Introduction
West Nile Virus (WNV) is an enveloped RNA virus with icosahedral symmetry and with a size of 45-50 nm belonging to the family Flaviviridae, genus Flavivirus (1). The virus, which was first isolated from a woman living in the West Nile region of Uganda in 1937, was noted with consecutive outbreaks in North Africa, Southern Europe, Israel and Russia in the 1990s and its spread has been accelerated with its passage to North America at the beginning of the 2000s. WNV outbreaks have been seen frequently especially in the Middle East and Europe in recent years (2, 3).
Transmission usually occurs with the mediation of the mosquitoes of Culex genus that feed on infected birds. Thus, birds are the natural sources and reservoir hosts of WNV. Also, it has been reported that transmission via blood and organ transplantation, transplacental route and breast milk was possible (4, 5).
In Turkey, The highest number of WNV cases were detected in 2010. In the study conducted by Kalaycioglu et al. between 2010 and 2011, 47 cases were diagnosed with WNV, and it was reported that the virus could be endemic in the western regions of Turkey (6).
Turkey is vulnerable to WNV cases and outbreaks due to the presence of infectious vectors and its geographical location on the flight paths of migratory birds.
This community-based study aimed to determine the seroprevalence of WNV in serum samples collected from the whole Manisa province in 2014 and the relation of this seroprevalence with sociodemographic and socioeconomic variables.
Materials and Methods
Study Population
The sera and data were collected between March 18, 2014, and June 22, 2014, for “Determination of the seroprevalence of some vaccine-preventable diseases in Manisa, 2014” Research Project (BAP) of Dokuz Eylül University.
This is a cross-sectional seroprevalence study and its population consists of all individuals over the age of two who are registered in the Manisa Province Family Medicine Information System (FMIS) (N=1,317,917). With OpenEpi software, version 3.01 (www.openepi.com), the smallest sample size was determined to be 1337 people with 2% expected prevalence, 0.75% standard deviation and 95% confidence interval. By considering the problems that may be encountered during data collection or problems such as the possibility of being unable to reach the people in the sample, insufficient serum, hemolysis, and lipolysis, 30% backup was added to this number and the sample size was calculated to be 1740. Of the targeted 1740 people, 1260 (72.4%) people accepted to be included in the study and 1255 people gave a blood sample for serum.
The study subjects were selected by using a simple random sampling method with the help of a database (Oracle, Austin, Texas, U.S.). The similarity of the demographic characteristics of the selected people such as age, sex and place of residence with those of the population was evaluated. The people who were chosen as a result of sample selection were invited to the Family Medicine Centers (FMC) in which they were registered by appointment, between March 2014 and June 22, 2014, by the FMC staff. Data collection forms were filled by pollsters by face-to-face interview method. During the interview, 10 ml venous blood sample was obtained by a nurse or midwife. The collected blood samples were sent to Manisa Public Health Laboratory on the same day and were frozen at -20 °C after being centrifuged and aliquoted. The serums were weekly transferred to Dokuz Eylül University Medical Microbiology Laboratory by cold chain and stored at -80 °C.
Questionnaire and Data Collection
The ages of the participants were classified within groups of 10-year intervals, their locations as “periphery” or “central district” in relation to the FMC region, and their educational statuses as “illiterate”, “literate”, “elementary school”, “middle school”, “high school”, “university and above”. Presence and type (hypertension [HT], diabetes mellitus [DM], cardiovascular disease [CVD]) of chronic disease if present were questioned. The location of the toilet (whether it was in or out of the house) was questioned for vector exposure. The monthly income of the participants was asked with an open-ended question and the yearly equivalized income per person pertaining to the year 2014 was calculated to be 3265 TL according to the equivalized income scale developed by Organization for Economic
Co-operation and Development (OECD). People with incomes lower than this income were considered to be low-income and those who had higher incomes than this were considered to be high income.
The mean altitudes of the districts and their areas in terms of square kilometers were obtained from the official website of Republic of Turkey Ministry of National Defence, General Directorate of Mapping. The population data of cities and districts pertaining to 2013 were obtained from the database of TURKSTAT website (Turkish Statistical Institute) (7-8). The provincial median altitude value was calculated to be 132 meters (m), the districts were separated into two as being below or above this value. Population densities were calculated separately by dividing the district populations by their areas and the provincial median was found to be 80 people/km2 and the districts were separated into two groups.
Laboratory Analysis of Samples
In the study, the specific antibody responses to WNV in the serum samples of the participants were examined by a commercial ELISA (Enzyme-linked immunosorbent assay) method according to the manufacturer’s instructions (Euroimmun, Lubeck, Germany). The antigen source used in the test is a recombinant, detergent-extracted glycoprotein E of WNV from the membrane fraction of human cells. Quantitative measurements were done for IgG-type antibodies using optical density and standard calibrators. All indeterminate results were considered to be negative.
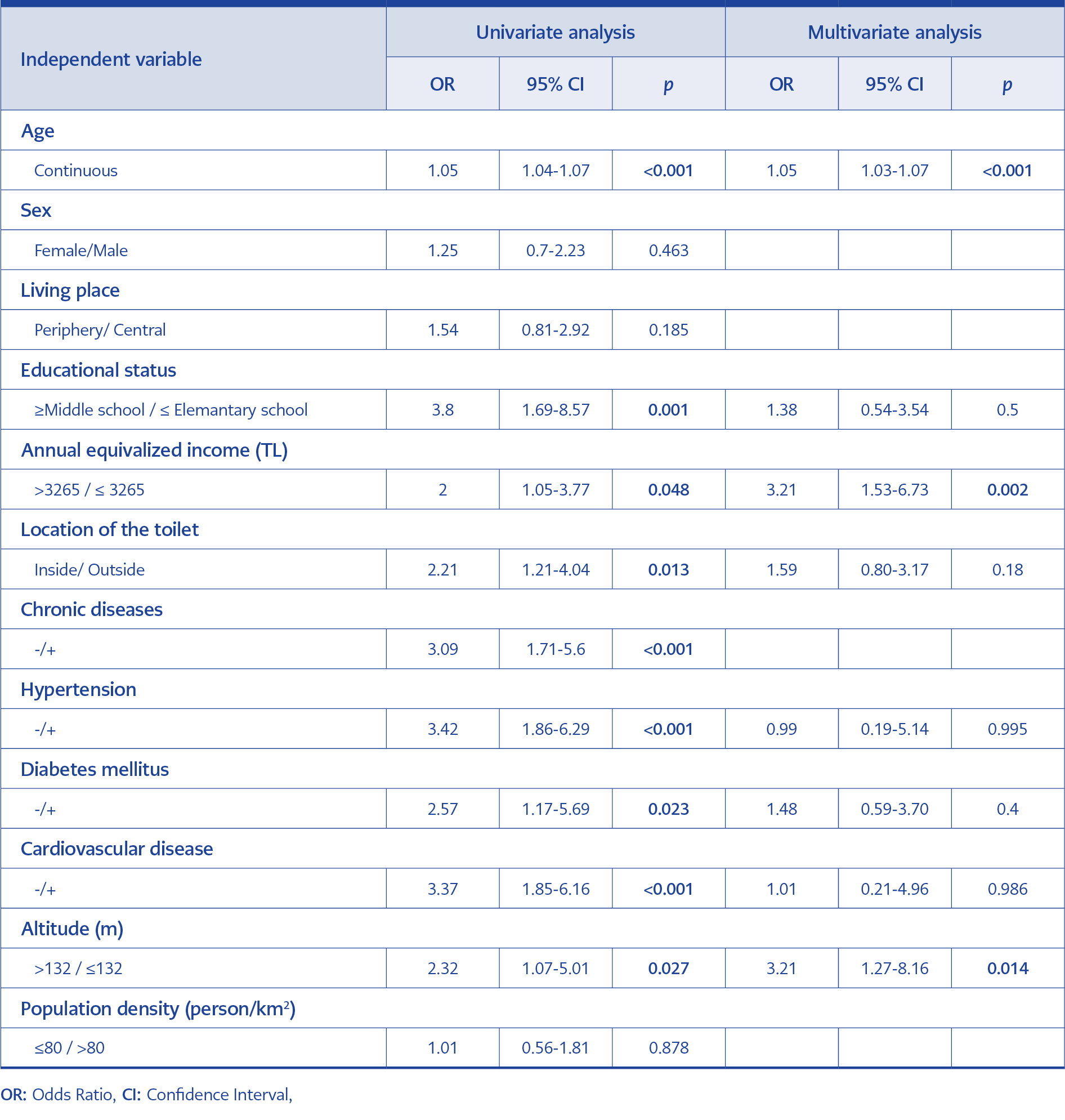
The statistical analysis was performed by using the Statistical Package for Social Sciences (SPSS) 22.0 (IBM Corp., Armonk, NY, USA). Chi-square test was used to detect the association of antibody positivity with gender, age group, presence of chronic disease, hypertension, diabetes, cardiovascular diseases, educational status, altitude, population density, and location of the toilet in the house. By including the significant variables logistic regression was performed for the prediction of seropositivity. Analyses were conducted within 95% confidence interval (CI) and p<0.05 values were set as significance. Dokuz Eylül University Non-Invasive Research Ethics Committee approved the study on August 13, 2018, with the decision number of ‘‘2018/17-07’’.
Results
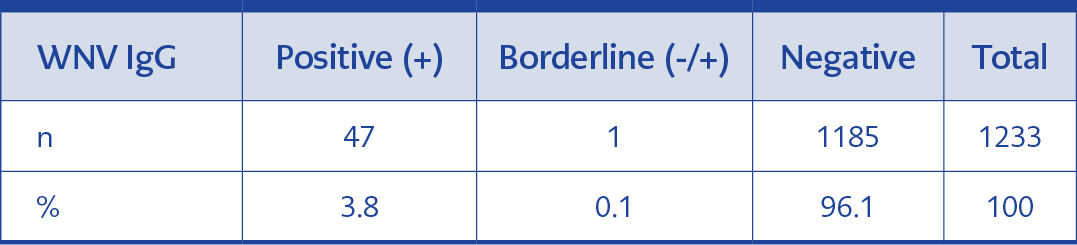
Of the 1255 people who consented to be included, 22 were excluded because of an insufficient sample, so the results were analyzed for 1233 people.
In the study group, 591 (47.9%) were males, 642 (52.1) were females and their ages varied between 2 and 82 (mean age 36.72 ± 21.09).
In the study, WNV IgG was positive in 3.8% (47/1233) and indeterminate in 0.1% (1/1233) (Table 1). Because the sera detected to be positive as a result of antibodies were not confirmed with the plaque reduction neutralization test (PRNT) method, the results were interpreted as probable antibody positivity and analysis findings were calculated only over positive sera.
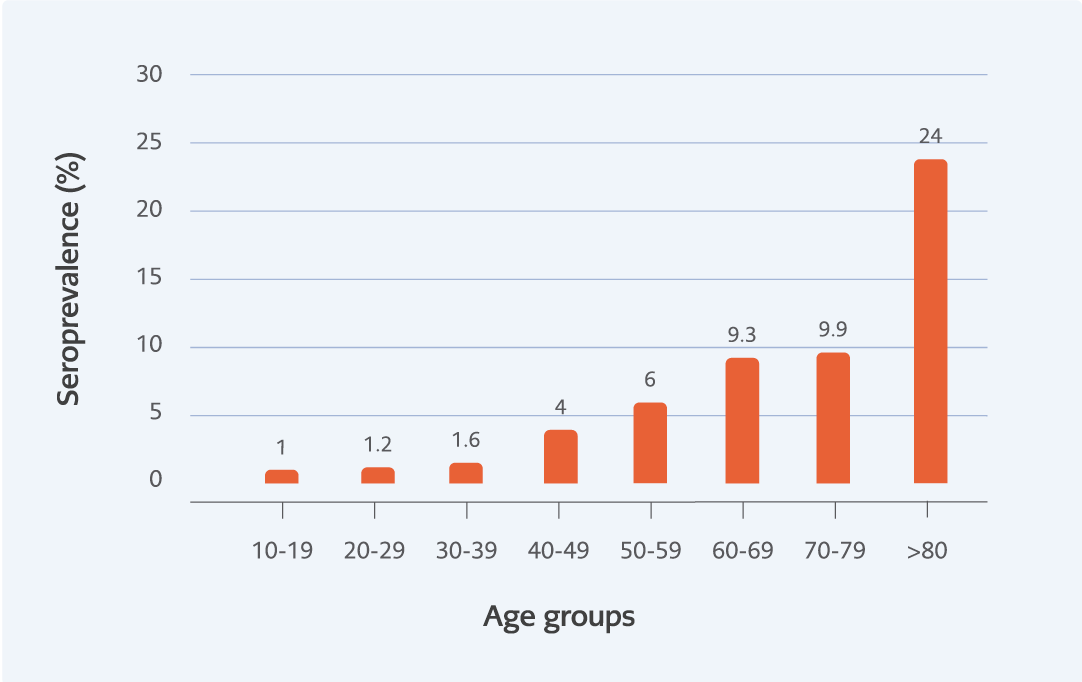
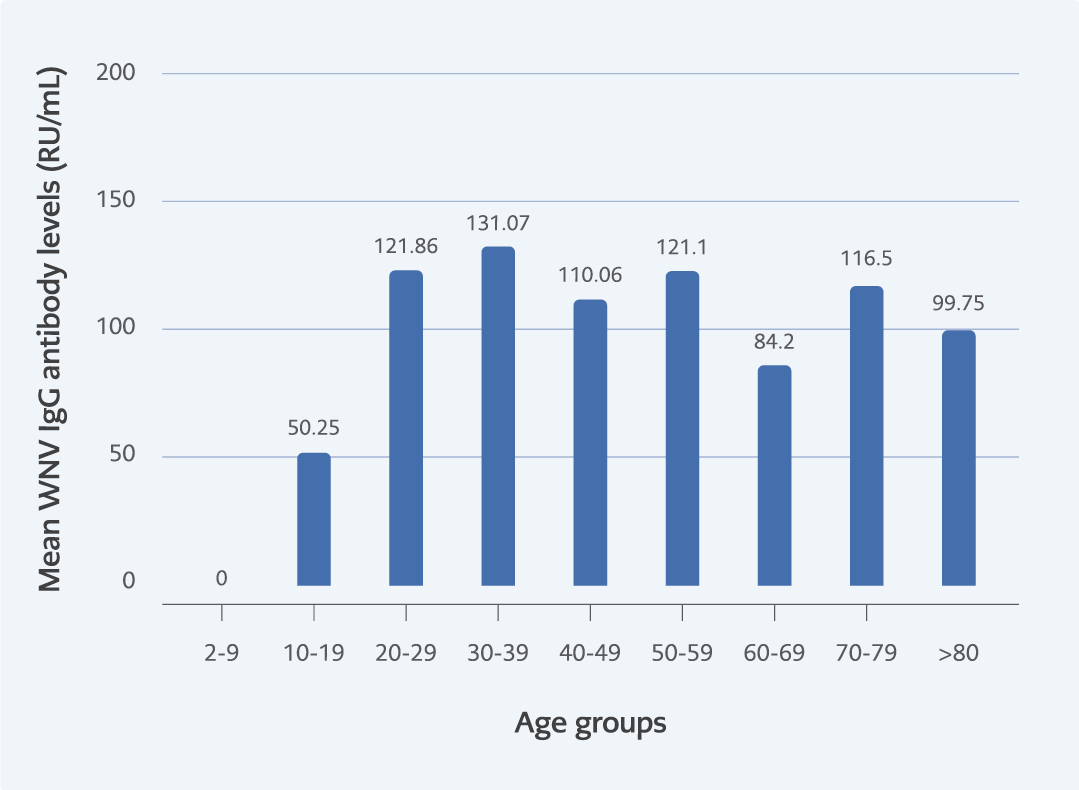
In this study, no association was found between gender and seroprevalence (p=0.46), but a significant correlation was found between seroprevalence and age groups (p<0.001). This difference was detected among individuals older than 40-49 age group (Figure 1). WNV IgG antibody level (RU/mL) was detected to be similar in all age groups (p=0.6) (Figure 2).
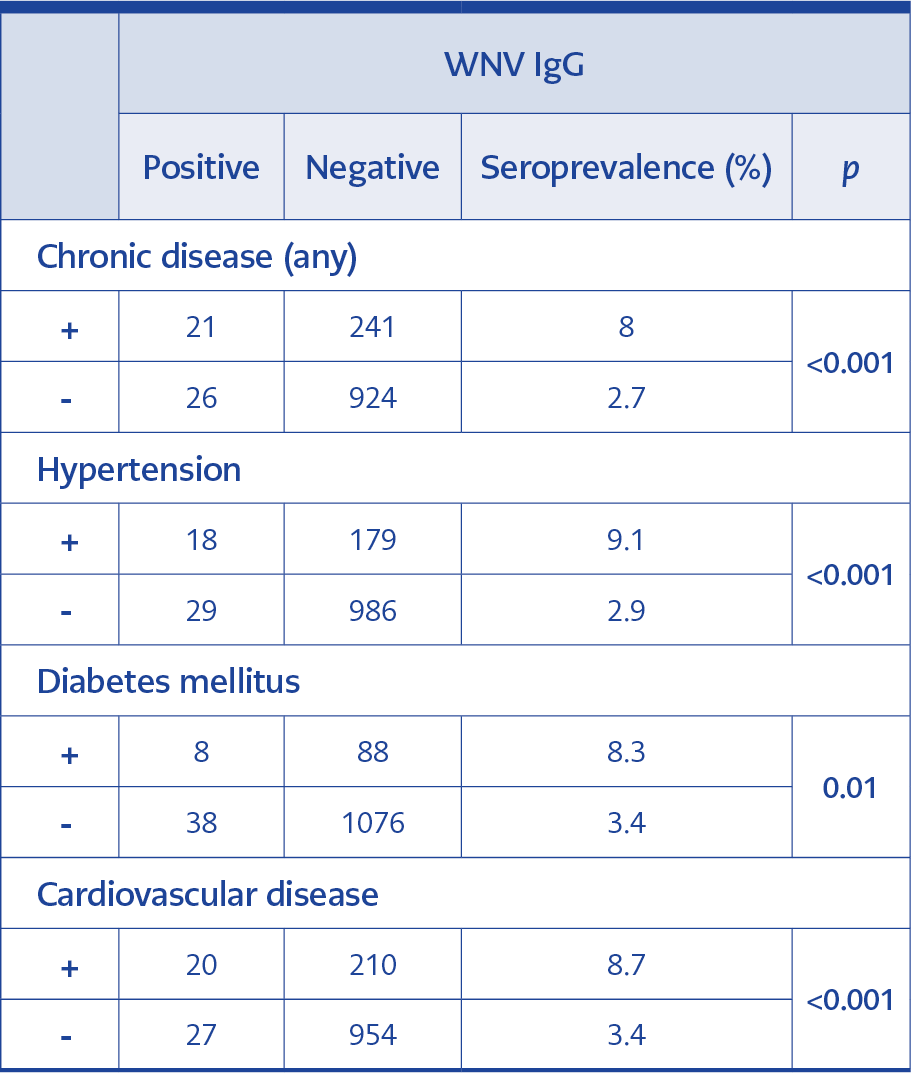
WNV IgG seroprevalence was found to be significantly higher among the people with HT, DM and CVD in comparison to those who did not have these diseases (Table 2).
WNV IgG seroprevalence was significantly found to be higher in adults who were illiterate, merely literate or elementary school graduates. This difference was observed to become more prominent as the level of education decreased (p<000.1). There was no difference in seroprevalence between central and peripheral FMC regions. When the examination was done according to annual adjusted income per person; WNV IgG seroprevalence was found to be significantly higher for those with low income (p=0.03) (Table 3).
The WNV IgG seroprevalence was found to be significantly higher among the participants who lived in the houses where the toilets were located outside of the house or toilets both inside and outside the house than those with toilets inside the house (p=0.01).
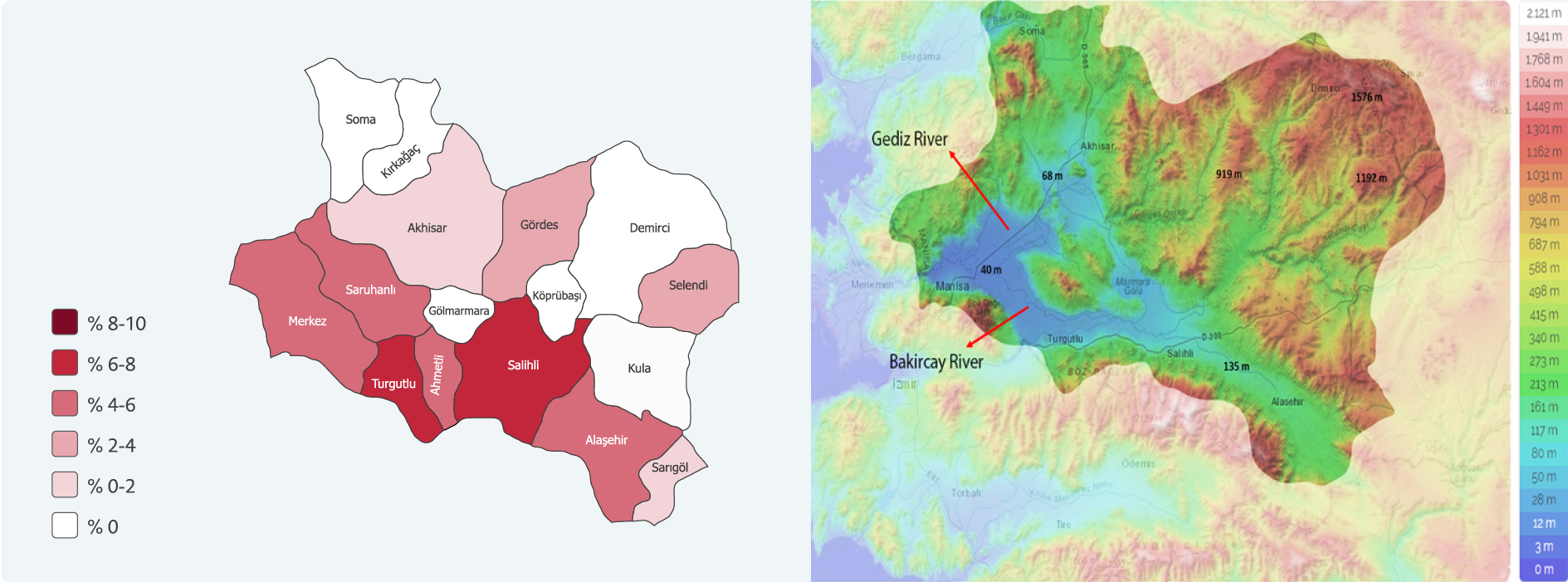
The people in the study group resided in the 17 districts within the Manisa province borders. The WNV IgG seroprevalence was detected to be higher in some districts. These districts were marked on geographical maps and it was seen that they were located in Gediz and Bakırçay river beds with relatively lower altitudes (Figure 3).
The WNV IgG seroprevalence in the districts with a median altitude value below 132 m was found to be significantly higher than those with a median altitude of more than 132 m (p=0.02). No difference was detected in seroprevalence between districts with a population density higher than 80 people/km2 and those with a population density less than 80 people/km2 (p=0.87).
In the multivariate analysis, one unit increase in age caused a significant increase of 1.05 times, an annual adjusted income per person less than 3265 TL increased it by 3.21 times and an altitude of 132 m or below increased it by 3.21 times (Table 3).
Discussion
It has been demonstrated in the studies conducted under experimental conditions that in recent years not only the prevalence but also the virulence has increased (9-12). However, difficulties caused by cross-reactions in the laboratory diagnosis of the disease and limitations in treatment options continue and there is no approved vaccine that can be used in humans (13). Thus, there is a need for epidemiologic data that are to be obtained from the studies in the world and our country and for protective measures that will be created with the use of this data. This study is a community-based WNV seroprevalence study and epidemiological data for Manisa province was obtained.
West Nile virus seroprevalence was reported as 11.0% in the 15-65 age interval in Iran by using the cluster sampling method and ELISA (14), it was reported as 24% in Egypt by using a random sampling method and PRNT (15). In a study conducted in Greece using a random sampling method from the general population and ELISA-PRNT tests, the mean seroprevalence in all age groups was reported as 2.1% and 1.5% respectively (16). In the present study, the surveillance was found to be 3.8% for Manisa province.
In three different studies conducted with healthy blood donors in our country between 2010 and 2015 with ELISA method, WNV seropositivity in 2821, 2516, 1200 serum samples were found as 2.4%, 0.99% and 1.6% respectively. While no virus was detected in the first of these studies with polymerase chain reaction (PCR), specific antibody presence was confirmed in nearly half of the samples that were detected to be positive with PRNT method in the other two studies (17-19). Blood donors usually consist of healthy young adults. However, in case a disease or infection finding is detected in the pre-donation examination of the blood donor candidates, these people are excluded from donation permanently or temporarily before blood is taken (20). WNV seroprevalence was detected to be low in blood donors in this study. This might be explained by the presence of people who cannot donate blood due to medical reasons and people older than 65 years of.
According to the annual data of the European Center for Disease Prevention and Control (ECDC), WNV infection is seen more frequently in men when compared to women (M/W ratio:1.8) (21). In the present study, while WNV IgG seroprevalence is similarly higher for men (M/W ratio:1.23), no significant difference was detected between the sexes (p=0.46).
In previous studies, it was reported that being over 50 years of age was a risk factor for being seropositive, that it increased seropositivity by 3.5 to 5.2 times and that seroprevalence also increased by increasing age (22-23). Similarly, we detected an association between increased age and increased WNV IgG seroprevalence significantly (p<0.001).
The results revealing a higher WNV seroprevalence in the study region may be due to the geographical characteristics of Manisa province. A big part of the province is located in the Gediz river basin and three dams and many streams as well as two important rivers, namely Gediz and Bakırçay, are located in this river basin (24). In this region, the genera of mosquitoes and their seasonal change throughout Manisa province, 3 of the Culex genus (Culex pipens, Culex martinii, Culex deserticola) were detected to be prevalent and C. pipens and C. martinii were reported as dominant species (25). Also, about 211 different bird species have been observed in this basin and changes are seen in their numbers during the migration seasons (26).
The presence of vector mosquito species is influenced by geographical conditions and altitude. In regions with low altitudes, vectors are more prevalent when compared with those in high altitudes (27). In previous studies, it was observed that viral spread was more frequently seen in regions with low altitudes (28) and that the risk decreased on mountainside (29). When the districts in this study were separated into two groups as those with a median altitude value below 132 m and those with a median altitude value over 132 m, WNV IgG seroprevalence was observed to be significantly higher in the districts with an altitude below 132 m (p=0.02). This significance continued also in the multivariate analysis and seropositivity risk was detected to be 3.2 times higher in people living in regions with an altitude below 132 m.
It has been demonstrated that increases in population density and housing population caused vector spread and increase (30,31) and that this situation increased and influenced the geographical distribution of the virus (29). The districts in the study region were separated into two groups as those with a population density over 80 people/km2 value and below 80 people/km2 value but no difference was detected in seroprevalence in terms of population density (p=0.97).
In low-income regions, increased vector activity (30), WNV seroprevalence (32) and neuroinvasive disease development risk (33) were observed to be higher than those in other regions. It has been reported that the presence of old houses in these regions could also be supplying appropriate reproduction areas for vectors (30). In the study, adjusted income per person was calculated with the OECD formula for 2014; WNV IgG seroprevalence was found to be significantly higher for people with an income below 3265 TL (p=0.03). The location of the toilet in the house was considered as constituting a risk factor for vector theme in addition to poverty indicator; WNV IgG seroprevalence was found to be higher for people with toilets outside the house in comparison to people with toilets in the house (p=0.01) (Table 3). However, in the multivariate model, it was observed that this significance disappeared. In addition, it was observed that the central and periphery of the living area did not affect the seroprevalence. When these data in the study were evaluated together, they suggest that the socio-economical factors specified in the previous studies could also be influential in WNV infection development in our country.
The impact of educational status in getting information on the disease and on the transmission routes has also been reported (34). Also, Rios et al. demonstrated that WNV and Saint Louis Encephalitis (SLE) virus cause a greater number of neuroinvasive diseases in people with a low level of educational status (33). In the study group, it was detected that as the educational level decreased, WNV IgG seroprevalence increased significantly (p<0.001). This difference was prominent, especially in illiterate or merely literate people or elementary school graduates.
The strong aspect of the study is that a sample comprising all populations over two years of age from the whole province chosen by random sampling was included in the study. The sample size is relatively big and it is considered to be reflecting the community. Although the results that were found to be indeterminate in the study were evaluated as negative, the fact that the positive antibodies were not confirmed by a method such as PRNT due to the lack of financial support may have caused the seroprevalence to be detected higher than it actually is, and this constitutes a limitation. Since there were no population-based seroprevalence studies on other flaviviruses in the region where the study was conducted, a healthy comparison could not be made in terms of cross-reactivity. In addition, detection of non-neutralizing antibodies to the nucleocapsid antigen and failure to detect neutralizing antibodies to envelope glycoproteins by ELISA may result in an overestimation of antibody seroprevalence.
Because there was no information in the health histories of participants related to a neuroinvasive disease, the lack of information on how many in this group with these risk factors developed neuroinvasive disease or death constitutes another limitation. Another limitation of the study is that the population densities were calculated roughly by dividing the district population by the district area. If a detailed population density map could have been created, a healthier analysis of whether there was a seroprevalence difference between the district centers which are thought to have a denser population and rural areas that presumably have a less dense population could have been made.
In conclusion, in the community based present study, which was conducted in the whole Manisa district, WNV IgG seroprevalence was detected to be 3.8% with ELISA method. In the study, no impact of the presence of an underlying disease, living in a rural area or population density was detected on seropositivity whereas an increase in age, low income and living in regions with a low altitude increased seropositivity significantly.