Introduction
Brucellosis is a zoonotic infectious disease caused by Gram-negative bacteria of the genus Brucella, which is transmitted from animals to humans by direct and indirect pathways. Brucellosis can be confused with various infectious and non-infectious diseases because of its nonspecific clinical signs and symptoms such as fever, sweating, fatigue, loss of appetite, headache, muscle and joint pain (1-3). The diagnosis is based on isolation of the microorganism from clinical samples, demonstration of antibody response in serological methods, or specific nucleic acid determination by molecular methods.
The definitive diagnosis of brucellosis requires isolation of the infectious agent from blood, bone marrow, cerebrospinal fluid samples, and from other clinical samples, including stool, wound, and joint fluid. However, the isolation rate of the bacteria may vary depending on the stage of the disease, previous antibiotic usage, the type and amount of the clinical sample, the type of bacteria, and the culture technique used. The positivity rate of blood cultures, therefore, varies between 15-90%. Bone marrow cultures have been shown to be more sensitive and have a shorter isolation time than blood cultures (1-4). Especially in chronic cases, the probability of isolating the agent is low. Thus, serological methods are gaining popularity in the diagnosis of brucellosis (5).
The specificity and sensitivity of the serological tests used in the diagnosis may also be affected by the stage and the severity of the disease (1). Rose Bengal test (RBT), Standard Tube Agglutination Test (STA), Standard Tube Agglutination Test with Coombs (C-STA), Immunocapture Agglutination (ICA) test, ELISA and newly developed Brucella Coombs Gel test (BCGT) are frequently used in the serological diagnosis of brucellosis. BCGT is designed for the rapid detection of the antibodies against Brucella bacteria in human serum by the Coombs method. BCGT, which is carried out in wells containing gel matrix and Coombs antibody, allows performing STA and C-STA together, without the need for a long time for incubation. Beyond being a simple test in serological diagnosis, BCGT results demonstrate high consistency with the results of the C-STA test (6). In this study, we aimed to determine the diagnostic value of BCGT in comparison with STA and ELISA methods.
Materials and Methods
Patients who admitted to Haydarpaşa Numune Training and Research Hospital, Department of Infectious Diseases and Clinical Microbiology between February 2016 and February 2017 were included in the study. Patients who were under 18 years old or pregnant were excluded from the study. Detailed medical history, physical examination findings, and laboratory or radiology findings were recorded for each patient. Ten cc venous blood samples were drawn, centrifuged, and serum samples were stored at -20°C until studying. Serum samples were dissolved and brought to room temperature before analysis. The STA test dilutions (Seromed, Istanbul) were made up 1/40 from 1/640 to titers, and ≥160 titers were considered as positive. Brucella-IgM / IgG (VIRCELL, Santa Fe, GRANADA, SPAIN) antibody levels were measured by ELISA. All tests were carried out in accordance with the manufacturer’s recommendations.
Brucella Coombs Gel test
This test takes place in microplate wells containing gel matrix and Coombs antibody. ODAK Brucella Coombs antigen (ODAK Brucella Coombs Gel test, Toprak Medical, Istanbul, Turkey) was added to the dilution wells on the plate, and this mixture was pipetted into the gel matrix microtubes. We applied dilution ranges of 1:40-1:5120 and considered ≥160 titers as positive. Visual assessment was performed after centrifugation of the gel matrix for 20 minutes. The samples were accepted as negative if pink-coloured brucella antibody subsides at the bottom of microtubes or as positive if pink-coloured antibody floats over the gel. The test procedure took approximately two hours, including incubations and preparation. Both screening and dilution could be performed with this test (6).
Study patients were categorized as acute (<8 weeks), subacute (8-52 weeks), and chronic (> 1 year) brucellosis according to the duration of their symptoms. Patients who had relapsing symptoms or signs of infection within 12 months after the completion of brucellosis treatment and increased levels of IgG class antibody or blood culture positivity were included in the relapse group. Patients completing treatment within the last six months were included in the previously treated group.
Statistical analysis
Statistical analyses were performed by the MedCalc Statistical Software version 12.7.7. Cohen Kappa analysis was used to examine the categorical variables. Kappa coefficient of 0 ≤ κ < 0.20 was accepted as no compliance, 0.20 ≤ κ < 0.40 was accepted as weak compliance, 0.40 ≤ κ < 0.60 was accepted as moderate compliance,0.60 ≤ κ < 0.80 was accepted as high compliance, and 0.80 ≤ κ < 1.00 was accepted as perfect compliance. The statistical significance was set as <0.05. Sensitivity and specificity calculations were performed using the following formulas:
Sensitivity = True positive cases/All positive cases = True positive cases/ (True positive cases + False negative cases) x100,
Specificity = True negative cases / All negative cases = True negative cases / (True negative cases + False positive cases) x100,
Positive predictive value (PPV) = True positive cases / (True positive cases + False positive cases),
Negative predictive value (NPV) = True negative cases / (True negative cases + False negative cases)
Results
A total of 42 patients were enrolled in the study. The mean age was 47.2 ± 15.9, and 50% of them were male. Twenty-seven of these patients were diagnosed as brucellosis according to the medical history, physical examination findings and serological tests while the diagnosis of brucellosis was excluded in 15 patients.
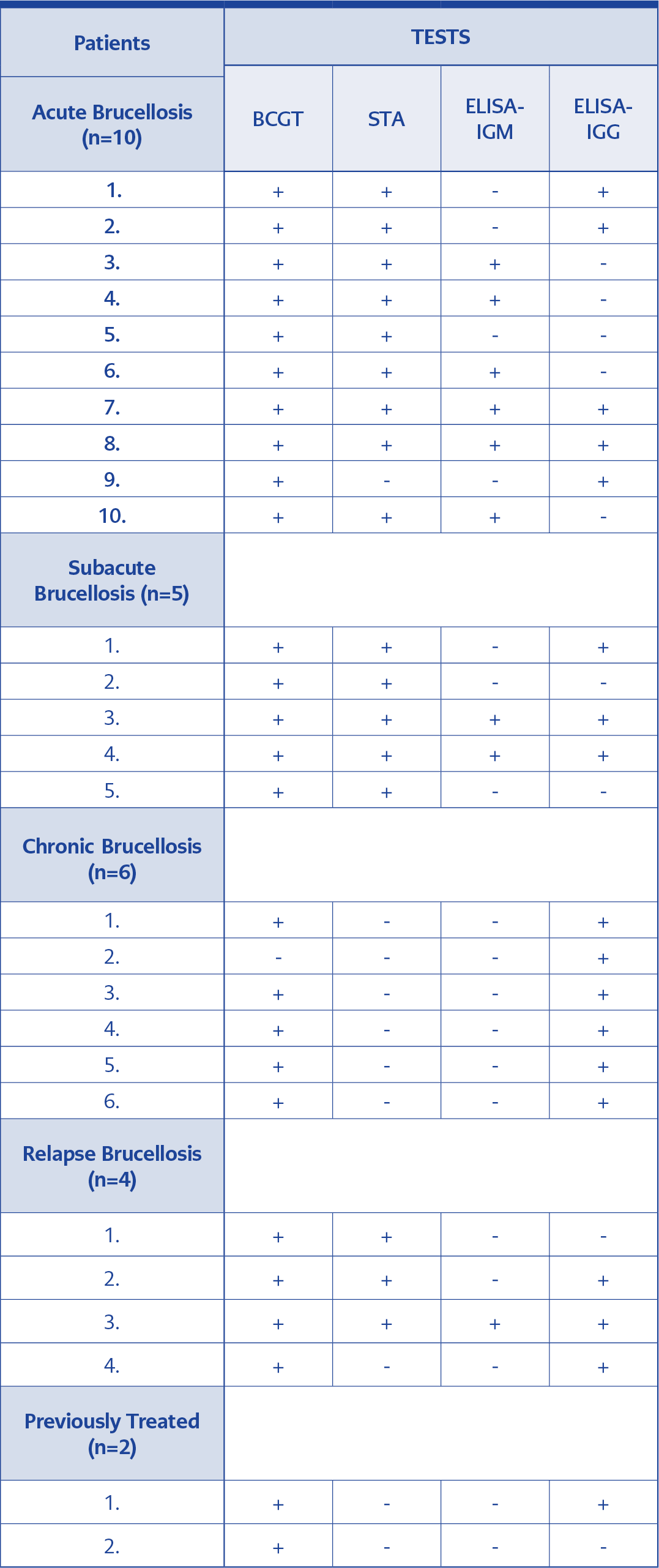
Thirteen (48.1%) patients were female, and fourteen (51.8%) were male out of 27 patients diagnosed as brucellosis. The mean age was 49.5 ± 17.6. When categorized according to the symptom duration, ten (37%) patients were accepted as acute, five (18.5%) as subacute, six (22.2%) as chronic, and four (14.8%) as relapsed brucellosis. Two (7.4%) patients were accepted as previously treated. BCGT was positive in twenty-six (96.2%) of the subjects, whereas STA was positive in 17 (62.9%) subjects, and Brucella-IgM and had Brucella-IgG were positive in 9 (33.3%), and 18 (66.6%) subjects, respectively. One patient’s BCGT, STA, and Brucella-IgM results were negative, while the Brucella-IgG result was positive. Patients’ serological test results are presented in Table 1, and the number and rate (%) of positive and negative serological test results are presented in Table 2.
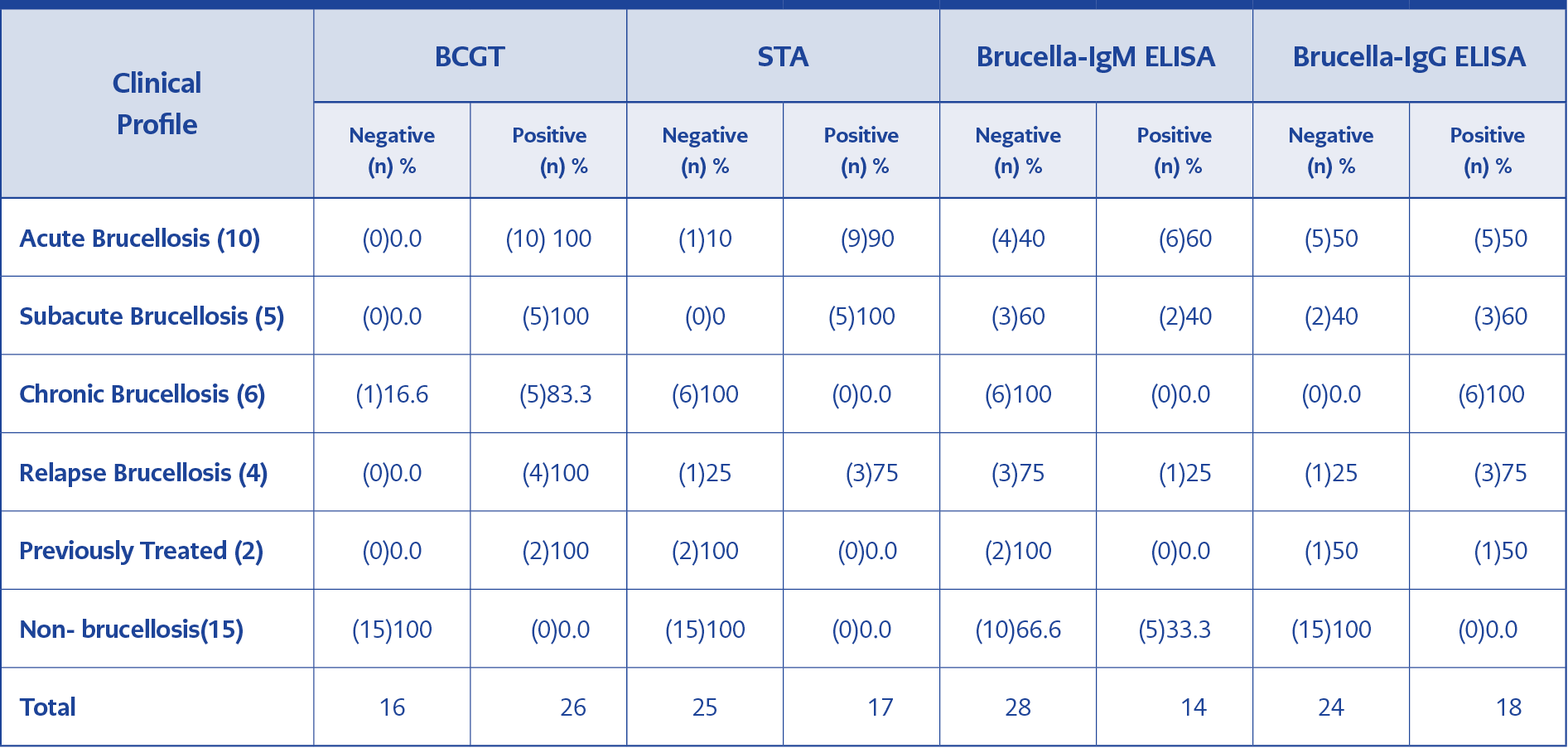
Table 2.The number and rate (%) of serological tests results according to the clinical profiles of the patients.
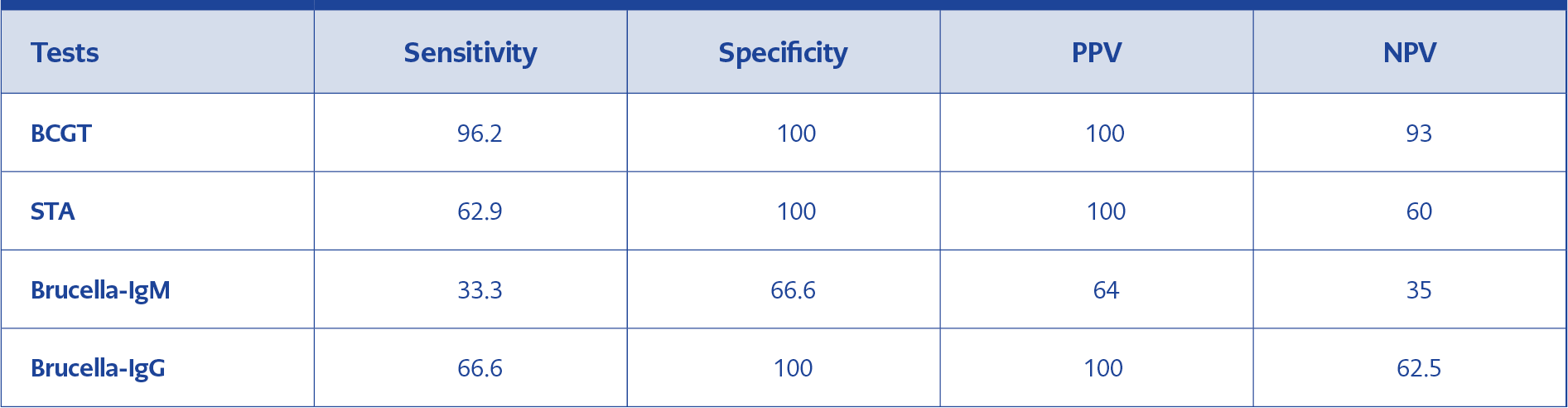
Among the tests performed in the study, BCGT demonstrated the highest sensitivity (96.2%) and specificity (100 %) in addition to the highest PPV (100%), and NPV (93%). The Sensitivity, Specificity, PPV, and NPV of all tests used in this study are presented in Table 3.
Kappa analysis showed that BCGT was moderately but significantly correlated with STA and Brucella-IgG (Kappa: 0.590, and Kappa: 0.539, respectively). However, there were no significant correlations between BCGT and Brucella-IgM (Kappa: 0.029).
When STA was accepted as a reference test, and its positivity titers were taken as ≥160, the sensitivity and the specificity of BCGT was found to be 100% and 64%, respectively.
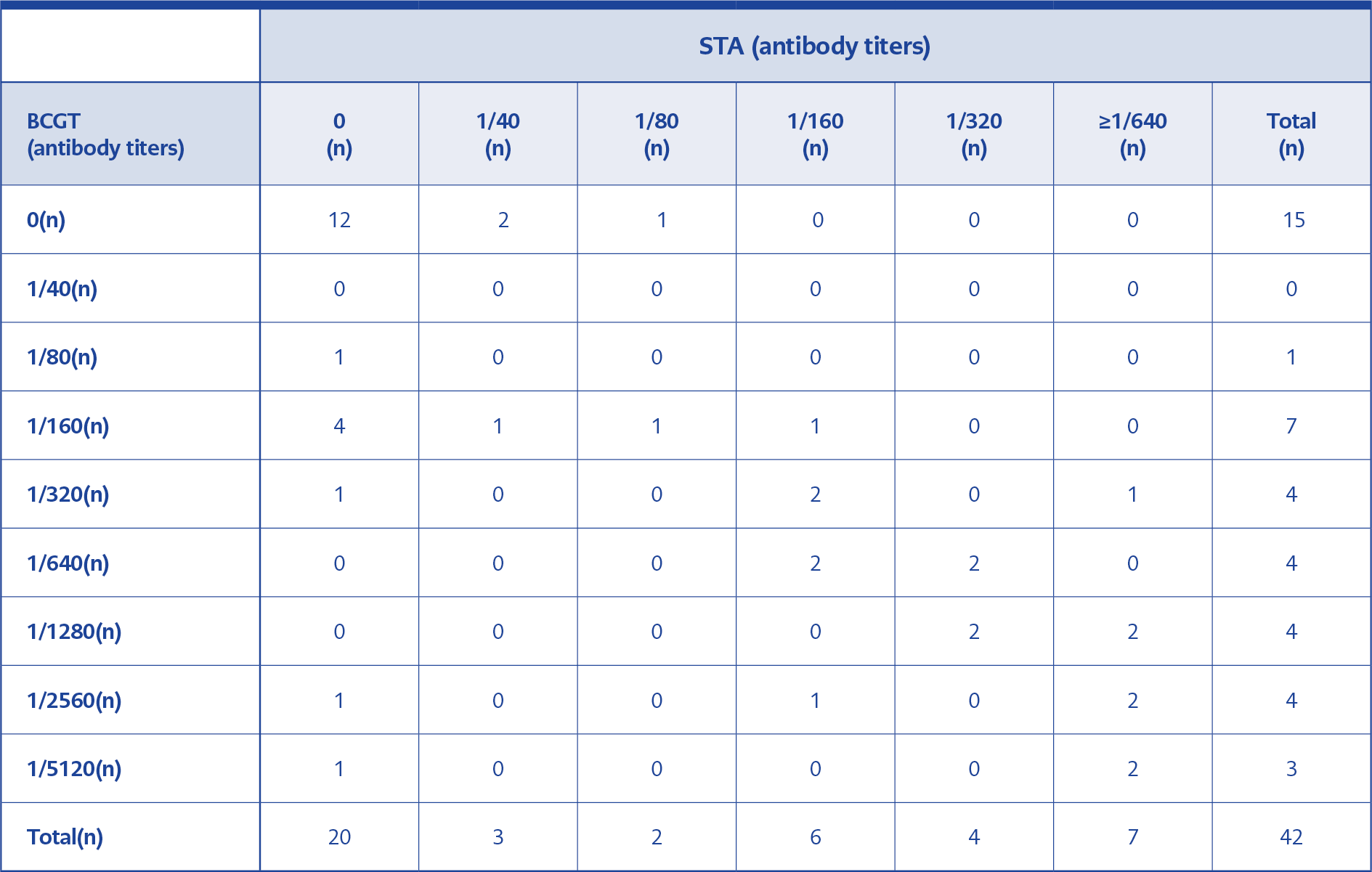
When we compared STA and BCGT titers, we found that the compliance rate was high in both at the levels of 1/160 and above. In two patients with BCGT positivity at 1/2560 and 1/5120 titers, STA was found negative. Brucellosis treatment of one patient was completed three weeks ago, and the other one was diagnosed as chronic brucellosis. A comparison of BCGT and STA results according to antibody titers is shown in Table 4.
Discussion
Serological methods, most commonly STA, are frequently used in the diagnosis of brucellosis (4). Total brucella antibodies (IgG, IgM, and IgA) are determined by STA (7). However, false-negative results may be observed with STA due to several causes, including the prozone phenomenon, sampling at the early stages of the disease, and the presence of blocking antibodies. On the other hand, some of the gram-negative bacterial infections may cause false-positive results (8). If false negativity is considered, C-STA is used as an extension of STA to determine further the presence of blocker or non-agglutinated immunoglobulins (7). Recently, BCGT, a much more practical method, has been developed. This test, which is performed in wells containing gel matrix and Coombs antibody, allows STA and C-STA testing at the same time. While 24-48 hr incubation is required in STA and C-STA, BCGT results can be obtained in less than 2 hours without incubation (6, 9). However, evidence regarding the implementation of BCGT in the diagnosis of the brucellosis is limited due to the novelty of this test. In a recent study by Türk Dagi et al., the sensitivity and specificity of BCGT in the diagnosis of the brucellosis was 94.6%, and 100%, respectively, which was significantly higher than that of the C-STA (10). In another study conducted by İrvem and colleagues, BCGT was found to have excellent compatibility with ICA and C-STA test (11). Among these tests, the ICA test, which is practical and fast, is applied with similar principles to BCGT. The most important advantage of BCGT over the ICA is that the 24-hour incubation time of ICA is less than 2 hours in BCGT, and results are taken in a shorter time with BCGT. In a study, of which the ICA was taken as a reference test, the sensitivity of STA, ICA, and BCGT were reported as 71.4%, 100%, and 100%, respectively, and the specificity was reported 100% for all three tests (12). In a study by Turhanoglu et al., the sensitivity and specificity of STA were 90.3%, 100% respectively, whereas the sensitivity and specificity of BCGT were 97.5%, 100%, respectively (13). In another study by Hancı et al., the sensitivity and specificity of BCGT were 100% and 82.2%, and the sensitivity and specificity STA were 53.4%, and 100%, respectively (14). In our study, we found that the sensitivity of BCGT as 96.2% and specificity as 100%, which is highly consistent with the results of the previous studies. We also found that the sensitivity of STA as 62.9% and its specificity as 100%. Our findings demonstrate a moderate correlation between STA and BCGT. The correlation shown between STA and BCGT is also consistent with the results of the previous research (15).
ELISA is the second most frequently used serological method to detect Brucella-IgM and IgG antibodies (16). Although there is enough data comparing ELISA and STA in the diagnosis of brucellosis, there are currently very few studies comparing the efficacy of ELISA and BCGT in brucellosis. The present study is one of the first studies comparing the diagnostic role of ELISA and BCGT in patients with brucellosis. Ozdemir et al. indicated that the sensitivity of ELISA IgM and IgG’s were 73.7% and 72.2% in brucellosis (17). We determined the sensitivity and specificity of IgM as 33.3%, and 66.6%, and the sensitivity and specificity of IgG as 66.6%, 100%, respectively. Compared to ELISA, the sensitivity and specificity of the BCGT to diagnose the brucellosis were significantly higher in our study. Moreover, no concordance was observed between BCGT and Brucella-IgM results, although there was a moderate correlation between BCGT and Brucella-IgG results. Previous data indicate that the ELISA test shows similar results with STA in the acute period, but is more sensitive in chronic and healed cases (18). In our study, we observed only one patient with chronic brucellosis who was diagnosed with IgG positivity. In acute cases, IgM can be found increased alone or with IgA and IgG. In chronic cases, IgG can be found increased alone or with IgA. In acute cases of the present study, while the sensitivity of IgM was 80%, IgG’s sensitivity was 90-100 %. In subacute cases, the performance of IgM decreases while IgG provides better results than IgM; however, in some cases, both IgM and IgG may be negative (19). In our study, nine of 26 patients who were positive for BCGT were IgM positive, and seventeen of them were IgG positive.
BCGT, STA, and Brucella-IgM were negative in one patient, while Brucella-IgG was positive. This patient, who was finally diagnosed as chronic brucellosis, was living in the endemic region and had complaints of sweating, arthralgia, headache, and lumbar spondylodiscitis for more than one year. The main difference of our study than others is that the sensitivity and specificity calculations are based not only on serological tests but also on clinical features.
Relapses may occur in 5-40% of acute brucellosis cases, and cultures may also be negative in these cases. Clinical findings and serological tests are of great importance in these patients (19). In one study, positivity rates of C-STA with brucella IgG, IgA, STA were 83%, 58%, 25%, respectively; and brucella IgM was negative in such patients with brucellosis. ELISA IgG, IgA, and Coombs accumulating titers are good indicators of relapse, while IgM is of poor value (20). In our study, BCGT, STA, Brucella-IgM and IgG positivity rates were 100%, 75%, 25% and 75%, respectively, in patients with relapse brucellosis.
The criteria used for the diagnosis of brucellosis and clinical features of the study groups vary in previous studies. This variation can somewhat explain the differences in our results and previous evidence.
In microbiology laboratories, in addition to the STA test, at least one agglutination test or ELISA Brucella-IgM/IgG study is recommended to improve the reliability of the results (7). The most important limitation of our study was that the limited number of cases and culture positivity rates affecting the comparability.
In conclusion, the results of the present study show that BCGT may be useful in the diagnosis of brucellosis due to its high sensitivity and specificity. However, as a new test, BCGT has to be studied further with culture-confirmed cases and control groups, so that it can take part in routine diagnostic laboratories.