continuous variables were presented as mean and standard deviation (SD) or median values and an interquartile range (IQR) of 25%–75%. The categorical variables were analyzed with a Chi-square test or Fisher exact test. The non-parametric variables were analyzed using the Mann–Whitney U test and the parametric ones with a Student t-test. In the univariate analysis, variables with a p-value of less than 0.20 and not correlated with each other were included in the logistic regression model. Values with a type I error level of below 5% were considered statistically significant.
Results
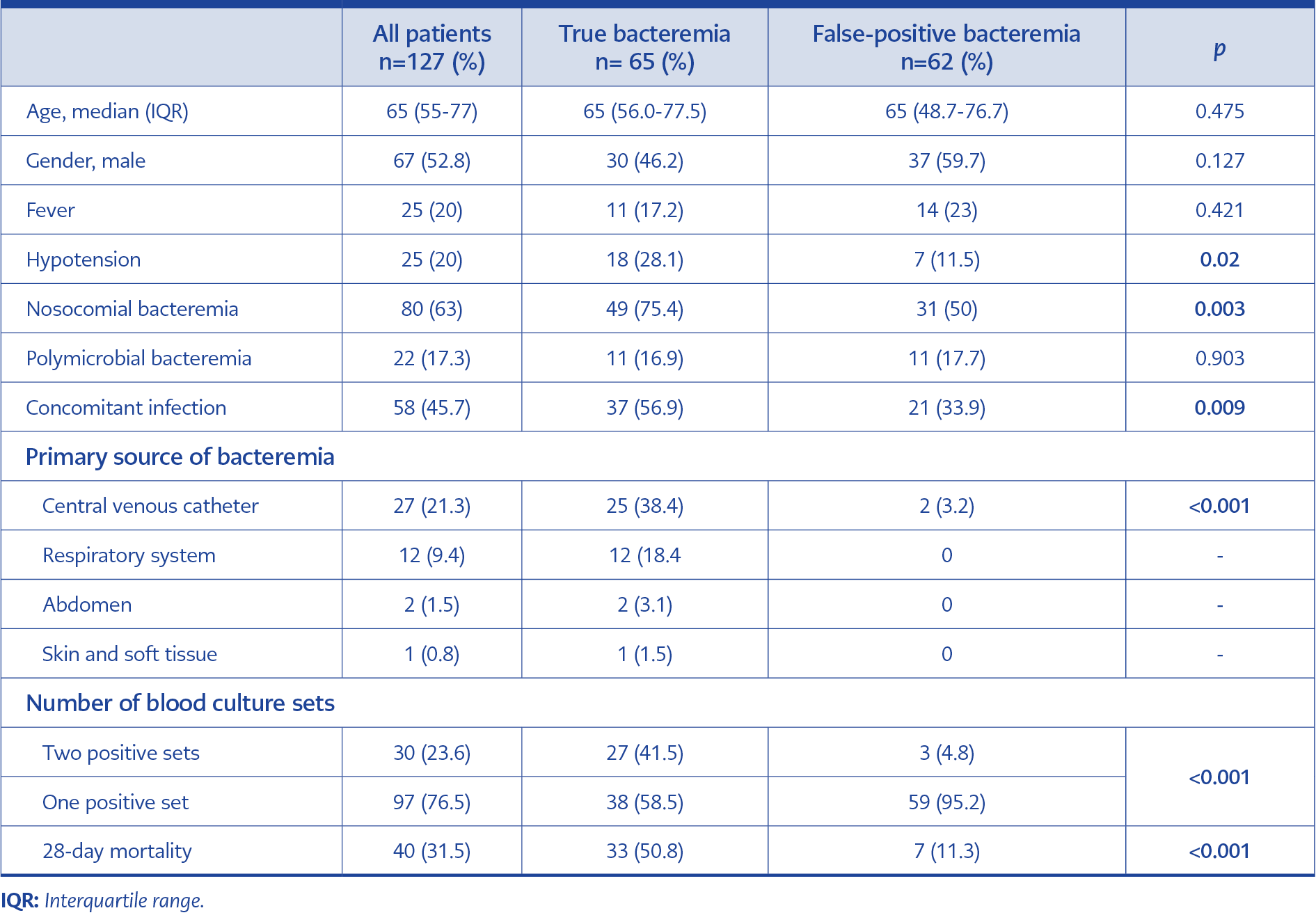
One hundred thirty-eight hospitalized patients with S. maltophilia bacteremia were initially screened, and 127 were included in the study. 11 patients were excluded from the analysis due to a lack of clinical data. True bacteremia was identified in 65 (51.2%) and false-positive bacteremia in 62 (48.8%) patients. The median age of all patients was 65 (IQR: 55-77), and 52.8% were male. The median age of true and false-positive bacteremia groups were 65 (56-77.5) and 65 (48.7-76.7), respectively. In addition, 61 (48.0%) patients were in the intensive care unit (ICU), 13 (10.2%) were in the hematology unit, 11 (8.7%) were in the oncology unit, and 42 (33.1%) were in other wards. Baseline characteristics and outcomes of patients with S. maltophilia bacteremia are shown in Table 1.
At least two sets of blood cultures were drawn in 73 (57.5%) patients and only one set in the remaining 54 (42.5%). Among patients with multiple blood cultures, two or more sets yielded S. maltophilia in 30 (41.1%) patients and one set in 43 (58.9%) patients. Most but not all patients with multiple positive blood cultures were included in the true bacteremia group. Three patients with multiple positive blood cultures whose clinical findings were not compatible with bacteremia were included in the false-positive bacteremia group. Forty-two (33.1%) patients had a detectable source of bacteremia. The most common sources were CVCs and the respiratory system. 95.2% of the patients with a detectable source of bacteremia were identified in the true bacteremia group (Table 1).
One hundred sixteen (84.1%) patients had monomicrobial bacteremia, and 22 (15.9%) patients had polymicrobial bacteremia with the following co-pathogens: coagulase-negative staphylococci (n=4), Pseudomonas aeruginosa (n=3), Achromobacter spp. (n=3), E. coli (n=2), Acinetobacter baumanni (n=2), Enterobacter spp. (n=1), K. pneumoniae (n=1), S. marcescens (n=1), Chryseobacterium spp. (n=1), E. faecium (n=1) , S. aureus (n=1), S. gordonii (n=1), Candida spp. (n=1).
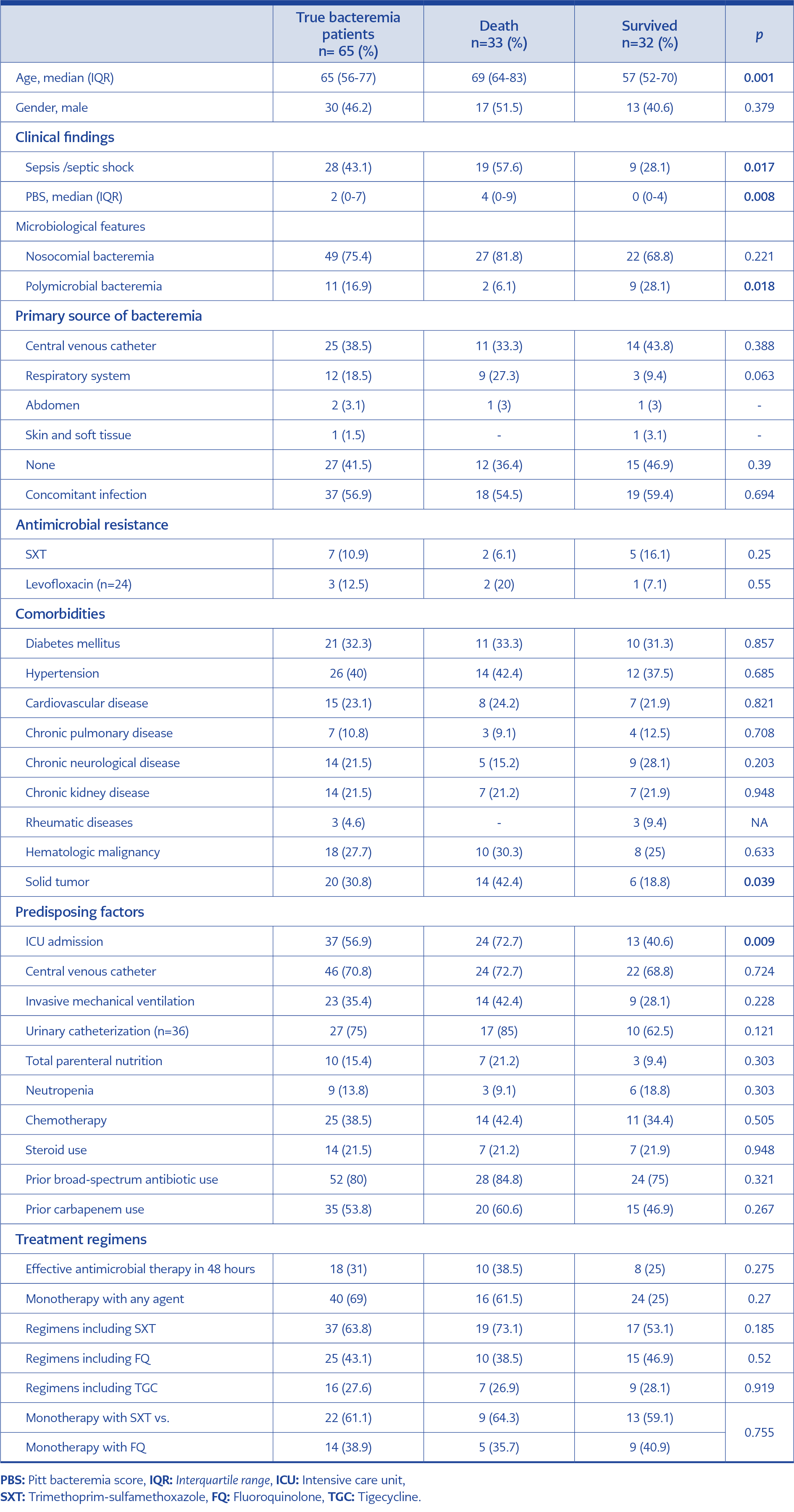
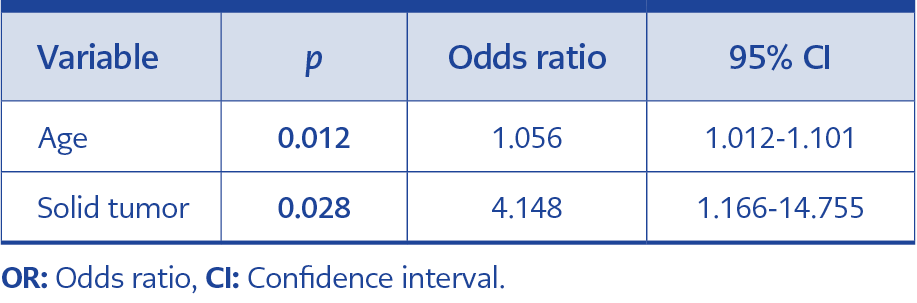
After identifying true bacteremia cases, we evaluated the 28-day mortality rate, risk factors related to 28-day mortality, and preferred treatment regimens. The 28-day mortality was 50.7%, and the risk factors associated with mortality of true S. maltophilia bacteremia cases are shown in Table 2. In multivariate analysis, age and solid tumor were the independent predictors of 28-day mortality (Table 3).
Among 65 true bacteremia cases, 58 (89.2%) received specific antimicrobial therapy. None of the patients in the false-positive bacteremia group had received antimicrobial therapy covering S. maltophilia. SXT and FQs, including levofloxacin and ciprofloxacin, were the most frequently preferred antibiotics, either with monotherapy or in combination. The percentage of used antimicrobials were as follows: SXT alone 37.9%, FQ alone 24.1%, SXT and tigecycline (TGC) 10.3%, SXT and FQ 6.9%, SXT and FQ and TGC 6.9%, FQ and TGC %5.2, ceftazidime 1.7%, SXT and ceftazidime 1.7%. The antimicrobial regimens did not significantly affect survival (Table 2).
Discussion
There are several difficulties in the diagnosis and treatment of S. maltophilia bacteremia. The gold standard and first-line diagnostic test in patients with bacteremia are regarded as blood culture analysis (16). The disadvantage of blood cultures is that they can easily get contaminated, leading to difficulties in patient management. Culture contaminants are typically constituted of Gram-positive bacteria, and Gram-negative bacteria are generally interpreted as true pathogens when isolated from blood cultures (9, 17, 18). However, this study presented a high frequency of false-positive S. maltophilia bacteremia in hospitalized patients. To our knowledge, no other study in the literature directly evaluated the false-positive S. maltophilia bacteremia rate.
S. maltophilia is an environmental pathogen, and the microbiological properties of the microorganism may facilitate the colonization of body parts and contamination of medical devices. Deviations from the aseptic technique during venipuncture or blood culture collection from central lines may easily lead to contamination. Contamination may also occur during the preparation of culture media or laboratory processing of the culture (19). Apart from contamination, short-term exposure to contaminated exogenous sources may also cause transient and self-limited bacteremia episodes. These clinically non-significant episodes may have been identified as contamination and contributed to our study’s high rate of false-positive bacteremia. A significant contributor to the high frequency of false-positive bacteremia was the outbreaks in our hospital in 2015 and 2021. The contamination source was not defined in the first outbreak, although a comprehensive outbreak analysis revealed that the contamination source was blood gas syringes in the second one. This outbreak ended shortly after the contaminated blood gas injectors were recalled. The common use of contaminated blood gas injectors may have led to clinically insignificant bacteremia in many patients and increased the frequency of false-positive bacteremia in this study.
Accurately diagnosing maltophilia bacteremia is challenging due to the high rate of false-positive cases and many confounding factors encountered during patient management. In the clinical setting, a case of bacteremia is generally assessed as true bacteremia if there are clinical findings compatible with bacteremia. On the other hand, S. maltophilia is a pathogen frequently involved in polymicrobial bacteremia, and other concomitant infections are also common in clinical settings. Therefore, in such patients, it is hard to attribute the clinical findings to S. maltophilia bacteremia. Additional parameters are needed to clarify the diagnosis. Multiple positive blood culture sets and the presence of a primary source of bacteremia are also significant indicators of true bacteremia. Literature also supports that multiple positive blood cultures are significant indicators of true bacteremia (11). Therefore, collecting at least two sets of blood cultures, including one aerobic and one anaerobic bottle in each set, is recommended for every patient with suspected bacteremia. In our study, the vast majority of the bacteremia episodes with ≥2 positive blood cultures were true bacteremia; however, clinical findings were not compatible with bloodstream infection in three patients, although ≥2 blood cultures grew S. maltophilia. All three patients had CVCs, and the colonization of CVC might have led to recurrent blood culture positivity. In this study, we used a combined evaluation of clinical findings with the number of blood culture sets and primary sources of bacteremia in order to distinguish true from false-positive bacteremia. Then, we assessed 28-day mortality and found that mortality was significantly lower in patients with false-positive bacteremia, although they did not receive antimicrobial therapy for bacteremia. This finding confirms that our criteria help distinguish false-positive cases.
In the second stage of the study, we evaluated the true bacteremia cases in terms of mortality. Our study’s 28-day mortality rate was high, compatible with other studies in the literature reporting mortality rates between 25% and 51% (8, 20, 21). The high mortality rate of our study is not unexpected because more than half of the patients were in the ICU; 58% had solid or hematological malignancy, and the median age was 65 years. In the literature, several risk factors were identified for mortality. Hematological malignancy, neutropenia, admission to an ICU, indwelling a CVC, mechanical ventilation, hypotension, high SOFA score, and sepsis or septic shock are the common factors associated with mortality in many studies (8, 21, 22). In our study, advanced age and an underlying solid tumor were independent predictors of mortality. Sepsis or septic shock, high PBS, and ICU admission were significantly high in patients with mortality in the univariate analysis; however, they lost their statistical significance in the multivariate analysis.
There is a concern about the rising antimicrobial resistance of SXT and FQs among S. maltophilia strains. Recent data reveals that the rate of SXT resistance ranges from 9.7% to 21.6% (13, 14, 23, 24). Levofloxacin resistance is also concerning due to the increased use of FQs and changes from 9.7% to 24% in different studies (14, 23, 24). In our study, SXT and levofloxacin resistance was not high and seemed lower than many other reports (10.9% and 12.5%, respectively).
Early and appropriate antimicrobial therapy is crucial for many infections, particularly bacteremia. However, the literature has conflicting data for S. maltophilia bacteremia on this issue. Kanchanasuwan et al., suggested that an appropriate empirical therapy started within 72 hours after obtaining blood cultures was associated with lower 30-day mortality in hospitalized patients with S. maltophilia bacteremia (25). By contrast, inadequate or delayed antimicrobial therapy was not related to mortality in many studies (20, 21, 26). Ebara et al., showed that patients without specific treatment for S. maltophilia had a paradoxically higher survival rate than those who received treatment, although this finding was not statistically significant (8). Our data also support that early adequate antimicrobial treatment did not significantly impact 28-day mortality.
SXT is the first-line treatment option in S. maltophilia infections; however, hypersensitivity, toxicity, or other adverse reactions and the rising antimicrobial resistance can limit its use. Therefore, other antimicrobials, including FQs, minocycline, TGC, and ceftazidime, are considered for the treatment. However, clinical data on alternative regimens is limited. A few studies examine the treatment options for S. maltophilia bacteremia, mainly focusing on the FQs as an alternative to SXT. A meta-analysis evaluated the clinical efficacy of FQs compared with SXT and demonstrated comparable effects of FQs on mortality to SXT (27). Our findings support that the treatment regimens, either combination or monotherapy, had a similar impact on survival. Therefore, regimens, including SXT, FQs, or TGC, can be used to treat S. maltophilia bacteremia. In the presence of polymicrobial bacteremia or accompanying other infections, TGC may be an alternative option, especially in combination with SXT and FQ, owing to its broad spectrum of activity. Further well-designed studies are needed to clarify these findings and to determine optimal treatment regimens.
Our study has several limitations. First, “true bacteremia” and “false-positive bacteremia” were defined retrospectively. This may have led to some errors in the categorization of the patients. Second, the single-center nature of the study does not exclude the possibility that the results may be affected by the center-specific conditions and practices. Third, the small sample size was relatively small to make a clear recommendation on mortality predictors and treatment alternatives.
In conclusion, our study reveals that isolating S. maltophilia in blood cultures does not always indicate a true infection, and the high rate of false-positive bacteremia should be considered during patient management. Environmental contamination should be investigated if there is an unexpected increase in the number of S. maltophilia cases. Multiple positive blood culture sets and the presence of primary sources of bacteremia, as well as clinical findings, are indicative of true bacteremia and should be carefully assessed during patient management. Age and underlying solid tumors are the factors independently associated with mortality. Advanced-aged patients with oncologic malignancy should be followed up closely. Antimicrobial regimens, including SXT, FQs, or TGC, have a similar impact on survival. Therefore, all these regimens can be preferred in patients with S. maltophilia bacteremia considering antimicrobial resistance and adverse effects or toxicity.