Introduction
Infective endocarditis is a clinical condition that affects the endocardial surface of the heart, involving natural or prosthetic heart valves and intracardiac devices. It is a high-cost and high-mortality infectious disease (1) with an increasing incidence and is more frequently diagnosed with advancing diagnostic methods. Along with well-defined cardiac (previous endocarditis, presence of intracardiac prosthetic materials, structural and congenital heart valve diseases) and non-cardiac (intravenous [IV] drug usage) predisposing risk factors for infective endocarditis, various conditions such as advanced age, chronic kidney failure, malignancy, chronic diseases, hemodialysis, and receiving healthcare have been reported as factors increasing the risk of infective endocarditis recently. Furthermore, the increased life expectancy with new treatments for chronic diseases and malignancies, as well as the application of advanced technology and novel techniques in healthcare and surgical procedures, have contributed to the increased incidence of endocarditis (2, 3, 4). Infective endocarditis is primarily caused by bacteria and fungi. The most commonly identified pathogens are staphylococci, viridans streptococci, and enterococci (5).
The diagnosis of infective endocarditis is based on the modified Duke criteria, and bactericidal and parenterally administered antimicrobial agents should be chosen for treatment. Combination therapy is preferred at the beginning of treatment (3). Despite all the advancements in diagnosis and treatment, the mortality of infective endocarditis continues to remain high (1).
This prospective study presented the epidemiological, clinical, laboratory, and prognostic characteristics of patients diagnosed with infective endocarditis at a university hospital, aiming to contribute to Türkiye’s data on infective endocarditis epidemiology and risk factors.
Materials and Methods
The study was conducted at Pamukkale University School of Medicine Hospital, a tertiary care facility with a capacity of 857 beds. Pamukkale University School of Medicine Clinical Research Ethics Committee approved the study on September 25, 2018, with decision number 18. Patients aged 18 and older with a definite or possible diagnosis of infective endocarditis were included in the study. The modified Duke criteria (definite/probable) were used for the diagnosis of endocarditis (3). Patient data were recorded prospectively.
As factors predisposing to endocarditis, the registration form included congenital and degenerative heart diseases, as well as rheumatic heart disease-associated valve damage, prosthetic valves, cardiac pacemakers, cardiac conditions, and non-cardiac chronic diseases such as diabetes, hypertension, chronic obstructive pulmonary disease, prior cerebrovascular disease, cancer, chronic kidney failure, and HIV infection. In cases of prosthetic valve endocarditis, those occurring within the first year after surgery were defined as early prosthetic valve endocarditis, while those occurring after one year were considered late prosthetic valve endocarditis.
Cardiac and non-cardiac factors that could predispose to endocarditis, such as the presence of intracardiac prosthetic devices, congenital and structural heart valve diseases, IV drug usage, other comorbidities, and probable causes of bacteremia, including previous hospitalization, endoscopy, colonoscopy, bronchoscopy, dental extractions within the past six months, and a history of recent diarrhea within one month before diagnosis, were recorded. Infective endocarditis that developed in patients who received healthcare and invasive procedures in the last six months or after the patient’s admission was defined as healthcare-associated endocarditis (6).
C-reactive protein (CRP) elevation was considered at 5 mg/L, and procalcitonin elevation was considered at 0.5 ng/mL and above. Blood cultures were incubated using the BD BACTEC™ FX blood culture system (BD Diagnostics, USA). Gram staining and culturing on Columbia sheep blood agar and eosin methylene blue media were performed on blood culture bottles showing growth signals. Species identification and antibiotic susceptibility results of microorganisms were evaluated using the BD Phoenix™ 100 system (BD Diagnostics, USA).
Results
In our study, 67 cases of endocarditis were identified in 65 patients between 2018 and 2021. Of these patients, 40 were male, and 25 were female. The mean age of the patients was 52.5±14. Twenty-two (33%) of the patients had underlying cardiac diseases that predisposed them to infective endocarditis. Among them, 7 (32%) had congenital heart disease, 8 (37%) had degenerative heart diseases, and 6 (27%) had heart valve diseases due to acute rheumatic fever. In 1 (4%) patient, both congenital heart disease and rheumatic heart valve disease were observed together. Hypertension (30.8%) and chronic kidney failure (20%) were the most common non-cardiac comorbidities. When possible, sources of bacteremia that resulted in endocarditis were examined, 53.7% of the cases had a history of hospitalization within the last six months, and 41.8% had a history of prior cardiac surgery. Central venous catheter usage was present in 15 (22.4%) cases. The rate of healthcare-associated endocarditis was 58.2%. There were five cases with IV drug use.
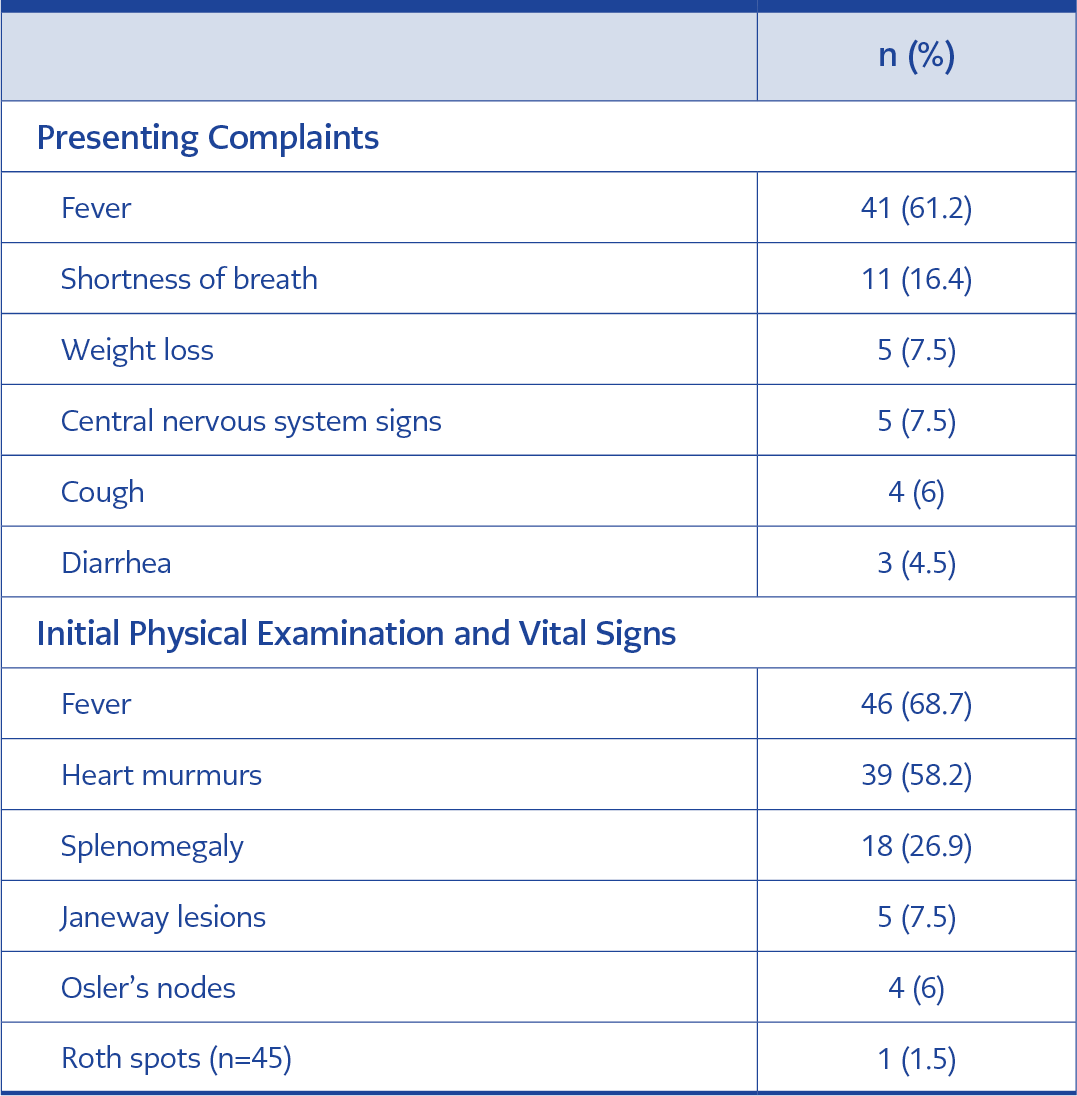
Table 1. Evaluation of the initial presenting complaint, physical examination, and vital signs in infective endocarditis (n=67).
Table 1 provides the patient’s presenting complaints and physical examination findings. The most common presenting complaint and physical examination finding was fever, which lasted an average of 21 days.
According to laboratory findings, the average CRP was found to be 120.8±90.7 mg/L, and it was high in 64 (95.5%) cases. Procalcitonin was examined in 42 cases, and it was high in 23 (54.8%) cases. Rheumatoid factor was tested in 35 cases and was positive in 26 (74.2%) cases.
When imaging findings were evaluated, transthoracic echocardiography revealed endocarditis findings in 49 (73.1%) cases. Vegetations were present in 43 (64.1%) cases. In three cases, there were findings of calcification, aneurysm, and chordal rupture in addition to vegetation. Imaging findings other than vegetations included graft separation in two cases and valve dysfunction in four cases. Transesophageal echocardiography (TEE) was performed in 40 (59.8%) cases. In 31 (46.3%) cases, vegetation was present alone. In addition to vegetation, two cases out of five had ruptures, two had dehiscence of the prosthetic valve, and one had fistulas in the valves.
TEE was performed in 15 cases out of 18 with no findings on transthoracic echocardiography (TTE). In TEE, vegetation findings were detected in 14 cases. Although there were findings on TTE, TEE was also performed in 24 cases. In addition to TEE and TTE vegetation findings, rupture was detected in one attack, valve dehiscence was detected in one attack, and valve fistula was detected in two cases. In addition, cardiac magnetic resonance imaging (MRI) was performed in two cases, and endocarditis findings were detected in both. There was evidence of endocarditis in five of the eight cases in which leukocyte-labeled scintigraphy was performed.
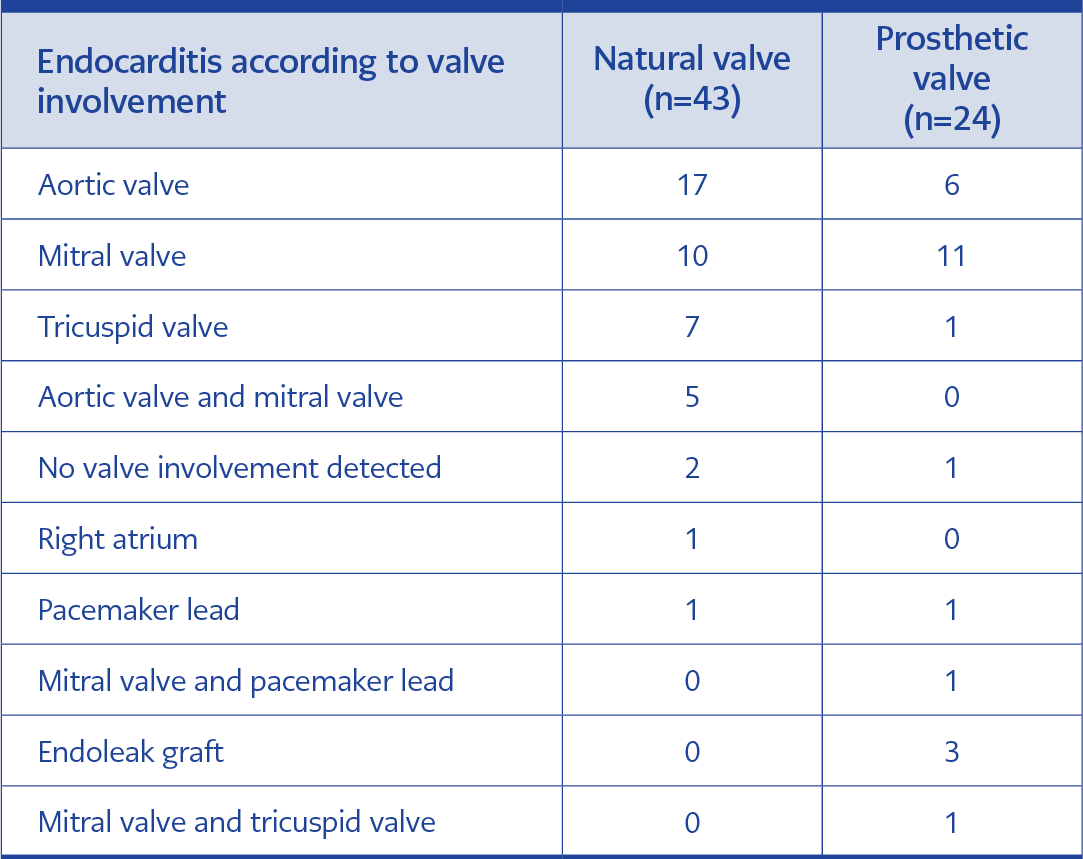
Forty-three (64.1%) cases were native valve endocarditis and 24 (35.9%) were prosthetic valve endocarditis. Pacemaker involvement was in 2 (3%) cases, and graft involvement was in 3 (4.5%) cases. Valve involvements are specified in Table 2.
In 41 (61.2%) cases, the causative agent of endocarditis was identified through blood culture. In two cases with negative blood cultures, growth was observed in surgical cultures. One was MSSA (methicillin-sensitive Staphylococcus aureus), and the other was Klebsiella pneumoniae endocarditis. In 21 (31.3%) cases, the causative agent was staphylococci. Of these, 14 were S. aureus, and seven were coagulase-negative staphylococci. The blood culture isolations are listed in Table 3.
Antibiotic treatment was initiated empirically in 46 (68.7%) cases, and in 21 (31.3%) cases, it was initiated specifically based on the causative agent. Ampicillin-sulbactam (IV ampicillin 2 g every 8 hours and IV ampicillin-sulbactam 2 g every 8 hours) plus gentamicin 3 mg/kg, vancomycin plus gentamicin, or daptomycin 9 mg/kg plus gentamicin were used in 23 (50%), 6 (13%), and 9 (19.5 %) cases, respectively. Vancomycin and daptomycin-containing regimens were preferred mainly for healthcare-associated endocarditis.
Surgical treatment was performed in 38 (56.7%) cases. Emergency surgery was performed in 3 (14.2%) of them, priority surgery was performed in 11 (52.4%), and elective surgery was performed in 7 (33.4%). Surgical complications occurred in 7 (18.4%) cases. Among these, 4 (57.2%) were cardiac tamponade, 2 (28.5%) were paravalvular leaks, and 1 (14.3%) was a wound site infection. Complications were identified in 44 (65.6%) cases. Among these, 33 (75%) had septic emboli, and 22 (50%) had cardiac complications. In 12 (27.3%) cases, multiple complications were identified.
Out of the 67 cases identified in 65 patients, 32 (49.2%) resulted in death within 180 days. Among them, 3 (9.3%) were due to non-endocarditis-related causes. The average time to death was 19 days.
Discussion
The average age of infective endocarditis varies depending on the development level of the countries. In two multicenter studies in TÜrkiye, covering the years 2005 and 2012 and 2000 and 2013, with 248 and 325 patients, respectively, the average age was 47 years (7-8). In our study, the average age was similarly found to be 52.5. The reason for infective endocarditis occurring at a younger age in developing countries can be explained by the higher prevalence of congenital heart diseases and rheumatic heart valve diseases, which predispose individuals to endocarditis.
Similarly, in our country, heart valve diseases due to acute rheumatic fever (ARF) are the first risk factor among cardiac risk factors (3). In this study, the rate of valve disease due to ARF was 31%. There are well-defined predisposing risk factors for infective endocarditis, including the presence of intracardiac prosthetic devices, history of previous infective endocarditis, structural or congenital heart valve diseases and IV drug use. However, recent studies have shown that the risk of infective endocarditis is also high in patients with some comorbidities such as advanced age, chronic kidney disease, malignancy, solid organ or bone marrow transplantation, or in patients having hemodialysis, or long-term healthcare with IV catheterization.
When the relationship between chronic diseases and endocarditis is examined, it is suggested that hypertension may be associated with an increased risk of endocarditis because of the chronic damage it can cause to the valve structure and heart endothelium (9). Chronic kidney failure can predispose individuals to endocarditis due to immunosuppression caused by uremia and degenerative heart disease with calcification of heart valves (10-11). Although the exact mechanism by which those comorbidities or interventions increase the risk of infective endocarditis is not fully understood, it is thought to be a result of the endothelial damage they cause or the frequency of bacteremia they increase.
Intravenous catheter usage is another factor that can lead to the development of endocarditis in patients with kidney failure, and it has been shown that there is a higher incidence of endocarditis in patients with catheter usage compared to those using fistulas (10), probably due to increased risk of bacteremia among patients undergoing dialysis via a central venous catheter. In our cases, the most common chronic diseases were hypertension and chronic kidney failure. Ten of the patients with chronic kidney failure (80%) were undergoing hemodialysis, and nine of them had central venous hemodialysis catheters. Additionally, in individuals using IV drugs, the rate of endocarditis has been found to be between 2% and 20%, and the risk of endocarditis is 20 times higher in IV drug users compared to the general population (12). In the United States, the rate of endocarditis among IV drug users increased from 15.3% to 29.1% between 2010 and 2015. This increase has been associated with the rising frequency of endocarditis among young individuals (13). Due to the direct endothelial damage and thrombosis caused by drug use and the higher bacterial load introduced into the systemic circulation through contaminated injectors, right heart involvement is more common in these patients (12). In our study, the average age of the five patients with IV drug use was found to be 26.2, and the rate of tricuspid valve involvement was 60%.
When examining the relationship between endocarditis and healthcare, the frequency has been reported to be between 10% and 34%, with the majority of cases (84%) being catheter-related (6). In this series, the rate of healthcare-associated endocarditis was found to be 58.2%. Among those associated with healthcare, 76.9% had a history of hospitalization, 46.2% had a history of surgery in the last six months, and 35.9% had central venous catheter usage.
It is noted that fever is observed in approximately 90% of endocarditis patients and is the most common presenting symptom (14). In our study, the number of patients with fever as a complaint was 41 (61.2%), and the number and rate of patients with fever at admission were 46 (68.7%). The average duration of fever complaints was 21 days.
A study examining endocarditis laboratory findings, showed that S. aureus was more frequent in endocarditis cases with elevated CRP, procalcitonin levels, and leukocytosis (15). Another study found that procalcitonin levels were higher in those with a preliminary diagnosis of endocarditis than in those in whom endocarditis was excluded. In addition, increased procalcitonin levels were found to be higher in patients with fever of 38°C and above, patients with positive blood cultures, patients with symptoms lasting less than five days, and patients on immunosuppressives (16). In our study, 70% of the cases in which elevated procalcitonin was detected had bacteremia.
The preferred initial imaging method for patients suspected of endocarditis is transthoracic echocardiography, which is as sensitive as TEE for detecting natural valve endocarditis (17). One study noted that TTE has suboptimal vegetation detection sensitivity but a high negative predictive value (18). In this study, the rate of detecting endocarditis findings with TTE was 78.6% in natural valves, 100% in early prosthetic valves, 60% in late prosthetic valves, and 50% in graft and pacemaker infections. Technically, TTE may be inadequate in approximately 20% of obese patients, those with chest wall abnormalities, and those with chronic lung diseases (19-20). In our cases, 93.3% of patients who did not have endocarditis findings on TTE were found to have findings on TEE. Therefore, although TTE is the preferred imaging method for endocarditis, TEE can be useful for diagnosing cases of suspected endocarditis.
A multicenter study involving 25 developed and developing countries, found culture positivity in 90% of cases (21). In a study conducted in TÜrkiye, the positivity rate for blood cultures was 65% (7). In this study, similarly, causative agents were identified in 61.2% of cases (43 cases), while no causative agent was found in 38.8% (26 cases). The most commonly identified agent was S. aureus, which is consistent with the literature. Due to its preventive effect on complications such as sepsis and endocarditis-related septic embolism in infective endocarditis, it is recommended to start antibiotic treatment immediately after blood cultures are taken (22). The national consensus guideline recommends the combination of ampicillin-sulbactam 12 g and gentamicin 3 mg/kg for community-acquired endocarditis (3). However, the maximum dose for sulbactam is stated as 4 g. In this study, we administered empirical treatment to 50% of the patients in four equal doses, with a total dose of 12 g of ampicillin and 3 g of sulbactam. 69.5% of patients receiving this combination therapy had community-acquired endocarditis. Treatment was changed according to the microorganism growing in 55% of these patients.
Surgery is another component of treatment for endocarditis. In this study, the surgical procedure was performed in 56.7% of the cases, and 29.8% of were operated on within the first seven days. Studies examining the effect of surgical treatment on mortality found that it reduced mortality when comparing those who received only medical treatment and those who received medical treatment together with surgery (21). In this study, the most common reasons for surgery were found to be heart failure (32.8%) and septic embolism (14.9%); surgical complications were 18.4%. The mortality rate after surgery was found to be 36.8%.
Septic embolism was our study’s most frequently detected complication, with 50.7%. Embolism was most frequently detected in the cerebral (26.3%), spleen (6.8%) and lungs (6.1%) (23). Central nervous system embolism rates were similarly determined 26.8% in this study.
We found a higher mortality rate than the national consensus report (3). The reasons for the high mortality may be the high rate of central venous catheter use in our study, the high rate of healthcare-associated infections, and the fact that the most common cause of bacteremia is staphylococci. It can also be considered that the study was conducted during the COVID-19 pandemic period.
In conclusion, after examining cases in the first six months, our findings regarding the epidemiology and prognosis of infective endocarditis pointed out that it is still a disease with a high mortality rate. To reduce mortality, preventing healthcare-associated infections and managing medical and surgical treatment for endocarditis in a multidisciplinary manner are believed to be beneficial.