Introduction
COVID-19, caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), is a zoonotic infection that emerged in Wuhan province of China in December 2019. SARS-COV-2 is a Betacoronavirus that is classified in Riboviria realm, Nidovirales order, and Coronaviridae family (1). Although the fatality rate of SARS-CoV-2 (2.6%) is lower than SARS-CoV (11%) and MERS-CoV (35-50%), it was much more severe worldwide because of its high transmission rate, and millions of people have died due to COVID-19 (2). According to the World Health Organization (WHO), as of September 6, 2023, there have been 770,437,327 confirmed cases and 6,956,900 deaths due to COVID-19 worldwide. As of September 4, 2023 a total of 16,023 new COVID-19 cases have been reported in the last seven days (3).
Clinically, it can be asymptomatic or have a broad spectrum of clinical manifestations ranging from simple upper respiratory tract infection to pneumonia, acute respiratory distress syndrome (ARDS), multiple organ failure, shock, and death (4). In people who have recovered from the disease, the symptoms and clinical signs that occur with the effect of the disease can persist for a long time and adversely affect the quality of life of the person. The term ‘long COVID’ or ‘post-COVID-19 syndrome’ emerges when these symptoms, common among some people following acute SARS-CoV-2 infection, are evaluated. Long COVID describes the persistence and emergence of a heterogeneous group of symptoms at least 12 weeks after acute infection (5). Although there is no precise definition of long COVID, the most common symptoms reported are fatigue and dyspnea that persist for months after having acute COVID-19. Other persistent symptoms include cognitive and mental disorders, chest pain, arthralgia, palpitations, myalgia, anosmia, dysgeusia, cough, headache, and gastrointestinal and heart problems (6). Long COVID-related data revealed that rehabilitation support is needed for symptom-adjusted physical activity and personalized exercise programs. Therefore, appropriate and personalized exercise programs seem promising and effective therapy to alleviate post-COVID symptoms, accelerate the recovery process, and help them improve their autonomy, functionality, and quality of life (7). In our study, we aimed to evaluate the long-term symptoms of COVID-19 and the impact of these symptoms on the individual’s quality of life. We also aimed to show the effect of the SARS-CoV-2 vaccine on these symptoms and quality of life.
Materials and Methods
Our descriptive and cross-sectional study included all patients who applied to Bozok University Research and Practice Hospital Department of Infectious Diseases and Clinical Microbiology between April 2020 and December 2021. Of 625 patients, 45 died during the follow-up period, 49 could not be reached, and ten refused to participate in the study. As a result, the study was conducted with 521 patients. Patients were evaluated at least three and at most 12 (mean=7.43±3.33) months after diagnosis.
The survey, consisting of 60 questions, was conducted face-to-face and over the telephone. The first part contained 18 questions about the sociodemographic features and COVID-19 symptoms of the participants. When defining post-COVID symptoms, patients were asked about symptoms that did not exist before but developed and persisted or worsened after COVID-19. In the second part, there was the medical outcomes study (MOS) 36-Item Short-Form Health Survey (SF-36) and European Quality of Life 5 Dimensions 3 Level Version (EQ-5D-3L) Scale to determine the quality of life of the participants. The researchers read the questions and options to the illiterate participants and collected their answers. Descriptive analyses were performed to provide information about the general characteristics of the study groups. Data on continuous variables were presented as mean±standard deviation, and data on categorical variables were presented as n (%). Analysis of variance (ANOVA) were used to compare the groups. The statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) 15.0 (IBM Corp., Armonk, NY, USA). The statistical significance level was accepted as p<0.05. Informed consent was obtained from all participants.
The study was approved by the Institutional Ethics Committee of Bozok University with the number 2017-KAEK-189_2021.05.26_08. All procedures were performed in accordance with the ethical standards of the Declaration of Helsinki.
The MOS 36-Item Short-Form Health Survey (SF-36)
Ware and Sherbourne developed it to assess the quality of life of individuals (8). Its Turkish validity and reliability study was performed by Koçyiğit et al. in 1999 (9). The scale consists of a total of 36 questions and eight sub-dimensions, including general health (GH), bodily pain (BP), social functioning (SF), mental health (MH), energy-vitality (VT), emotional role limitations (RE), physical role limitations (RF) and physical functioning (PF). Each sub-dimension has a score between 0 and 100, and the higher the score, the higher the quality of life level.
European Quality of Life 5 Dimensions 3 Level Version (EQ-5D-3L) Scale
The scale was developed by the European Quality of Life Research Group (EuroQol) in 1987 and consists of five three-choice items, including mobility, self-care, usual activities, pain/discomfort, and anxiety/depression. In addition, there is a visual analog scale with a score between 0 and 100 based on the individuals’ self-assessment of their current health status. According to the answer item selected in the five-question section, the index score is obtained by subtracting the total obtained by multiplying by a certain coefficient from 1. As the index score approaches 1 or the visual analog scale approaches 100, quality of life increases.
Results
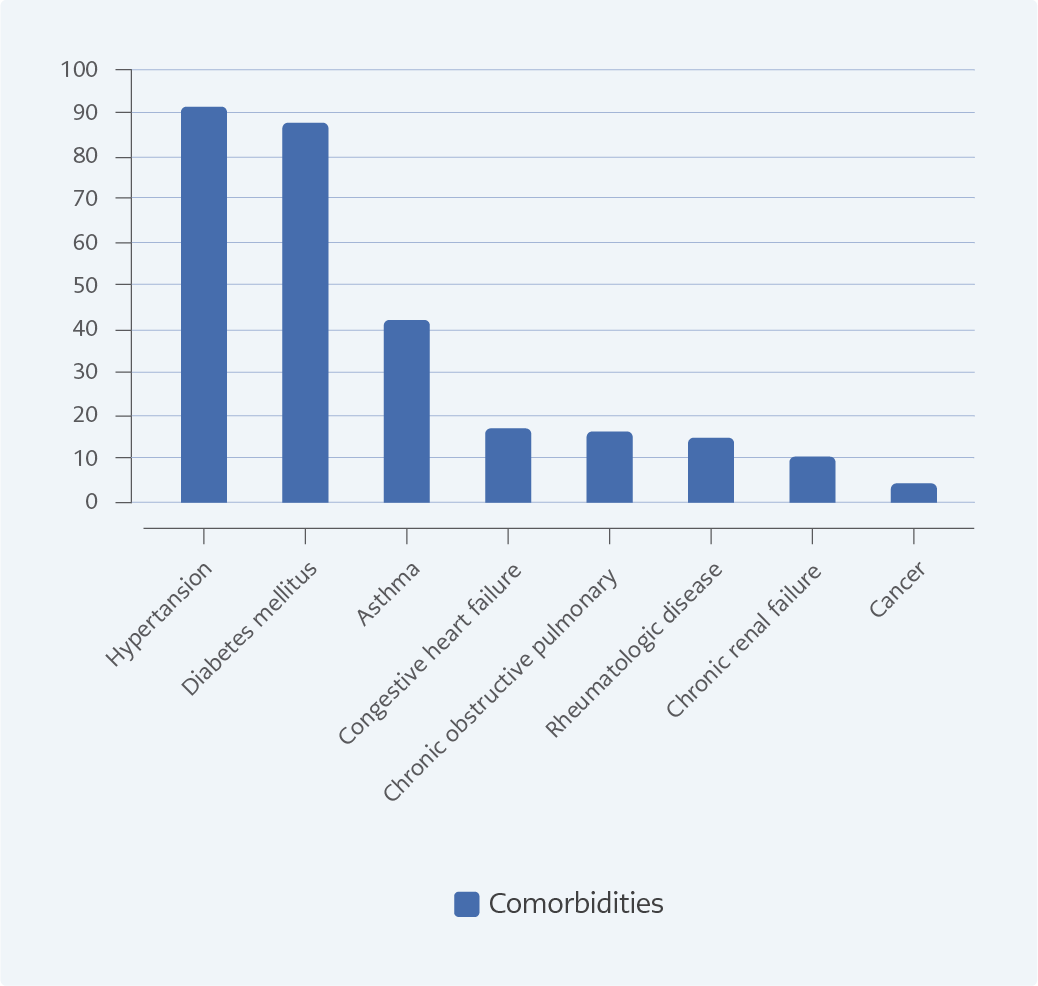
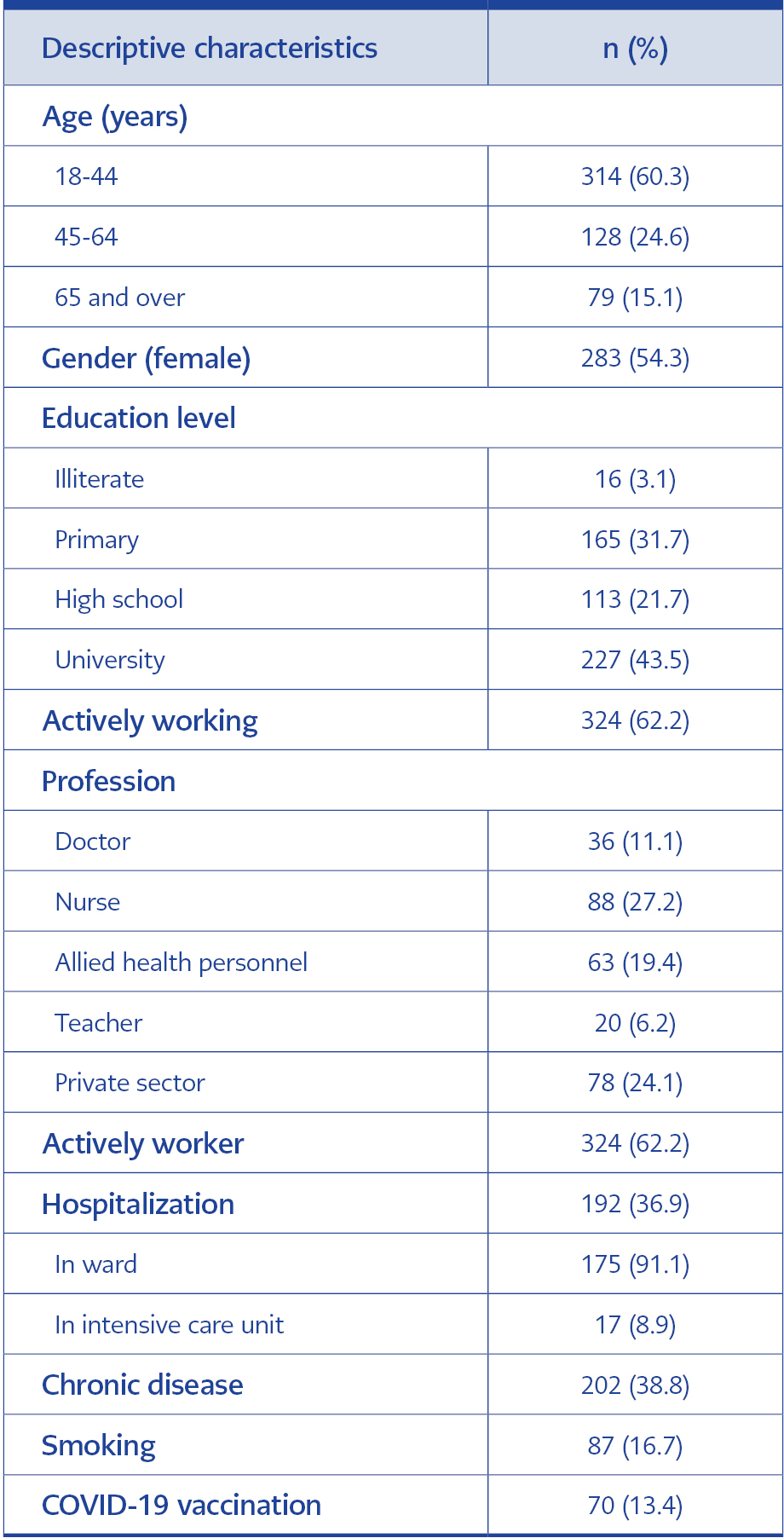
A total of 521 (54.3% female, 45.7 % male) patients were included in the study. The mean age of the study group was 43.45±16.91 (18-89) years, and 60.3% were in the 18-44 age group; 38.8% had comorbidities. Of the participants, 13.4% stated that they had received at least one SARS-CoV-2 vaccine, while 86.6% stated that they had not received any COVID-19 vaccine. Only three participants reported having a second COVID-19 course during the study. Demographic characteristics and SARS-CoV-2 vaccination status of the patients are summarized in Table 1. The chronic diseases of the participants are shown in Figure 1.
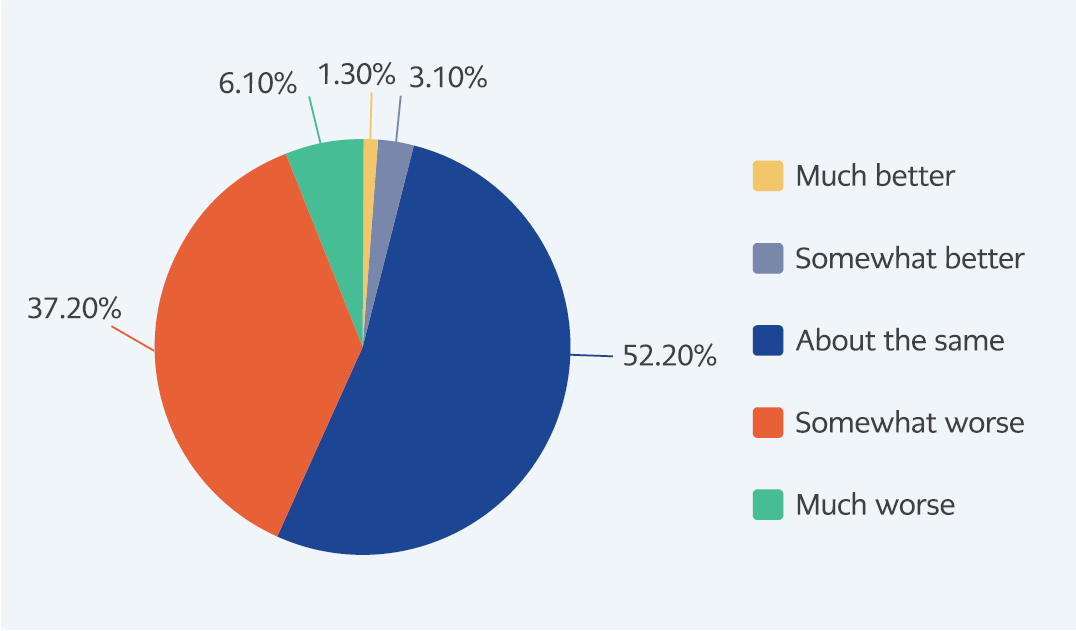
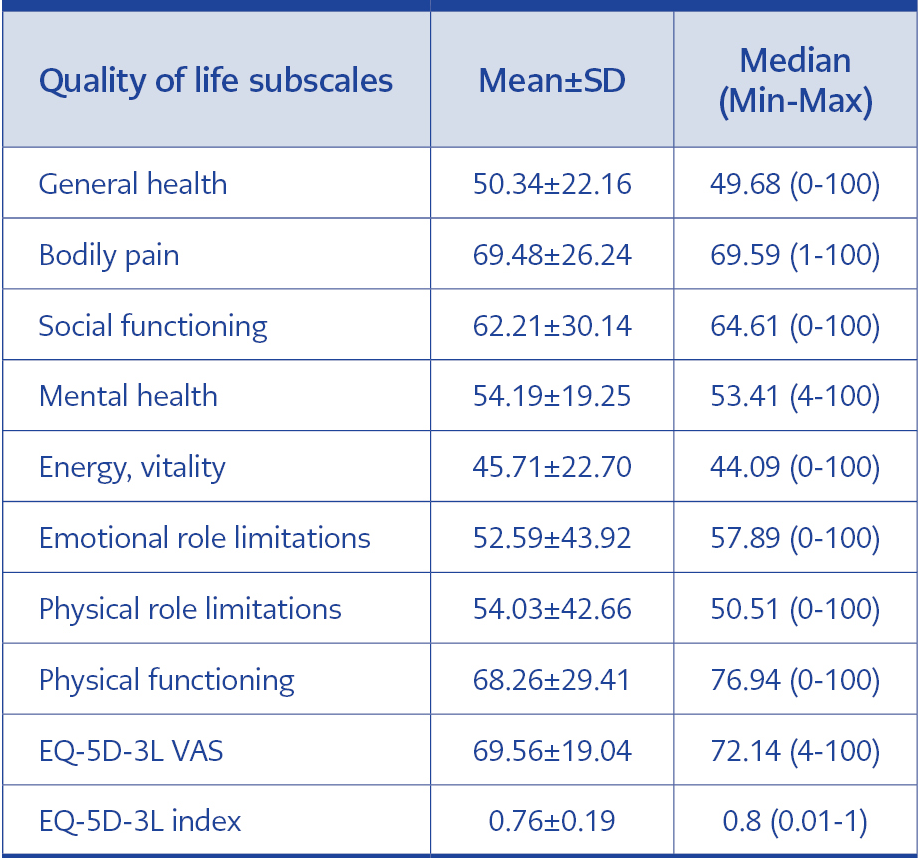
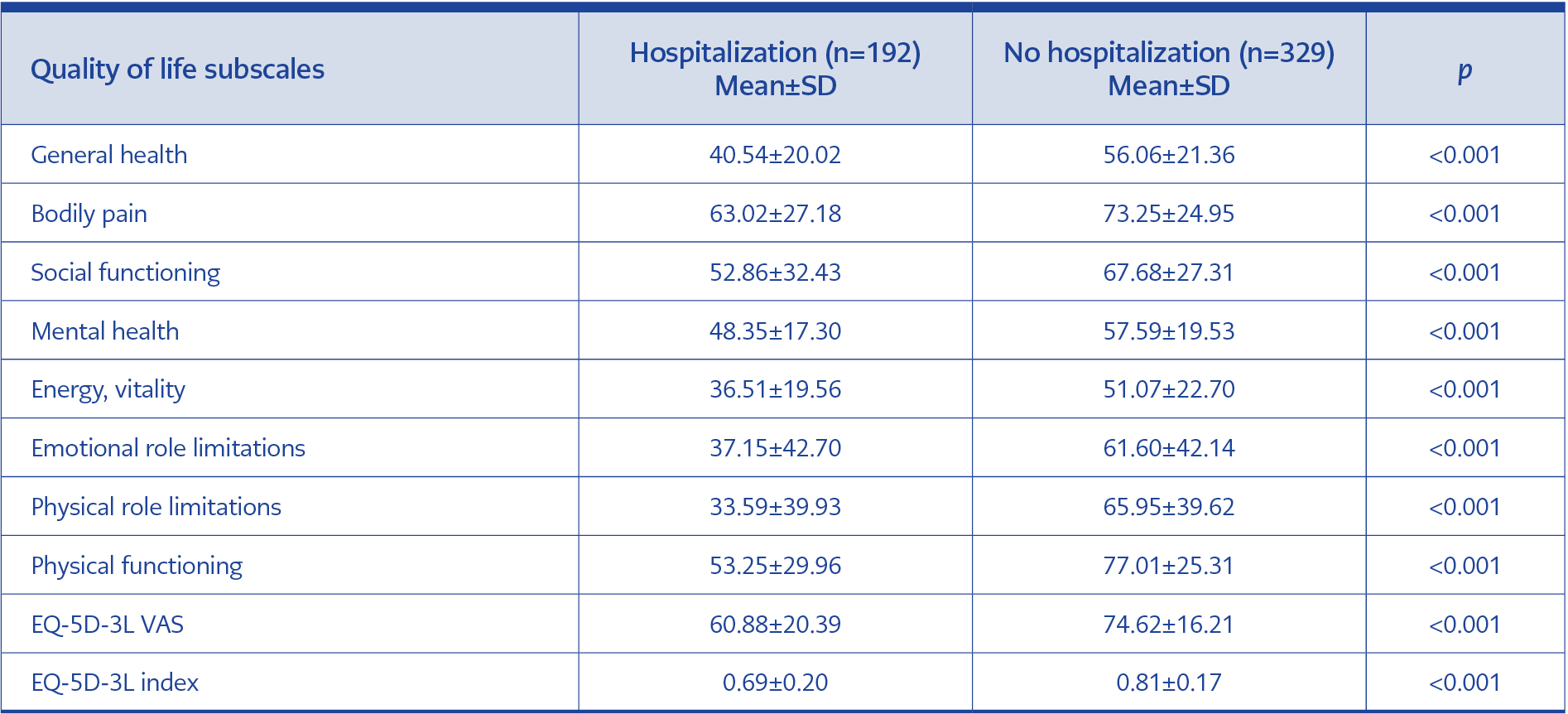
Only 19% of participants did not report any post-COVID symptoms, while the prevalence of participants reporting at least one post-COVID symptom was 81%. The three most commonly reported post-COVID symptoms in our study were weakness-fatigue (55.3%), dyspnea (25%) and sleep disorders (20.9%). Other complaints experienced by the participants in the post-COVID-19 period were found as cough (14.2%), distraction (12.5%), chest pain (11.1%), anosmia (10.4%), hair loss (9.4%), loss of taste (9%), depression (8.6%) and others (13.6%). Compared to pre-infection, 43.3 % of participants reported that their health status was worse than before, as shown in Figure 2. SF-36 and EQ-5D-3L subscale scores of the participants were found as shown in Table 2.
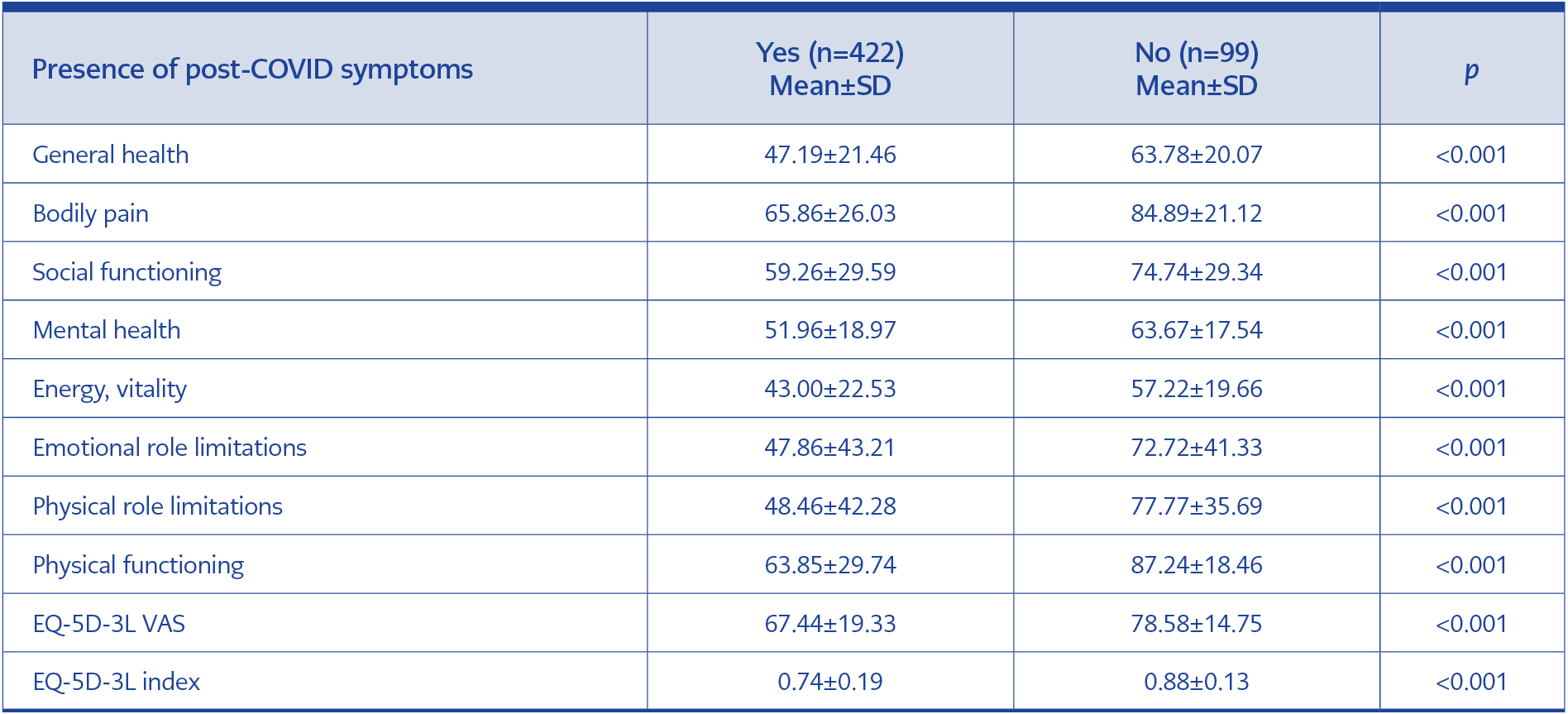
Table 3 Comparison of quality of life subscale scores according to the presence of post-COVID symptoms.
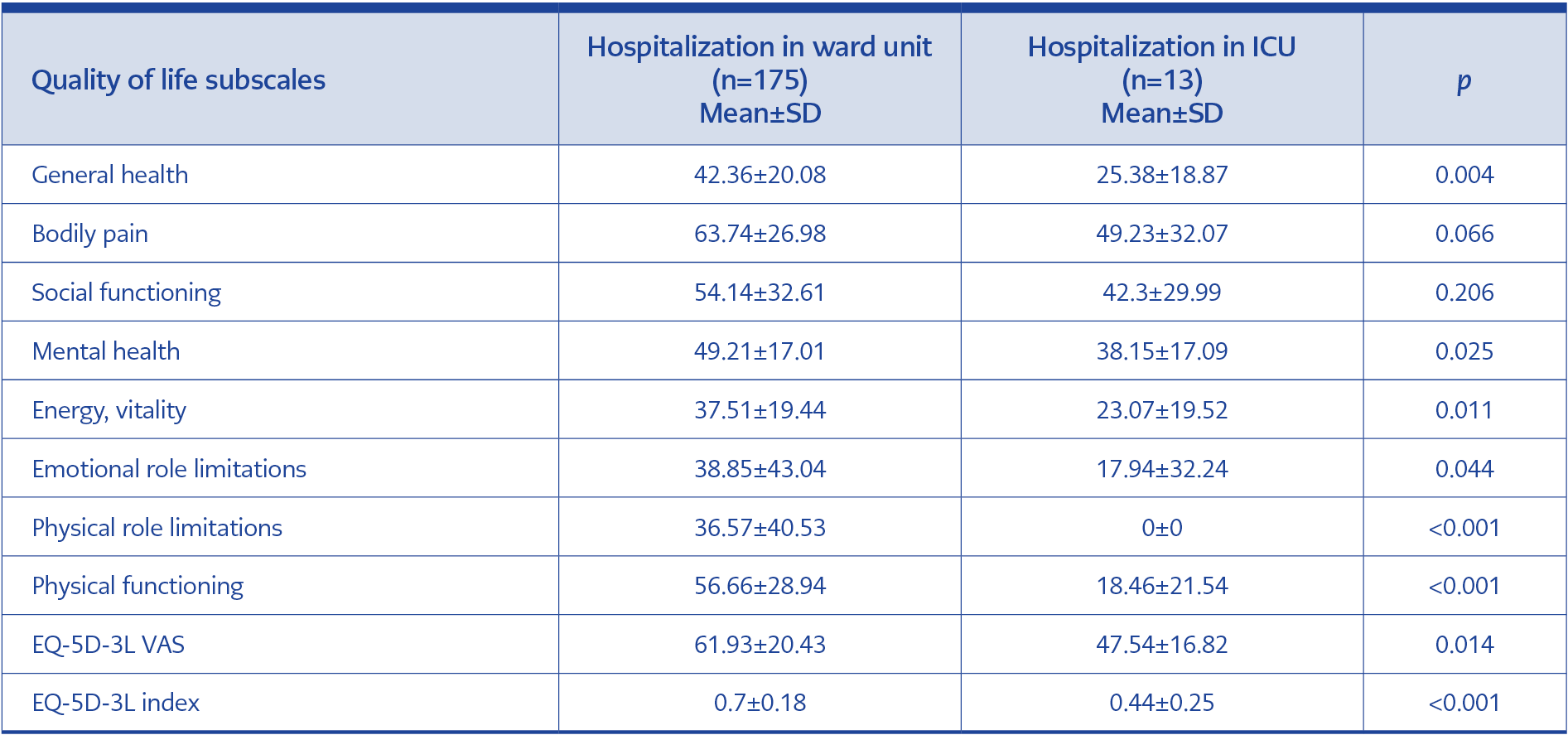
As shown in Table 3, those with post-COVID symptoms scored significantly lower on all subscales than those without symptoms. Compared to women, men scored higher on both scales. The difference between men and women was statistically significant on all SF-36 and EQ-5D-3L sub-dimensions except the mental health sub-dimension of the SF-36. According to age groups, the 18-44 age group had the highest, and the 65 years and older group had the lowest scores. We also compared participants according to their hospitalization status due to COVID-19 and found that hospitalized participants had significantly lower scores on all subscales than non-hospitalized participants. Participants hospitalized in the ward had higher scores than those in the intensive care unit (ICU). Except for BP and SF, this difference was statistically significant in EQ-D-3L and SF-36 subscales. The quality of life scores of the participants according to their hospitalization status due to COVID-19 and the place of hospitalization are shown in Table 4 and Table 5.
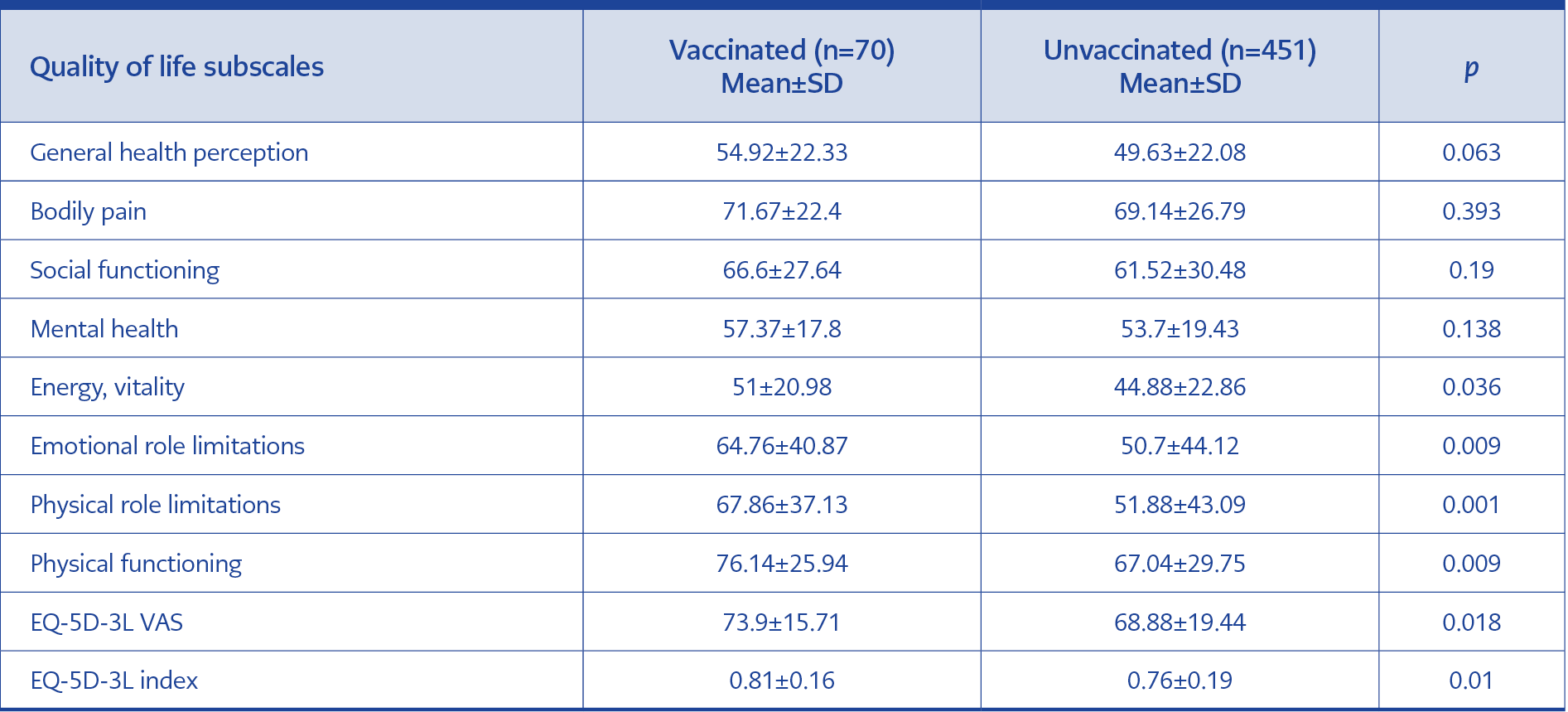
Compared to COVID-19 vaccination status, it was observed that vaccinated participants had higher scores on both scales, as shown in Table 6. However, there was a statistically significant difference between the groups in VT, RE, RF, and PF subscales, while the difference in GH, BP, SF, and MH subscales was not statistically significant. The difference between vaccinated and unvaccinated groups was statistically significant in all EQ-5D-3L scales.
When we compared the quality of life scores according to active working status, the actively working group had the highest score in all sub-dimensions in a statistically significant manner.
Discussion
Since the beginning of the pandemic, the effects of pandemic conditions, quarantine measures, and therefore limited physical activity on society and individuals, and their reflection on the quality of life have frequently been investigated. Although there are many studies on the quality of life during the COVID-19 period and post-COVID symptoms in our country, there are limited studies on to what extent and in which sub-dimension the quality of life of people with the disease is predominantly affected. Only 99 (19%) did not report any ongoing or newly emerging symptoms after COVID-19, while 422 patients (81%) reported suffering from at least one of these symptoms in our study. WHO estimates that approximately 10-20% of COVID-19 patients have persistent symptoms after acute infection (10). This rate is 5-32% according to the CDC (Centers for Disease Control and Prevention) and 22-40% according to the ESCMID (European Society of Clinical Microbiology and Infectious Diseases) (11, 12). The high prevalence of long COVID in our study may be related to the relatively low patient population compared to WHO, CDC, and ESCMID data, which were obtained by analyzing millions of patient data and many publications. In addition, most of the individuals who participated in our study got the disease when any SARS-CoV-2 vaccine was not administered in Türkiye. This may be associated with the low number of people vaccinated against COVID-19 and the high prevalence of long-term COVID-19 in our study. In a study, the prevalence of persistent symptoms was 75.9%, but the vaccination status of patients was unknown (13). In a study conducted by Taboada et al. in Spain during the pre-vaccine period, 71 patients hospitalized in ICU due to COVID-19 were examined six months later in terms of quality of life and persistent symptoms, and the prevalence of persistent symptoms was reported as 84% (14). In a systematic review of 45 studies including 9751 participants who had COVID-19 between January 1, 2020, and March 11, 2021, the median rate of individuals reporting at least one persistent symptom was 73% (15). A study by Durmaz and Yılmaz reported that 37.3% of vaccinated individuals and 61.7% of non-vaccinated individuals had prolonged symptoms (16). As in many studies, the most common long COVID symptom in our study was weakness-fatigue, with a rate of 55.3%. The other three most common symptoms that the participants complained of after COVID-19 were dyspnea (25%), sleep-wake disorders (20.9%) and cough (14.2%). In our study, symptoms such as memory disorders, headache, menstrual irregularities, and perspiration were among the other symptoms, constituting 13.6% of the long COVID symptoms. Desgranges et al. evaluated patients five months after COVID-19 and found that fatigue was followed by taste and smell disturbance (22%), dyspnea (16%), headache (12%), memory impairment (11%), hair loss and sleep disturbance (10%) (17). Despite varying rates, the persistence of COVID-19 symptoms remains stable across multiple studies, demonstrating the need for long-term follow-up of patients recovering from COVID-19 infection.
Compared to the pre-COVID-19 period, 43.4% of participants reported that it was worse, and 4.4% reported that it was better. In a study by Liska et al. on patients with prolonged COVID symptoms, it was found that 38.2% of patients were in worse condition and 48.8% were much worse condition compared to the pre-infection period, while 8.5% of patients were in the same condition and 4.5% were better condition (18). In this study, the percentage of patients reporting worse compared to the pre-COVID-19 period was higher than in our study (87% vs. 43.4%). The differences may be because only patients with long COVID symptoms were evaluated in the study, and the majority of patients (83.7%) were women. However, it is questionable whether these rates can explain the high difference, and it would be healthier to make an evaluation considering the time elapsed after COVID-19, the presence of long-term COVID-19 symptoms, age, and gender distribution.
In an article investigating the SF-36 norm values of the Turkish population, it was found as follows: PF=83.8±20.0, RP=86.3±24.9, BP=82.9±18.9, GH=71.6±16.1, VT=64.5±12.9, SF=91.0±12.9, RE=90.1±19.4, MH=71.0±11.0. When compared with these data, it was observed that the quality of life of our participants was quite low in all SF-36 sub-dimensions. While the highest difference is in RE (90.1±19.4 – 52.5±43.9), RP (86.3±24.9 – 54.03±42.22) and SF (91.0±12.9 – 62.21±30.14) sub-dimensions, the lowest difference is between BP (82.9±18.9 – 69.48±26.24) and PF (83.8±20.0 – 68.26±29.41) (19). As in norm values, we found that female sex and advanced age negatively affected quality of life. The difference between VT scores was not significant in norm values, whereas it was statistically significant in our study. This may be due to the fact that female sex is a risk factor for long COVID and the negative effect of long COVID on VT. It may also mean that vitality is more affected in women with COVID-19 compared to men. Qu et al. compared COVID-19 survivors with norm values and reported that quality of life decreased in COVID survivors, but advanced age did not cause a significant decrease in quality of life except for PF and GH. In this study, MH was higher in females, whereas in our study, it was higher in males but not statistically significant (20).
In our study, the EQ-5D-3L scores of the individuals were in parallel with the SF-36 in terms of being lower in older age and female gender. Todt et al. evaluated patients before hospitalization and three months after discharge to assess the quality of life of patients hospitalized for COVID-19 and reported a general decrease in the EQ-5D-3L index at three months (21). In a study using the EQ-5D-5L, it was reported that there was no difference between ICU and ward patients, whereas, in our study, the quality of life of hospitalized patients was significantly lower compared to non-hospitalized patients and ICU patients compared to ward patients (22). Taboada et al. evaluated patients six months after ICU admission and reported the EQ-5D index=0.7054±0.2514, EQ-VAS=66.36±18.26 (14). These rates were lower than the scores of our participants who were evaluated six months after the disease (EQ-5D-3L index=0.79±0.21, EQ-VAS=71.66±17.49). This data supports the negative impact of ICU hospitalization on quality of life (of our participants, 63% were outpatients, 36% were inpatients, and only 6% of patients were followed up in ICU). Participants who were hospitalized during acute COVID-19 had lower scores than non-hospitalized participants on all subscales. The highest difference was seen in the RP and RE sub-dimensions, and it was thought that this might occur as a result of the conditions that occur in the hospital (such as limited physical activity, negative effects of the hospital atmosphere on the patient’s psychological health, drug side effects). Similarly, when patients hospitalized in the ward and ICU were compared, it was found that hospitalization in the ICU significantly reduced quality of life except for BP and SF. The effects of hospitalization on quality of life are shown in Table 4, and the effects of ward or intensive care hospitalization are shown in Table 5. Severe respiratory distress, impaired motor movements due to muscle atrophy, and secondary infection may cause this condition, which is more common in patients hospitalized in the ICU. Neuronal damage may be the cause of the low BP score.
Those who received the COVID-19 vaccine were found to have higher scores than those who did not. However, a statistically significant difference was found between VT, RE, RF, and PF subscales, while the difference between GH, BP, SF, and MH subscales was not found to be significant. The impact of SARS-CoV-2 vaccine on quality of life is shown in Table 6. Our study is important because it shows the impact of the vaccine on the quality of life in people who have had COVID-19.
One of the main limitations of our study is that participants’ quality of life scores before COVID-19 are not known. For more meaningful data, it seems more appropriate to compare the quality of life of patients at the time of initial presentation and at the end of the study period. In order to minimize this aspect, the quality of life scores calculated in our article was compared with the quality of life norm values of the Turkish population. When the article on norm values was evaluated, it was seen that the sample was selected in accordance with the general population, without discriminating between sick/healthy, hospitalized/non-hospitalized, male/female, and presented as SF-36 quality of life norm values of the Turkish population. In addition, during the face-to-face and telephone interviews with our patients, it was emphasized that they should answer the questions by comparing them with the pre-COVID-19 period. Another limitation of our study is the low number of vaccinated patients. In this period, when vaccination rates are increasing, new studies can be conducted with more participants.
The COVID-19 pandemic has had destructive effects all over the world, causing millions of people to fall ill and lose their lives. As a result of our study, the following are the things that can be done to alleviate symptoms, accelerate the healing process, and improve the quality of life after COVID-19:
Physical activity and personalized exercise programs adjusted according to symptoms should be developed, and patients should be included in appropriate rehabilitation programs.
A separate unit should be established to evaluate patients for complications after COVID-19, and physicians from all branches should be assigned to this unit.
Hospital and intensive care unit stays of patients hospitalized due to COVID-19 should be kept as short as possible.
Patients should be encouraged to return to active working life as early as possible.
In order to minimize the long-term complications of COVID-19 on individuals and improve the quality of life, public information campaigns on the importance of vaccination should be organized effective vaccination policies should be developed.