Introduction
The coronavirus disease 2019 (COVID-19) pandemic, with the increasing number of cases, has been a major health challenge worldwide. In addition to being a significant cause of morbidity and mortality, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection maintains its importance because of prolonged symptoms after acute disease in the post-infectious period (1). Prolonged symptoms can significantly affect the quality of life and time to return to daily activities and work (2). Although now adequate data can describe the core symptoms, course, and prevalence of ‘long COVID’, the exact pathogenesis of the full range of ‘long COVID’ symptoms has not been explained (3).
The World Health Organization (WHO) defined post-COVID conditions (long COVID) as patients who had a history of probable or confirmed COVID-19, usually three months from the onset, with symptoms lasting for at least two months and cannot be explained by an alternative diagnosis (4). Nevertheless, the literature has many different proposed descriptions of ‘long COVID’ (5). The prevalence of ‘long COVID’ differs in each study because different methods were used in the studies, including national surveys or patients-led (5). The persisting symptoms of ‘long COVID’ constitute a heterogeneous group, affecting multiple organ systems, including muscle and joint (myalgia, arthralgia), respiratory (dyspnea, cough, chest pain), and nervous (taste/smell alteration, concentration difficulty) systems (1). The most common symptoms are fatigue, dyspnea, and cognitive dysfunction, which may persist for up to six months (4, 6).
Possible risk factors for ‘long COVID’, such as biomarkers, patient and clinical characteristics, have been researched in previous studies. Female sex, more than five initial symptoms, and acute COVID-19 severity were found as the most prominent risk factors (6). Nevertheless, most studies found no relationship between disease severity and ‘long COVID’ (6). ‘long COVID’ patients with severe disease are most likely to be men 50 years; on the other hand, patients with ‘long COVID’ after non-severe acute infection are most likely to be younger women (7). Inconsistent results for risk factors were found in the literature. While the severity of acute COVID-19 was not found as an independent predictor of post-COVID syndrome by Moreno-Perez et al., Chudzik et al. showed that severe COVID-19 was one of the risk factors for developing ‘long COVID’ syndrome (8, 9). These inconsistent risk factors for ‘long COVID’ can be explained by the differences in research methods, such as variances in reporting and clinical and demographic characteristics of the selected patient cohorts (10). This study aimed to describe the persisting symptoms three months after COVID-19 and the risk factors associated with ‘long COVID’.
Materials and Methods
This is an observational cross-sectional study performed in Turkey. The study cohort includes 1301 COVID-19 patients diagnosed with SARS-CoV-2 PCR positivity in the first 18 months of the COVID-19 pandemic (March 2020-September 2021). The survey was conducted for all participants within the same period in 2022. All participants were employees of one of the biggest national banks (Halkbank) in Turkey from all over the country. The demographic characteristics of employees of the bank represented the general Turkey population in the scope of age and comorbidities. Informed verbal consent to participate in the study was obtained. Over telephone calls, participants were questioned about the usual symptoms associated with ‘long COVID’. The participants were asked to answer the questionnaire according to symptoms that persisted for three months after the date of their own past COVID-19 between March 2020 and September 2021. These symptoms included myalgia, arthralgia, back pain, dizziness, fatigue, balance disorders, difficulty in perception or concentration, sleep disorders, loss of taste or smell, tinnitus, cough, dyspnea, palpitation, diarrhea, and loss of appetite. The questions were structured by a team of infectious disease specialists, neurologists, physical medicine and rehabilitation physicians, general practitioner, and intensive care physicians. The survey also included data about demographic (sex, age, comorbidities, body mass index [BMI], smoking, weight, height) and clinical (disease severity, oxygen requirement, hospitalization, and admission to intensive care unit because of COVID-19) characteristics of the participants. Clinical syndromes associated with COVID-19 were classified according to the WHO case definitions (11). Participants answered a total of 31 questions.
The presence of ≥1 symptom, persisting ≥3 months after COVID-19, was defined as ‘long COVID’. The frequency of persisting symptoms after COVID-19 and the risk factors associated with ‘long COVID’ were analyzed. The adult participants who answered all questions were included in the study.
Statistical analysis was performed using Statistical Package for Social Sciences (SPSS) 23.0 (IBM Corp., Armonk, NY, USA). The results of descriptive statistics were reported as median (min-max) for variables that were not normally distributed. Categorical data were reported as frequencies and percentages. Subjects were classified into two groups based on the ‘long COVID’ definition. Mann-Whitney U, the Chi-square test was used to assess whether there was a significant association between categorical variables. The statistical significance was set as p<0.05. The risk factors for the development of ‘long COVID’ were identified by multivariant logistic regression analysis.
Results
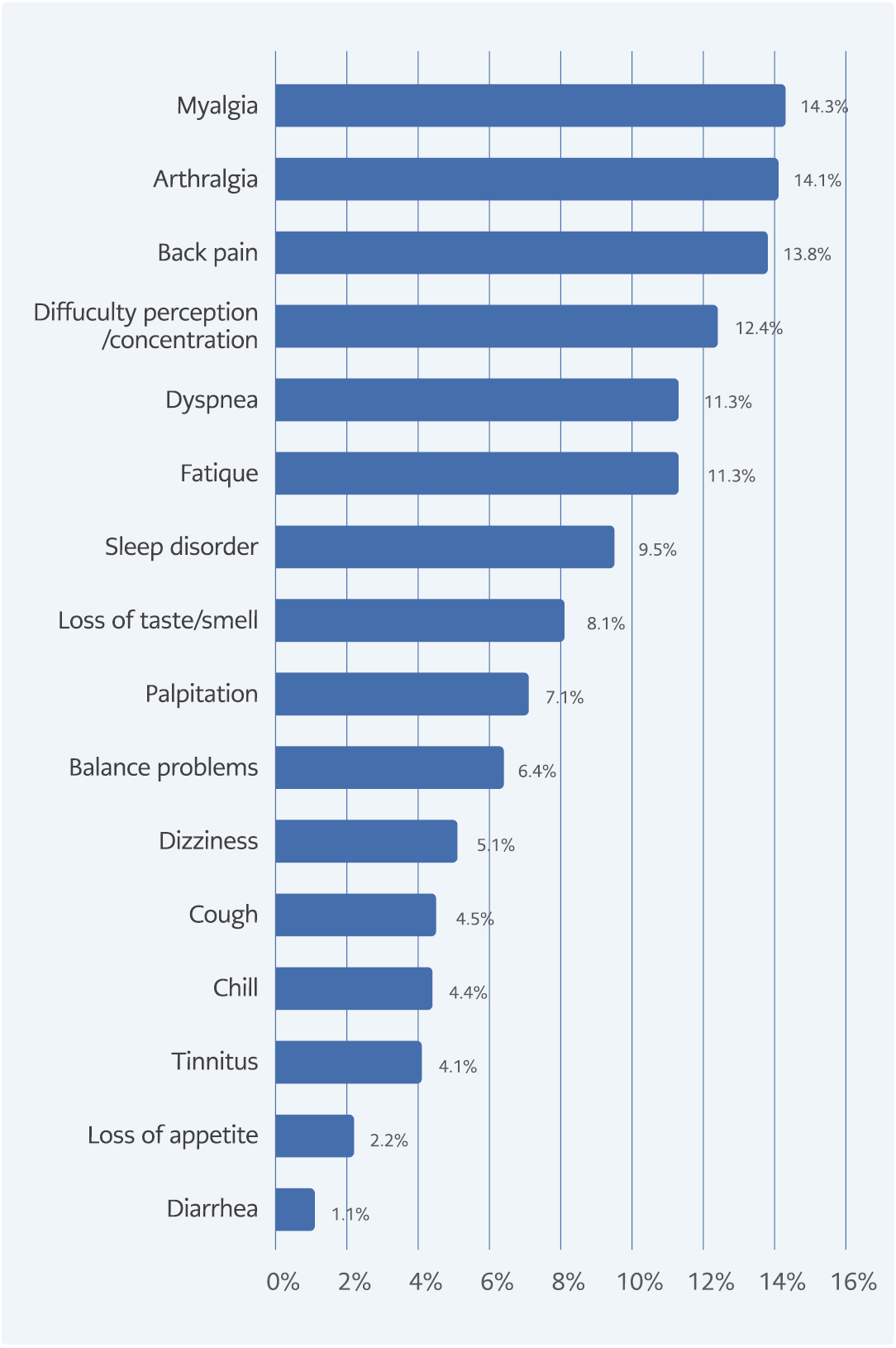
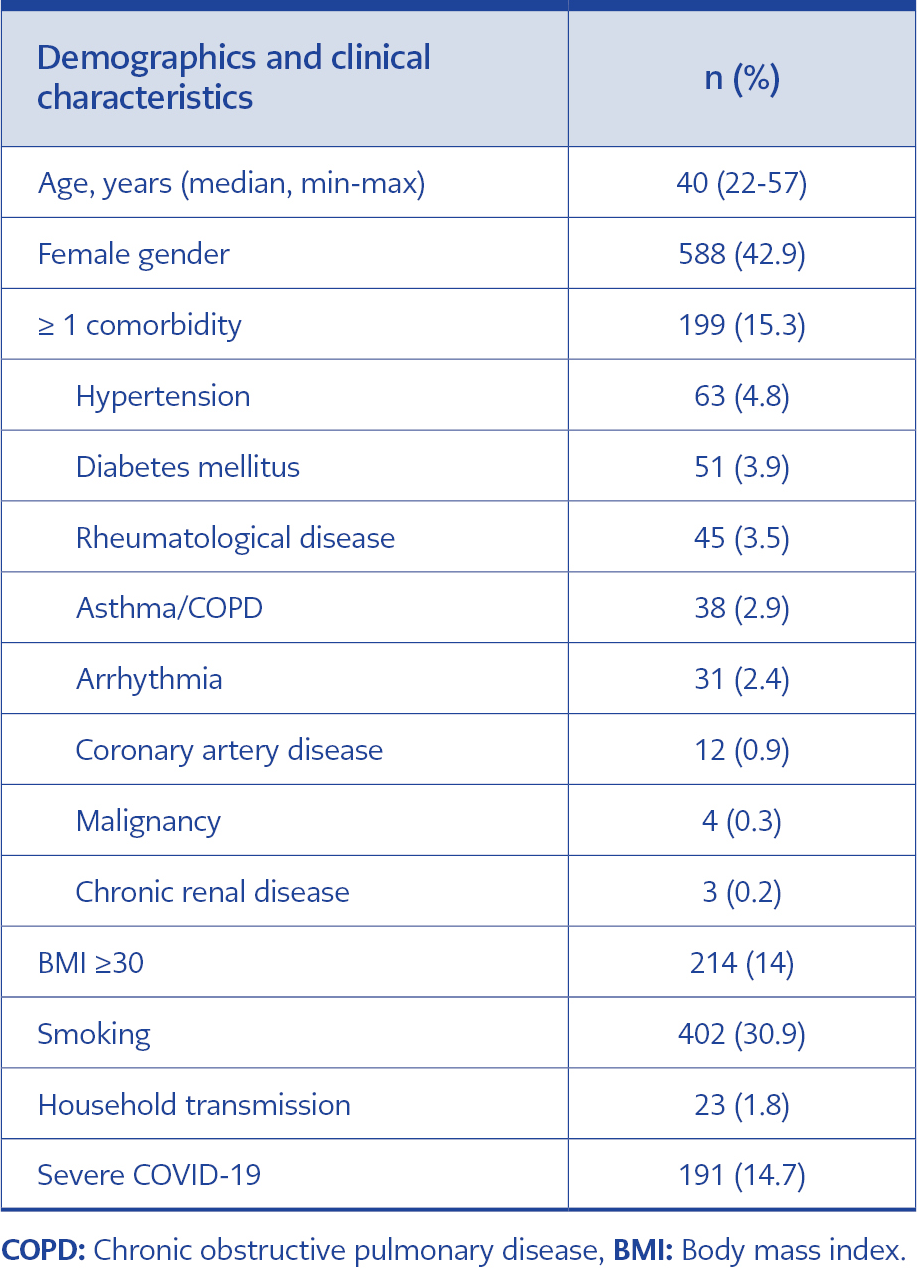
A total of 1301 patients participated in the survey. All of the participants answered the questions completely. There was not any patient who did not give informed consent or respond to the survey. The median age of participants was 40 (22-57), and 558 (42.9%) were women. The percentage of patients with a BMI ≥30 was 14%. One hundred ninety-nine (15.3%) patients had at least one comorbidity. The most common comorbidities were hypertension (4.8%), diabetes mellitus (3.9%), and rheumatological disease (3.5%). Eighty-five percent of the patients had mild-moderate disease, and 14.7% were followed up with severe COVID-19. Household transmission was only seen in 1.8% of the patients (Table 1). Two hundred fifty-seven (19.8%) patients were defined as ‘long COVID’ with the presence of ≥1 symptom persisting ≥3 months after COVID-19. Patients suffering from ‘long COVID’ reported a wide range of persistent symptoms. The most commonly observed symptoms among patients with ‘long COVID’ were myalgia (14.3%), arthralgia (14.1%), and back pain (13.8%) (Figure 1a-b). The musculoskeletal and neurological systems were the most commonly affected by the long-term effects of COVID-19.
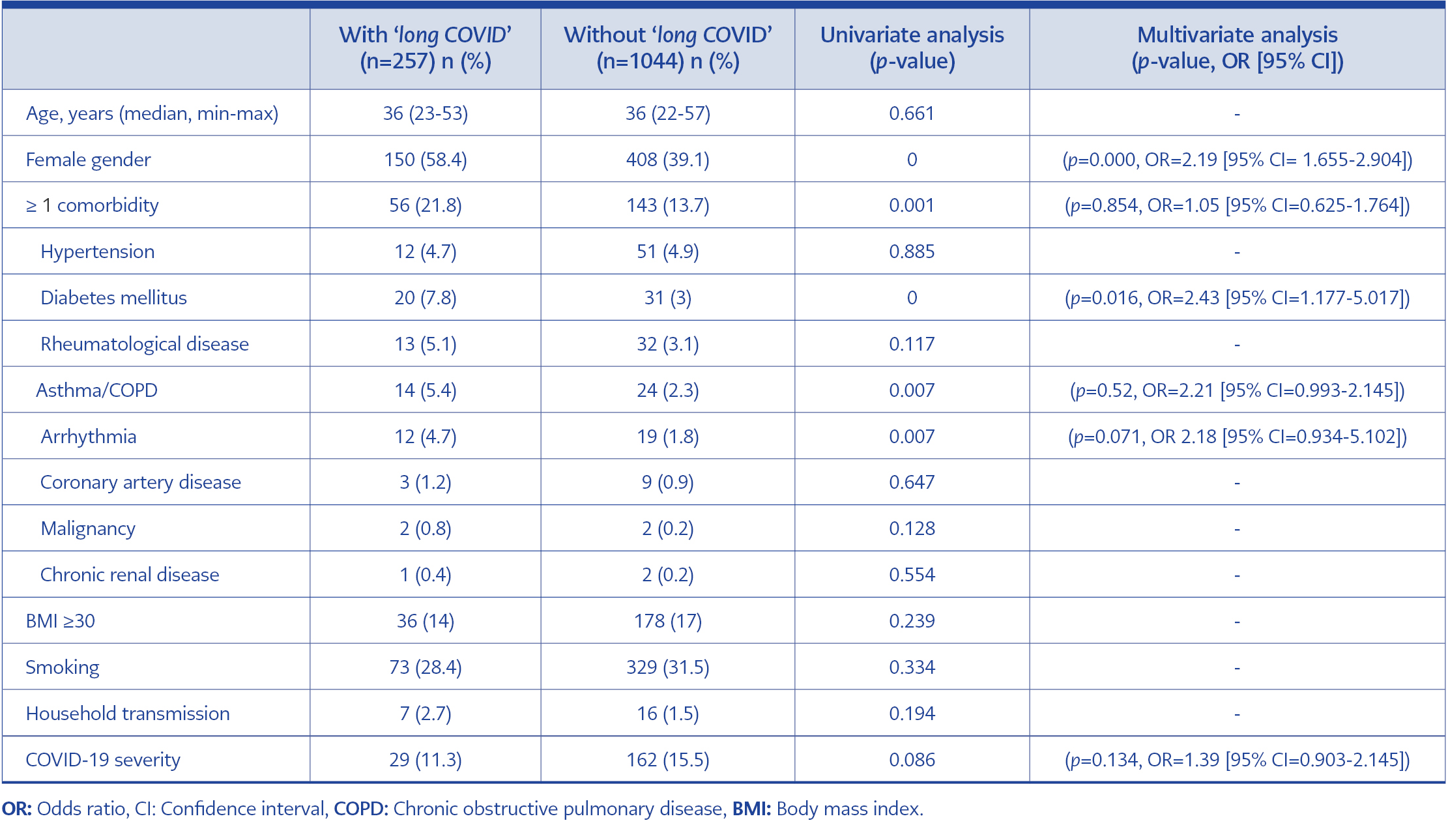
When the participants with or without ‘long COVID’ were compared, there was no significant difference between the two groups in terms of age and COVID-19 severity. The prevalence of being of female gender, having at least one comorbidity, diabetes mellitus, asthma/chronic obstructive pulmonary disease (COPD), and arrhythmia were higher in the patients with ‘long COVID’. Female gender (p=0.000, OR=2.19 [95% CI=1.655-2.904]) and diabetes mellitus (p=0.016, OR= 2.43 [95% CI=1.177-5.017]) were identified as independent risk factors for the development of ‘long COVID’ by multivariate logistic regression analysis (Table 2).
Discussion
The most recent number of people living with ‘long COVID’ has exceeded 65 million and is increasing daily (12). ‘long COVID’ has been associated with a broad range of signs/symptoms and public health impact (13). COVID symptoms lasting longer than three months resulted in a decreased quality of life in 57% of patients (2). ‘Long COVID’ is being seen in an increasing number of patients with persistent, debilitating symptoms after SARS-CoV-2 infection and have yet to be exactly explained by the mechanisms (14). This syndrome might also result from multisystemic involvement of COVID-19, immune dysregulation, autoimmunity, or neuroinflammatory events (15, 16).
The prevalence of ‘long COVID’ differs in each study because of the heterogeneity of studies, variability of definitions, and follow-up durations. In the literature, the frequency of patients with one or more prolonged COVID-19 symptoms is reported to be between 22% and 40% (17). The most commonly observed symptoms among patients with ‘long COVID’ were fatigue (31-58%), dyspnea (24-40%), musculoskeletal pain (9-19%), anosmia/dysgeusia (10-22%), and cognitive impairment (12-35%) (5, 15, 18-21). Although fatigue is one of the most frequent ‘long COVID’ symptoms, its prevalence in our cohort was low (11.3%); however, the range of rate reported in patients with ‘long COVID’ was extensive. In parallel with our results, the rate of fatigue was reported as 14.7% in a study by Augustin et al. (22). However, Amin et al. reported that fatigue was seen in 39.5% of patients, and this high rate was associated with underlying comorbidities (23). The low rate of fatigue in our study could be explained by our participants’ younger age and low incidence of comorbidities. Karaarslan et al. showed that 43.2% of participants had at least one rheumatic and musculoskeletal symptom at six months (24). Similarly, musculoskeletal symptoms (total 43.2%) consisting of myalgia (14.3%), arthralgia (14.1%), and back pain (13.8) were the most commonly described long-term effects of COVID-19 in our study.
There was a broad range of risk factors for the development of ‘long COVID’ in the literature. Previous studies suggested that the risk for ‘long COVID’ is higher in female sex, older age, patients who need hospitalization or oxygen therapy, and patients with comorbidities such as asthma (13, 25-27). The studies also have conflicting results regarding the risk factors for ‘long COVID’. For example, ‘long COVID’ symptoms have been increasingly reported by patients even with mild disease (who did not require hospitalization, oxygen or intensive care support) (14). Also, in our study, there was no significant difference between the two groups regarding COVID-19 severity. The fact that mild or severe disease has no effect on the development of ‘long COVID’ shows the importance of long-term effects of the disease beyond the acute infection burden.
Iwasaki et al. reported that people with ‘long COVID’ after non-severe COVID-19 are most likely to be younger women (36-50 years) whose acute infection could trigger adverse physiological responses. On the other hand, patients who survived after severe infection are mostly older men (50 years) with lingering tissue damage and scarring (7). Most of our participants who had ‘long COVID’ were young (median age of 36) women (58.4%) and suffered non-severe acute COVID-19 (88.7%). Female gender (p=0.000) and diabetes mellitus (p=0.000) were found as independent risk factors for ‘long COVID’. The pathophysiology and causes of ‘long COVID’ in our participants could be explained with these mechanisms.
Our study has several limitations. First, it is a retrospective study with a small number of patients. Due to the retrospective design and depending on participants’ self-reports, the memory factor may have caused bias in the answers. Second, the majority of the participants had mild diseases and were of younger age; these characteristics may also have affected the study results. In the literature, studies emphasize the importance of vaccination in the prevention of ‘long COVID’. However, we could not determine the impact of vaccination on the development of ‘long COVID’.
In conclusion, the fact that mild or severe illness has no effect on ‘long COVID’ points to the importance of long-term effects of the disease beyond acute infection burden. In order to detect ‘long COVID’ patients early and to provide appropriate multidisciplinary rehabilitation, it is important to know which factors put patients at risk for ‘long COVID’.