Introduction
In early December 2019, interstitial pneumonia of unknown origin emerged in Wuhan, the capital of China’s Hubei province. The pathogen was identified as a novel beta coronavirus called severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and the disease caused by SARS-CoV-2 was later named coronavirus disease 2019 (COVID-19) (1). As of writing this article, more than 400 million confirmed cases and over five million deaths were reported worldwide (2). Around 13 million confirmed cases and more than 90 thousand deaths were reported in Turkey (3). Meanwhile, nearly 40 years after the virus became known, human immunodeficiency virus (HIV) infection continues as a global health epidemic. Since the epidemic’s beginning, approximately 76 million people have been infected with HIV, and millions have died from HIV-related causes. By 2020, there were about 38 million HIV-positive patients globally, 28,000 being in Turkey. It is estimated that 690,000 people died from HIV-related causes in 2020 (4, 5). As the COVID-19 pandemic spread globally, more HIV-positive patients became infected with SARS-CoV-2. Our knowledge of the simultaneous management of co-infected patients and the course of COVID-19 in HIV infection is limited. Prolonged inflammation due to cytokine release is a known mechanism in HIV infection, and it has been associated with increased intestinal permeability and bacterial translocation. Proinflammatory cytokine serum levels have been independently associated with morbidity and mortality in HIV-positive patients (6, 7). In COVID-19, the massive and uncontrolled cytokine release known as cytokine storm plays a critical role in acute respiratory distress syndrome and multiple organ dysfunction (8). Due to the overlapping pathophysiological characteristics of HIV and SARS-CoV-2 infections, the management of co-infected individuals is a unique situation.
Studies have identified obesity/high body mass index (BMI), as a risk factor for severe COVID-19, possibly due to chronic inflammation that impairs immune responses to pathogens (9,10). It was also reported that the risk of severe COVID-19 is higher in older people and those with comorbidities such as cardiovascular disease, chronic lung disease, and diabetes (11). However, our knowledge about the effect of these clinical conditions on the course of HIV and SARS-CoV-2 among co-infected patients is limited.
We aimed to share our experiences among HIV and SARS-CoV-2 co-infected patients followed at the tertiary referral hospital in the Black Sea region, Turkey.
Materials and Methods
Within the scope of the study, medical records of HIV and SARS-CoV-2 co-infected patients diagnosed between March 2020 to June 2021 were analyzed retrospectively.
The diagnosis of COVID-19 was made by detecting SARS-CoV-2 ribonucleic acid (RNA) by real-time polymerase chain reaction (RT-PCR) testing of nasopharyngeal and/or oropharyngeal swab samples individuals suspected of COVID-19 using Biospeedy® SARS CoV-2 Triple Gene RT-qPCR (Bioeksen R&D Technologies Inc., İstanbul, Turkey) kits.
Patients with any signs and symptoms such as fever, headache, sore throat, cough, weakness, loss of taste and smell without shortness of breath or with normal lung radiological imaging were defined as patients with mild COVID-19. Severe COVID-19 criteria were specified as respiratory frequency above 30/min, SpO² below 94% in room air, PaO²/FiO² below 300 mmHg, or more than 50% infiltration in the parenchyma on radiological imaging (12).
BMI was defined as body weight divided by body height and expressed in kg/m2. Patients were classified as underweight (under 18.5 kg/m2), normal weight (18.5 to 24.9 kg/m2), pre-obese (25 to 29.9 kg/m2), and obese (above 30 kg/m2) based on the calculated value (13).
All HIV-positive patients on antiretroviral therapy (ART) continued their current treatment throughout the course of COVID-19.
The Ethics Committee of Ondokuz Mayıs University approved the study with the decision number of 2021/25.
Results
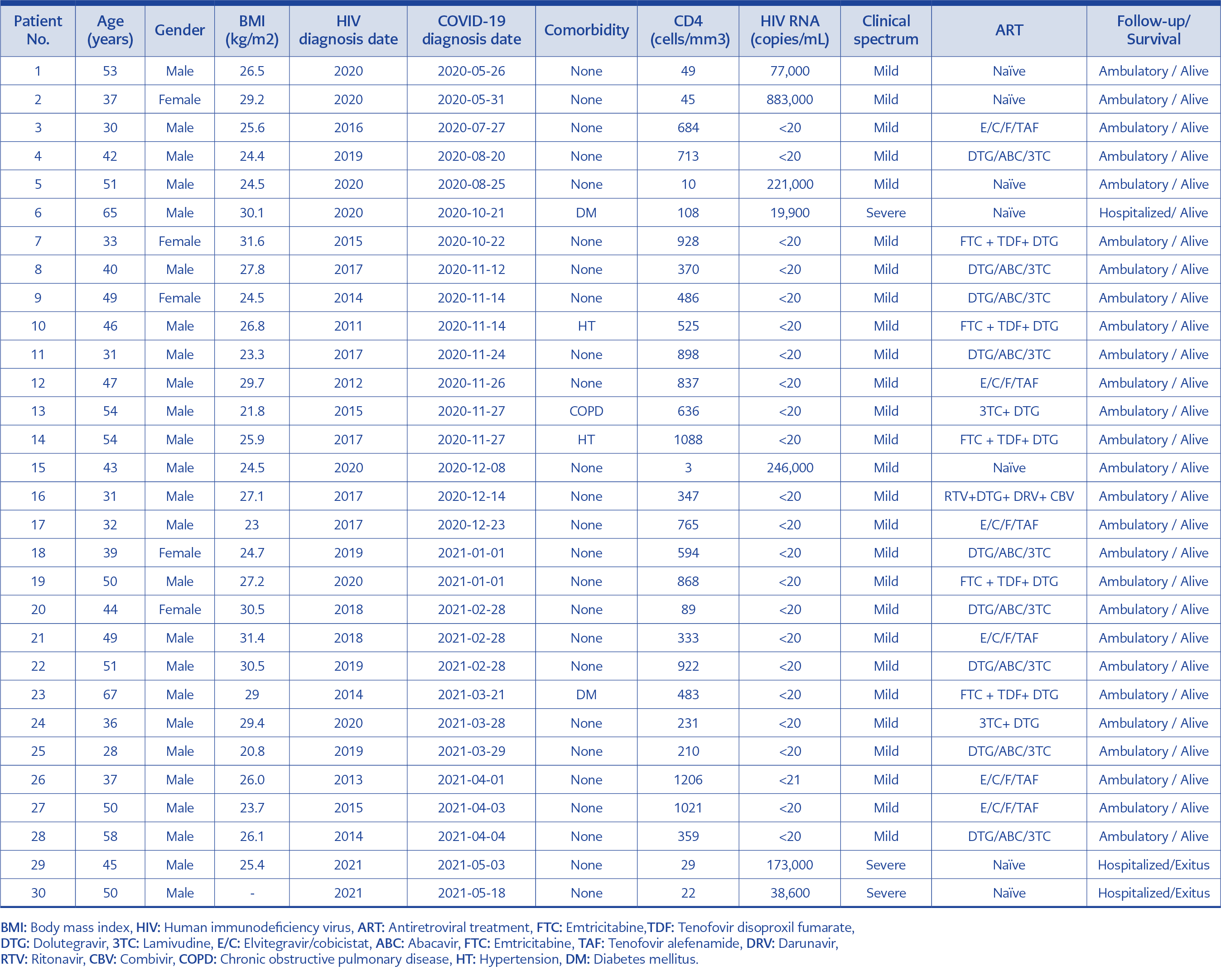
During the study period, 30 HIV and SARS-CoV-2 co-infected patients were followed. Five (16.7%) patients were female, 25 (83.3%) were male, and the mean age was 44.5 ±10.2 years. Twenty-three (76.7%) patients were known to be HIV-positive before admission to the hospital; in seven (23.7%) patients, the diagnosis of HIV infection was made in the routine examinations performed after the diagnosis of COVID-19.
All patients known to be HIV-positive were on ART and virologically suppressed (HIV-RNA <20 copies/ml). The mean CD4 T cell count among ART-experienced patients was 634±305 /mm³ and 38±35 /mm³ in ART-naïve patients. The median HIV-RNA viral load was 173,000 (min.19,900, max. 883,000) copies/ml for newly diagnosed HIV-positive patients.
Twenty-seven (90%) patients, five (17%) female and 22 (73%) male, presented with mild symptoms and were not hospitalized. Among non-hospitalized patients, 24 (85.1%) were on ART, and three (14.9%) were ART-naïve. Three (10%) patients who met the criteria for severe COVID-19 were hospitalized. Hospitalized patients were male and ART-naïve, and among them, two (6.7%) had died.
BMI values of 29 patients were available, and the mean BMI was found to be 26.5±2.9 kg/m2. The mean BMI for 27 outpatients was 26.5±2.9 kg/m2. Two of the three hospitalized patients had a BMI value, and the mean BMI was 27.7±3.3kg/m2.
At the time of COVID-19 diagnosis, five patients had a history of comorbidity. Two patients had arterial hypertension, two had diabetes mellitus (DM), and one had chronic obstructive pulmonary disease (COPD). Except for one of these patients (Patient No: 6), who was ART-naïve and had an uncontrolled DM, the other four patients were both on ART and medication for comorbidities (Table 1).
Discussion
Lower CD4 T cell count is expected in COVID-19, and lymphopenia is more pronounced in severe cases (14,15). In this context, it has been predicted that low CD4 T cell counts seen in advanced stages of the HIV infection will even be more manifested in SARS-CoV-2 co-infected patients and may lead to more severe immunological and clinical consequences (16). However, despite this prediction, studies on the clinical outcome of co-infection with COVID-19 and HIV have yielded mixed results.
A large-scale population-based retrospective cohort study from the United Kingdom (UK) found that HIV-positive patients have a higher mortality risk of COVID-19 (17). To evaluate the epidemiology of SARS-COV-2 and HIV co-infection and to detect mortality associated with COVID-19, in a systematic study involving 22 studies with 20,982,498 participants in North America, Africa, Europe, and Asia, HIV-positive people have been found to have a significantly higher risk of SARS-CoV-2 infection and death from COVID-19 than HIV-negative individuals (18). In a cohort study conducted in New York State, United States (US), people diagnosed with HIV infection experienced worse COVID-19-related outcomes than people who were not diagnosed with HIV. A previous HIV infection diagnosis was associated with higher rates of serious illness requiring hospitalization, and the advanced HIV infection stage was found to increase the risk of hospitalization (19). In a cohort study conducted with HIV-positive and HIV naïve COVID-19 patients admitted to a hospital in Wuhan between January and April 2020, it was found that the positive conversion rate of IgG for SARS-CoV-2 was lower in HIV-positive patients, which indicated that HIV-positive patients were in a disadvantaged situation when affected with COVID-19 (20).
Contrary to the study mentioned above, there are also studies reporting limited or no effect of HIV infection on the clinical course and survival of COVID-19. A retrospective cohort study of all COVID-19 suspected and confirmed cases hospitalized in Iran found that HIV infection was not a risk factor for increasing disease severity and risk of death in COVID-19 (21). A study evaluating patients with COVID-19 in acute care hospitals in New York, US, between March 2020 and April 2020 found that HIV-positive patients may not experience significantly worse outcomes from SARS-CoV-2 infection than non-HIV patients (22). Retrospective analysis of 32 SARS-CoV-2 and HIV co-infected patients confirmed at German HIV centers between March and April 2020 showed that 91% of patients recovered, and 76% were classified as mild cases. Study findings did not support excessive morbidity and mortality in symptomatic patients (23).
In our study, mortal cases (patient no: 29 and patient no: 30) were ART-naïve, severely immunosuppressed, had no comorbidities and were male. However, for patients with similar characteristics (Patient no: 1 and patient no: 15), the course of COVID-19 was mild, and the patients followed up ambulatory. Considering the immunological status of these aforementioned patients, it seems unlikely that HIV infection, which is the cause of immunosuppression, is alone responsible for mortality in COVID-19 co-infection.
In this study, the absence of death or severe clinical course in any of the patients on ART suggested that ART may have a protective effect on COVID-19. Antiviral activity of tenofovir (TFV) against SARS-CoV-2 has been demonstrated in in-vitro studies (24). In addition, a case-control study on middle east respiratory syndrome (MERS) reported that lopinavir/ritonavir (LPV/r) might be effective as post-exposure prophylaxis in healthcare workers (25). However, a higher rate of COVID-19 infection was reported among HIV positive patients on ART, including tenofovir alafenamide (TAF) or tenofovir disoproxil fumarate (TDF) users, in a prospective cohort in Spain (26). In addition, a randomized open-label study in 199 adults with severe COVID-19 found no clinical or virological benefit with lopinavir/ritonavir (27). Therefore, until now, there is no clear evidence that the use of ART agents can treat or prevent COVID-19, and it would not be accurate to say that the mild course of COVID-19 and the absence of mortality in patients on ART in our study can be attributed to ART. In our study, all patients on ART survived, compared to 72% of ART-naïve patients. Therefore, more broad-based assessments are needed to determine whether ART impacts the course of COVID-19.
Studies have shown that the course of COVID-19 is more severe in patients with comorbid conditions (28). In a study that analyzed data from 1590 laboratory-confirmed hospitalized patients from 575 hospitals in China between December 2019 and January 2020, patients with comorbidities such as COPD, DM, hypertension, and malignancy had worse clinical outcomes than those without. An increasing count of comorbidities reported being correlated with poorer clinical outcomes (29). In a study conducted in Turkey in which four cases were evaluated, it was reported that SARS-CoV-2 and HIV co-infection improved in both ART experienced and ART-naïve cases, and it was concluded that comorbidities are an important factor in survival in oo-infected cases (30). In our study, there were five COVID-19 and HIV co-infected patients diagnosed with hypertension, COPD, and DM. Among these patients, the ART-naïve 65 years old male obese patient (patient no: 6) with an uncontrolled DM had a severe clinical course and required hospitalization, including a period in the intensive care unit. However, the other ART-experienced patients with comorbid conditions or similar BMI had mild symptoms and did not require hospitalization. Considering the case, we concluded that uncontrolled DM was mainly responsible for the severe clinical course rather than the effect of HIV infection or BMI.
In conclusion, HIV infection or the immune status alone does not increase the severity of the COVID-19 course and does not increase the mortality of COVID-19. The comorbidities of the patients, such as uncontrolled diabetes, have an impact on the disease course.