Introduction
The COVID-19 pandemic emerged in Wuhan in December 2019 and spread rapidly all over the world. The number of cases worldwide reached a total of 150 million as of May 2021 (1). While 14% of these cases required hospitalization with severe pneumonia, 5% required intensive care support. The mortality rate was shown as 2.3% (2).
Cytokine storm that might cause acute respiratory distress syndrome (ARDS), multiple organ failure, and mortality plays an important role in the course of severe COVID-19 cases (3). Interleukin-2 (IL-2), IL-2R, IL-4, IL-6, IL-8, IL-10, tumor necrosis factor-alpha (TNF-α), and interferon-gamma (INF-γ) are associated with severe COVID-19 cases (3-5). Thus, anti-cytokine therapies are widely added to the standard treatments (such as antivirals, anticoagulants, corticosteroids) in COVID-19 cases (6).
The most commonly used anti-cytokine therapies are IL-6 antagonists (tocilizumab). Although many clinical studies have evaluated the treatment efficacy of tocilizumab, different results have been reported about the treatment responses. In the initial published randomized controlled trials, no association was found between tocilizumab treatment and COVID-19 patient survival (7-9). However, contrary to those studies, tocilizumab has been associated with a lower frequency of intensive care unit (ICU) admissions, mechanical ventilation requirement, and mortality in recent randomized controlled trials and cohort studies based on real-life data (10-13). Based on the recent results, tocilizumab is identified among the suggested treatments for use in COVID-19 management guidelines (14, 15).
This study aimed to evaluate the impact of tocilizumab treatment on clinical improvement in severe COVID-19 cases and the factors affecting clinical response.
Methods
Study Design
This single-center, retrospective, descriptive study was conducted in hospitalized severe COVID-19 patients between 20 March and 01 December 2020.
Study Populations and Definitions
All patients older than 18 years old who were admitted with a diagnosis of severe COVID-19 pneumonia or met the criteria of severe COVID-19 pneumonia at follow-up and whose standard COVID-19 treatment were added tocilizumab were included in the study. In addition to pneumonia confirmed by chest imaging, the presence of oxygen saturation (SaO2) ≤ 90%, or the ratio of the partial pressure of oxygen to the fraction of inspired oxygen (PaO2/FiO2) ≤ 300, or respiratory rate ≥ 30 per minute were considered as severe COVID-19 disease (16).
Standard treatment was defined as combination therapy consisting of antiviral (hydroxychloroquine or remdesivir favipiravir), corticosteroid (6 mg/day dexamethasone or 40 mg/day prednisolone), and low molecular weight heparin (LMWH). Patients receiving treatment other than standard treatment before tocilizumab therapy (pulse corticosteroid, convalescent plasma, or IL-1 antagonist) were excluded from the study.
Study Protocol
During the study period, all eligible participants were enrolled consecutively. The patients’ demographical features, clinical characteristics, and laboratory results (hemograms, serum, D-dimer, ferritin, CRP levels) were recorded on the first day of tocilizumab treatment. The ordinal scale of clinical status scores for each patient was calculated and recorded on the first day of tocilizumab treatment, as shown in Table 1 (17). The same scale score was calculated on the 1st, 3rd, and 7th days of tocilizumab treatment and compared with the onset score of treatment. A decrease in the ordinal scale of clinical status score of ≥ 2 points was evaluated as a clinical response compared to the onset of treatment.
Statistical Analysis
All data were analyzed by IBM SPSS Statistics for Mac, version 25.0 for Mac OS X (IBM Corp., Armonk, N.Y., USA). The normality of the data distribution was determined by the Shapiro-Wilk test, histogram, and Q-Q plots. The categorical values of the patients were expressed as a number and a percentage; the continuous values were presented as a mean and standard deviation (SD) or median values and an interquartile range (IQR) of 25%–75%. The categorical values were analyzed with a chi-square test or Fisher exact test. The non-parametric values were analyzed using the Mann–Whitney U test and the parametric ones with the Student t-test. The ordinal scale of clinical status scores calculated on different days for the same patient was compared with Wilcoxon tests. The correlation between the clinical score changes between the 0th and 7th days of tocilizumab treatment and the changes in some laboratory parameters was evaluated by the Pearson correlation analysis. On the 7th day of tocilizumab treatment, patients were grouped as responders and non-responders and compared in terms of possible risk factors. Variables with a p-value of <0.2 in univariate analysis and not showing a high level of correlation with other variables were included in logistic regression models. When appropriate, 95% confidence intervals (95% CIs) were also calculated, and statistical significance was set as p <0.05.
Results
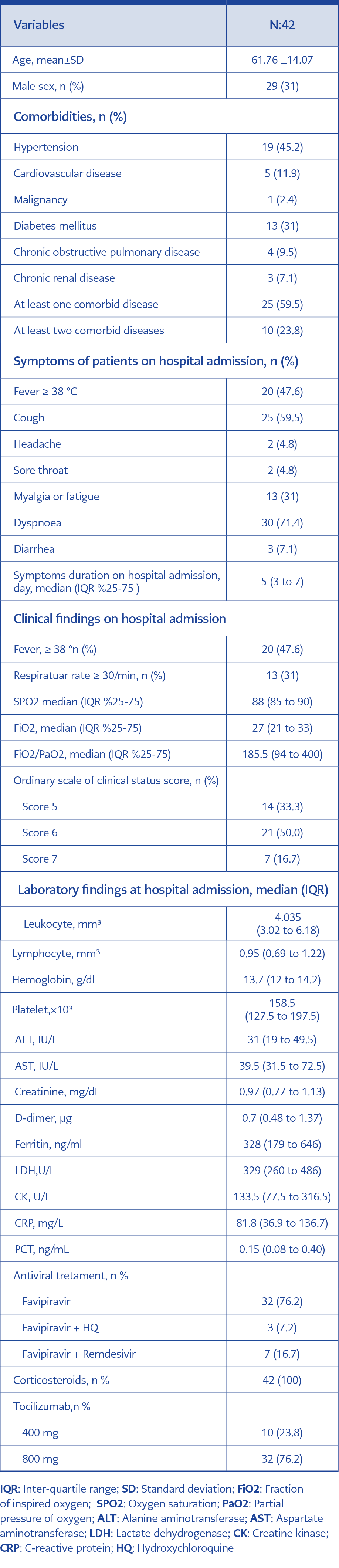
Forty-two patients were enrolled in the study. The demographical, clinical, and laboratory findings of patients at hospital admission was shown in Table 2.
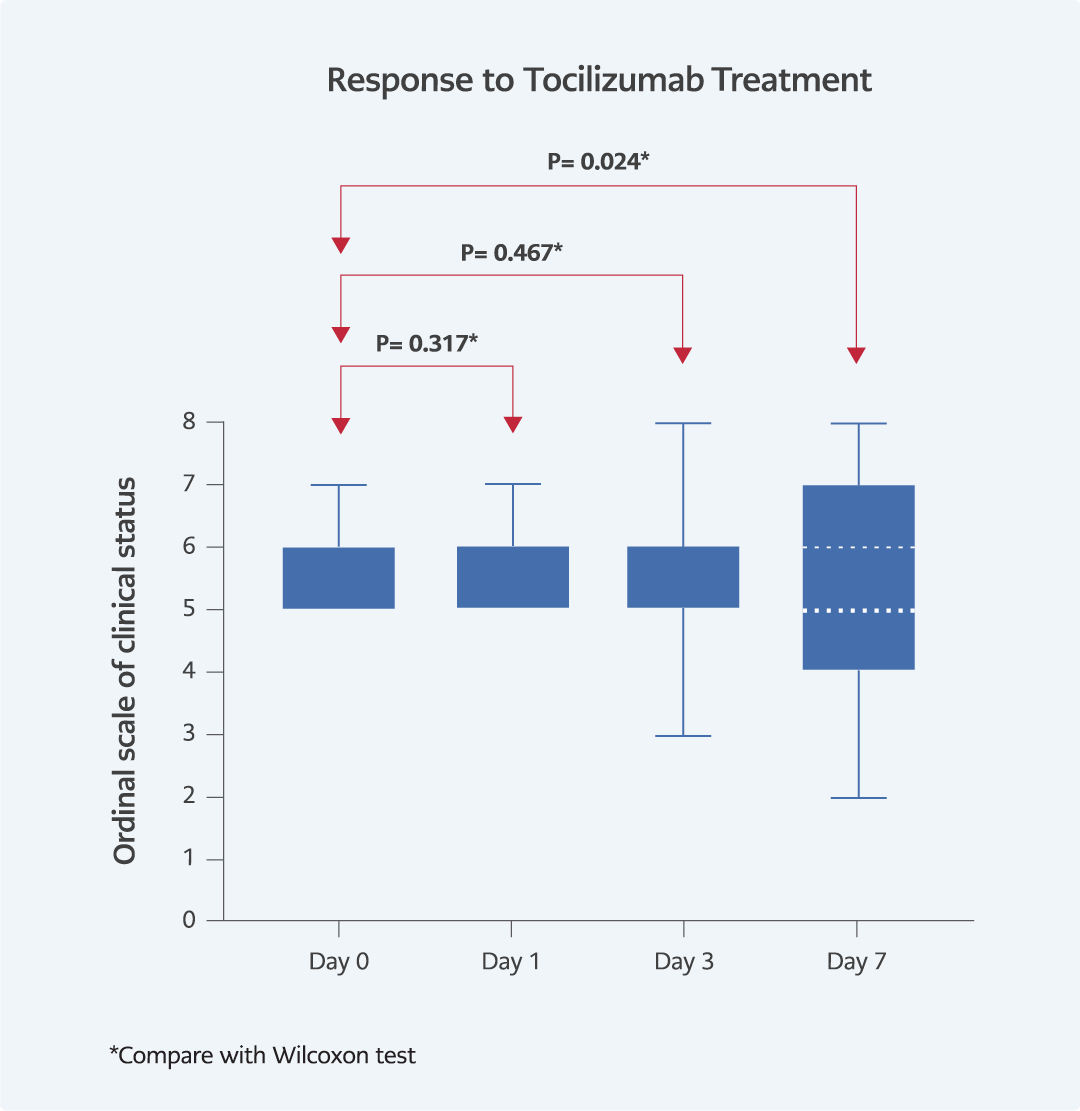
The median of the ordinal scale of clinical status scores (day 0) of the patients on the tocilizumab treatment days was 6 (5 to 6). The median of the ordinal scale of clinical status scores on the 1st, 3rd, and 7th days of tocilizumab treatment were 6 (5 to 6), 6 (5 to 6), and 5 (4 to 7), respectively. According to the comparison of the ordinal scale of clinical status score, there was no statistically significant difference between day 0 and day 1 (p=0.317, Wilcoxon, figure 1), day 0 and day 3 (p=0.467, Wilcoxon, as shown in the figure). When compared with the ordinal scale of clinical status score on day 0 of tocilizumab treatment, a statistically significant difference was found on the 7th day (p=0.024, Wilcoxon, as shown in the figure). Median of lymphocyte counts, ferritin, CRP, D-dimer levels on day 0 of tocilizumab treatment were 0.70 (0.48 to 1.00), 499 (315 to 667), 91.4 (52.7 to 136.5) and 0.71 (0.45 to 1.54), respectively. The median of lymphocyte counts, ferritin, CRP, D-dimer levels on the 7th day of tocilizumab treatment was previously 0.90 (0.74–1.52), 350 (350-428), 5.04 (3.92 – 6.76), and 2.05 (81.03 – 2.05). There was a significant difference in lymphocyte counts (p<0.001, Wilcoxon), ferritin (p=0,03, Wilcoxon), CRP (p<0.001, Wilcoxon) and D-dimer (p=0.05, Wilcoxon) levels between days 0 and 7 of tocilizumab. The correlation between the differences in the ordinal scale of clinical status score and the differences in lymphocyte count, ferritin, CRP, and D-dimer between the 0th and 7th days of tocilizumab treatment was evaluated and the Pearson correlation coefficient was -0.179 (p=0.255), -0.205 (p=0.193), -0.70 (0.48 to 1.00), -0.174 (p=0.269), 0.70 (0.48 to 1.00), 0.320 (p=0.039), respectively.
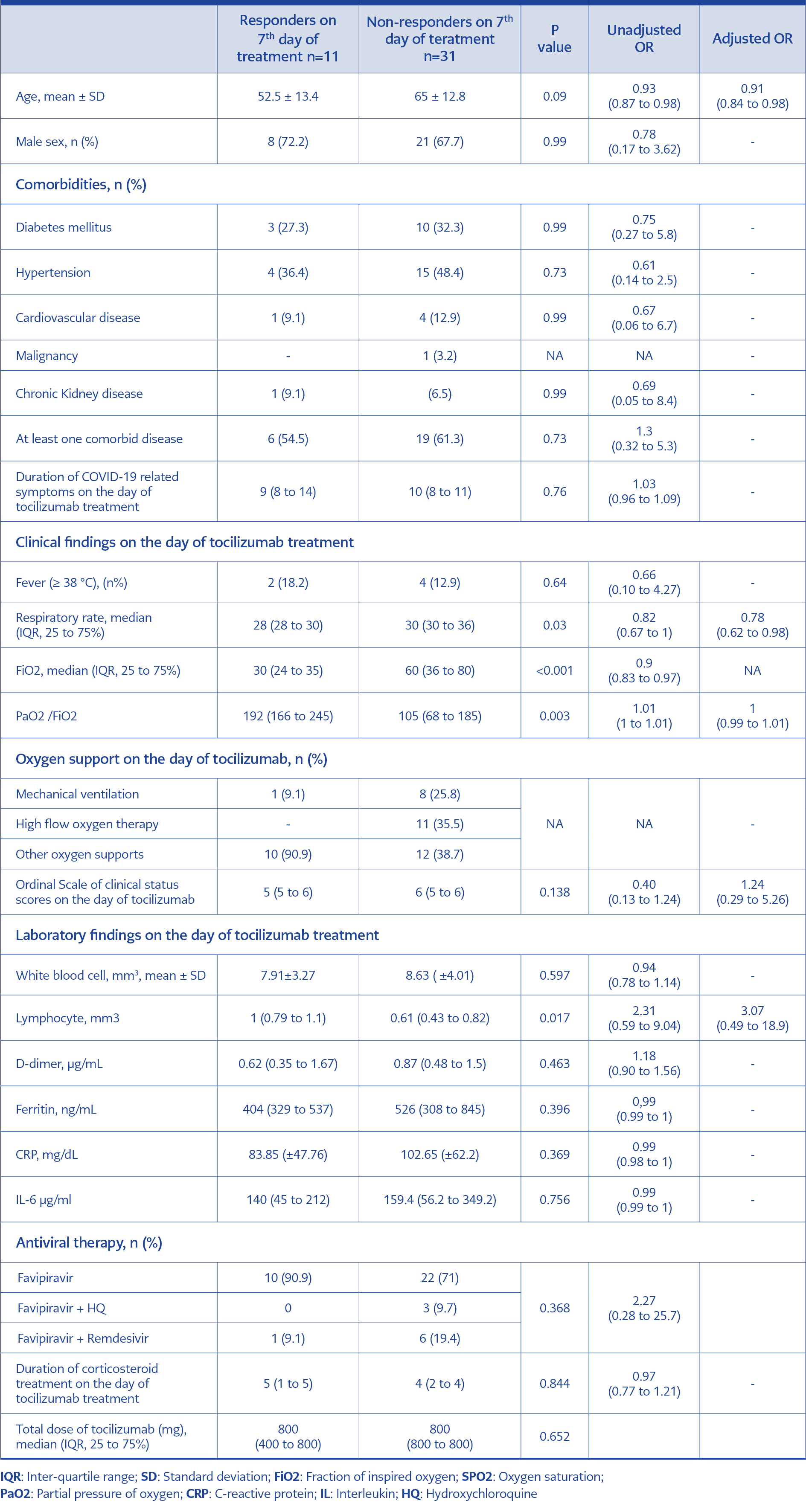
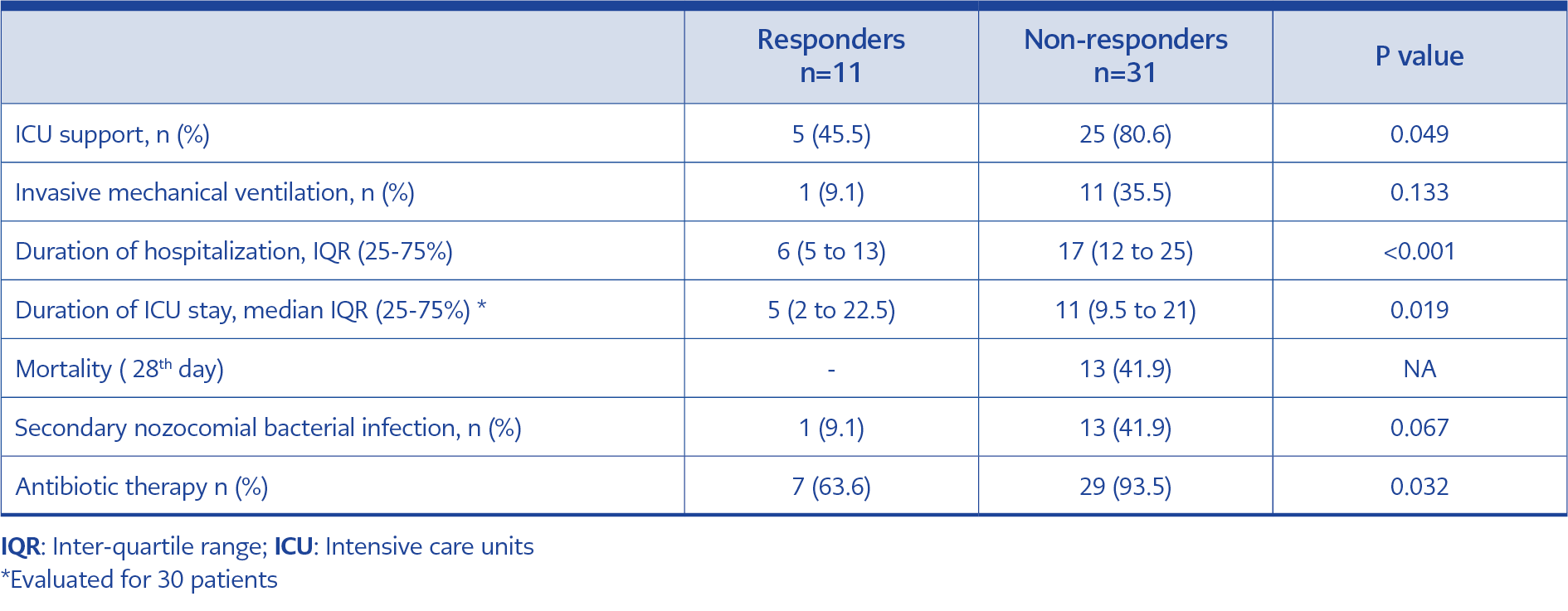
On the 7th day of tocilizumab treatment, the clinical response rate was 26.2% (n=11). The patients were grouped as responders and non-responders according to their clinical responses on the 7th day and compared possible risk factors, as shown in Table 3. Age (OR: 0.91, 95% 0.84-0.98) and respiratory rate (OR: 0.78, 95% 0.62-0.98) at the time of treatment were determined as factors affecting tocilizumab treatment response. Responders and non-responders were compared in terms of 28-day mortality, ICU support requirement, invasive mechanical ventilation, intensive care and hospital stay, secondary bacterial infection, and antibiotic use, as shown in Table 4.
Discussion
In our study, tocilizumab treatment was related to clinical improvement in one-fourth of severe COVID-19 cases. Being young and having low oxygen requirements (low respiratory rate and low oxygen requirement) is associated with the tocilizumab response. Patients with clinical improvement stayed shorter in hospital and ICU, needed low ICU support, and resulted in low mortality rates. No relationship was detected between laboratory parameters before tocilizumab treatment, including IL-6 levels, and clinical response.
In initial randomized controlled trials that used different inclusion and outcome criteria, the effect of tocilizumab treatment on patients’ survival was not detected (8, 9, 13, 18, 19). However, observational studies evaluating the efficacy of tocilizumab and meta-analysis of these studies showed that the tocilizumab therapy was associated with decreased oxygen support, mechanical ventilation requirement, and mortality rates (6, 7, 20-27). Current randomized controlled trials support this real-life observational data. In recent randomized trials, tocilizumab treatment is associated with shorter hospital and ICU stays reduced need for mechanical ventilation, and prolonged survival (10-13).
In the first week of the treatment, we achieved clinical improvement in one-fourth of the patients with tocilizumab therapy. However, we could not demonstrate the effect of tocilizumab on clinical improvement in the early period (day 1 and day 3) of treatment.
In some studies, the time of adding tocilizumab to the standard therapy was associated with the clinical response. The use of tocilizumab in the early period increased clinical response rates in patients with lower oxygen requirements, especially before the need for intensive care support or mechanical ventilation (8, 21, 25, 28, 30). Similarly, our study found that the frequency of clinical response increased with the use of treatment tocilizumab in young patients with lower oxygen requirements (low respiratory rate and high PaO2 / FiO2). However, there was no relationship between tocilizumab treatment response and the duration of COVID-19 symptoms. Therefore, tocilizumab should be added early based on clinical evaluation and increase in oxygen supply rather than disease and symptom duration.
Laboratory values such as ferritin, CRP, D-dimer, LDH, IL-6 levels, which are used to identify cytokine storm and are associated with severe disease and poor prognosis, are widely used for tocilizumab treatment indications (21, 29, 31-33). Current guidelines recommend CRP 75 mg/L cut-off values for defining systemic inflammation and the indication for tocilizumab therapy in severe COVID-19 patients (14, 15). However, the effect of these laboratory values in predicting the clinical response to tocilizumab treatment and the uncertainties in the limit values to be used for the treatment indication remain. In the study of Fernadez-Luiz et al., no relationship was shown between laboratory parameters other than LDH and clinical response to tocilizumab (7). Also, in our study, there was no relationship between the pre-treatment laboratory parameters of tocilizumab, including IL-6, and clinical response to tocilizumab.
One of the concerns for tocilizumab treatment in COVID-19 patients is the increased risk of secondary bacterial and fungal infections. In some small cohort studies, tocilizumab was associated with secondary nosocomial infections, especially when used in combination with steroids (34-36). However, there was no relationship between secondary infection and tocilizumab in randomized controlled studies and meta-analysis (6). In our study, secondary bacterial infection developed in one-third of the patients who received tocilizumab treatment. However, due to the absence of a comparison group in our study, the effect of tocilizumab use on the development of secondary bacterial infection could not be evaluated.
Our study has some limitations. First, since only patients using tocilizumab were included in the study, no comparison could be made regarding efficacy, side effects, and development of complications. Second, because there was no control group, we could not rule out the possibility that our results are dependent on the natural course of the disease or some known prognostic factors (such as age). Finally, because the study was based on the limited number of patient data from a single-center, obtained statistical results had a high risk of type 2 errors, and therefore the results could not be generalized.
In conclusion, using tocilizumab in younger patients who need low oxygen support increases clinical responses after treatment. However, since the effect of laboratory parameters in predicting the clinical response of tocilizumab is limited, treatment indications should be determined based on clinical parameters.