Introduction
At the beginning of the 21st century, three deadly epidemics related to coronaviruses have struck the world. Severe acute respiratory syndrome (SARS) originated in 2002, Middle East respiratory syndrome (MERS) in 2012, and finally COVID-19 caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in December 2019. COVID-19 first appeared as a respiratory disease in Wuhan city, Hubei province, China on December 31, 2019 (1). After a short time, SARS-CoV-2, the causative agent of the disease, was identified as a new β-coronavirus (2). The rapid spread of this epidemic from China to the whole world has caused global concern.
The disease continues to maintain its importance because there is no effective treatment and the vaccination is not at the desired level. Although the transmission rate and mortality rate vary from country to country, studies to determine the prognosis of COVID-19 are crucial. COVID-19 has a broad clinical spectrum ranging from asymptomatic infection to severe acute respiratory failure leading to death (1). Infected patients who initially present with mild symptoms may develop severe clinical manifestations later. Therefore, identifying risk factors that predict the prognosis of COVID-19 is critical.
In the majority of patients infected with SARS-CoV-2, the course of the disease is mild. The mortality rate is higher in severe and critical patients. Severe and critical patients are usually treated in the intensive care unit, while mild-moderate or non-severe patients are treated in the hospital’s inpatient ward or the outpatient department. Therefore, it is critical to identify the risk factors that predict disease prognosis on admission. Identifying risk factors at an early stage makes it possible to recognize patients at high risk. Thus, with close monitoring of these patients, earlier and more effective treatments can be administered, and in this way, mortality rates can be reduced. Indeed, studies have shown that some blood marker levels may be associated with COVID-19 severity and mortality (3).
In this retrospective observational study, we aimed to determine the factors that predict mortality in COVID-19 by examining the relationship between laboratory and demographic data at admission and the severity of COVID-19 in patients hospitalized with positive real-time polymerase chain reaction (RT-PCR).
Materials and Methods
Study Design and Participants
This retrospective cross-sectional study included patients who were SARS-CoV-2 RT-PCR-positive and admitted to a 350-bed public hospital between March 17 and September 21, 2020. RT-PCR assay nucleic acid amplification test was used directly with Bio-Speedy SARS CoV-2 Double Gene (ORF1ab and Nucleocapsid) RT-qPCR (Bioeksen Inc., Turkey). Real-time PCR was run on the CFX96 Touch System (Bio-Rad, USA). The threshold level to calculate the number of threshold cycles (Ct) was 200 RFU. A cycle threshold value less than 37 was defined as a positive result, and a cycle threshold value of 40 or more was defined as a negative test. Pregnant women (10 patients), patients hospitalized for another diagnosis who were found to have a positive SARS-CoV-2 RT-PCR test after initial admission (5 patients), and patients who did not accept and discontinued treatment (2 patients) were excluded. Informed consent was waived because the data used in the study were anonymous.
The Ethics Committee of Batman Training and Research Hospital approved the study on May 25, 2021, with decision number 274.
Data Collection
The data of patients, including age, gender, underlying disease (comorbidities), and laboratory test results, were obtained from the hospital database and patient records. The need for intubation, hospitalization in the intensive care unit (ICU monitoring), mortality and discharge, duration of hospitalization and number of days with symptoms on admission (i.e., on which day of their symptoms the patients presented to the hospital) were recorded. A specific period was not calculated as the mortality day. To compare relevant variables, the patients were divided into three groups according to the clinical severity of COVID-19 determined by their baseline clinical findings at admission: the mild-moderate patient group, the severe patient group and the critical patient group. When forming the patient groups, the following criteria were also considered (4):
1. Mild: Patients without pneumonia,
2. Moderate: Patients with pneumonia with respiratory symptoms but not accompanied by hypoxia,
3. Severe: Patients with a respiratory rate of 30/min or more, oxygen saturation less than 94%, PaO2/FiO2 < 300, pulmonary infiltration greater than 50% (involvement of more than 50% of the total lung parenchyma area as pulmonary infiltration or ground-glass opacities on thorax computed tomography) and dyspnea,
4. Critical: Patients with respiratory failure, septic shock, and multi-organ failure requiring mechanical ventilation.
In addition, the patients were divided into two subgroups by whether they died or survived to investigate the association of relevant variables with mortality.
Differences between groups in age, gender, underlying diseases (comorbidities), and laboratory blood test results (hemogram, neutrophil-to-lymphocyte ratio [NLR], C-reactive protein [CRP], procalcitonin, blood sedimentation, ferritin, lactate dehydrogenase [LDH], sodium, albumin, liver and kidney function tests, D-dimer, and cardiac enzyme results) were examined on the first day of hospitalization and statistically analyzed.
Statistical Analysis
All data analyses were performed with Statistical Package for Social Sciences (SPSS) 22.0 (IBM Corp., Armonk, NY, USA). Descriptive data were given as mean, standard deviation, numbers and percentages. Chi-square and Fisher’s exact tests were used for group comparisons, and Student’s t-test was used for comparisons of normally distributed continuous variables. The Kruskal-Wallis test, one of the nonparametric tests, was used to compare continuous variables that did not conform to the normal distribution. Results with a p-value of less than 0.05 were considered statistically significant. The severity of the disease was analyzed using the ordinal logistic regression model, and the development of mortality was analyzed using the multivariate logistic regression model.
Results
Descriptive Findings
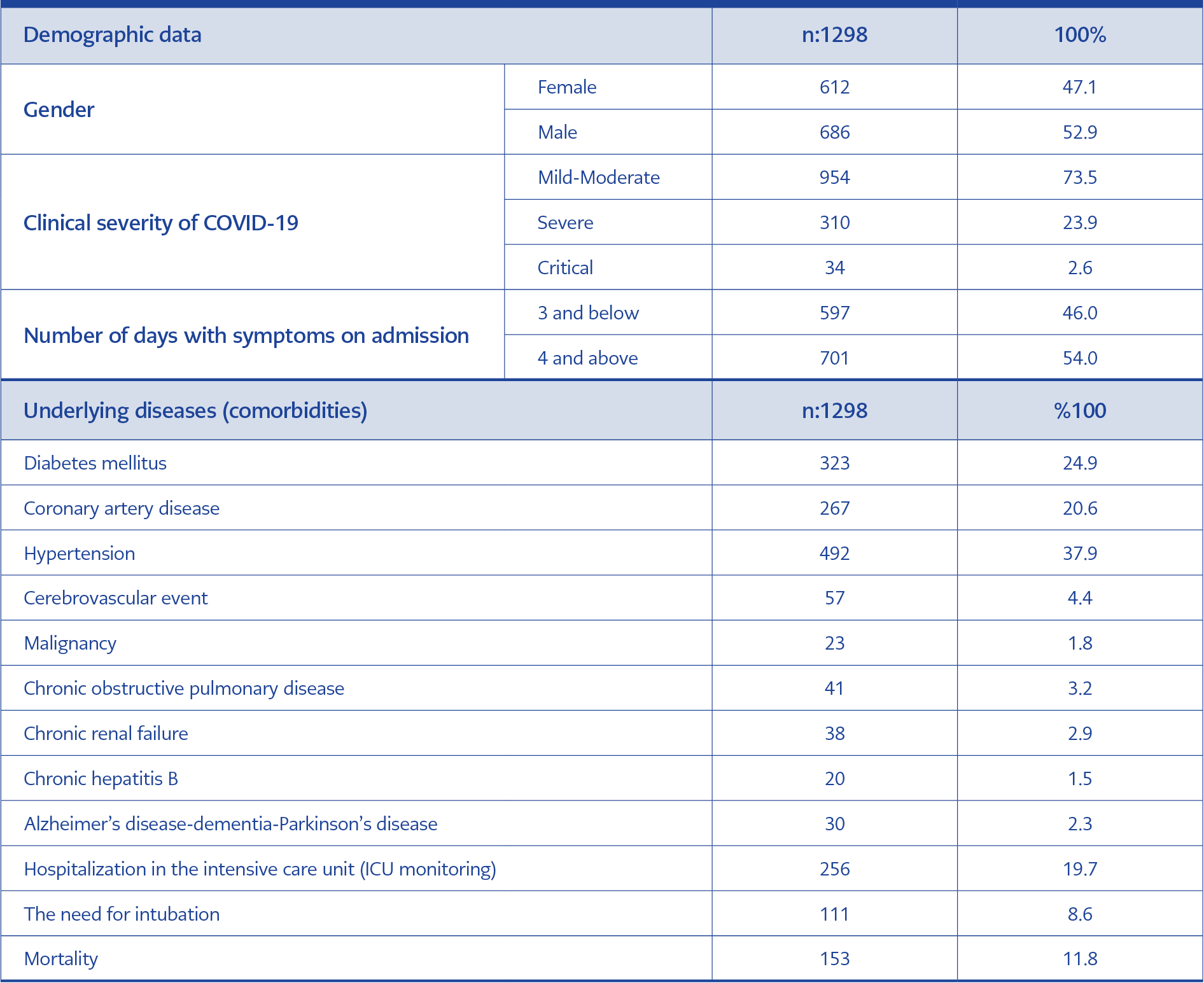
Information from 1315 SARS-CoV-2 RT-PCR-positive patients hospitalized and monitored with a provisional diagnosis of COVID-19 at Batman Regional State Hospital between March 17, 2020, (when the first case of COVID-19 was detected in Batman) and September 21, 2020, was retrospectively analyzed. One thousand two hundred ninety-eight patients aged 18 years and older were included in the study. Of the subjects, 686 (52.9%) were male. The mean age of the participants was 57.9±18.2 years. Nine hundred fifty-four (73.5%) patients had mild to moderate disease courses, 310 (23.9%) had severe disease courses, and 34 (2.6%) had critical disease courses. Of the study patients, 1042 (80.3%) were monitored in the ward and 256 (19.7%) in the ICU. One hundred fifty-three (11.8%) of all patients resulted in mortality (Table 1).
Comparative Findings
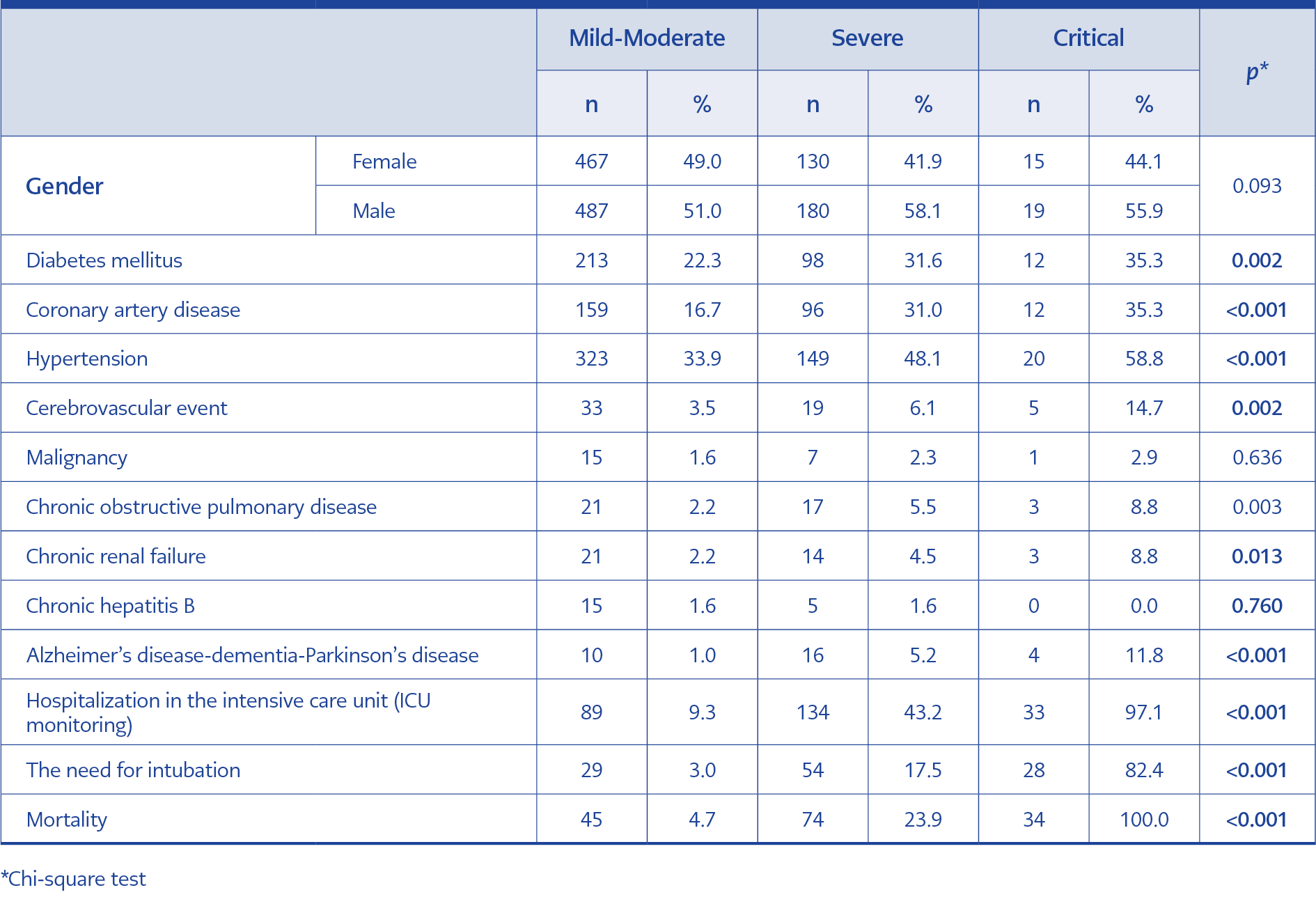
The rate of ICU monitoring was 9.3% in the mild-moderate patient group, 43.2% in the severe patient group, 97.1% in the critical patient group.
The mortality rate was 4.7% in the mild-moderate patient group, 23.9% in the severe patient group and 100% in the critical patient group. Finally, the need for intubation was detected at a rate of 3.0% in the mild-moderate patient group, 17.5% in the severe group and 82.4% in the critical group (p< 0.05) (Table 2). A significant correlation was found between clinical severity of disease and diabetes mellitus (DM), coronary artery disease (CAD), hypertension (HT), cerebrovascular event (CVE), chronic obstructive pulmonary (COPD), chronic renal failure (CRF), Alzheimer’s disease-dementia-Parkinson’s disease (p< 0.05) (Table 2).
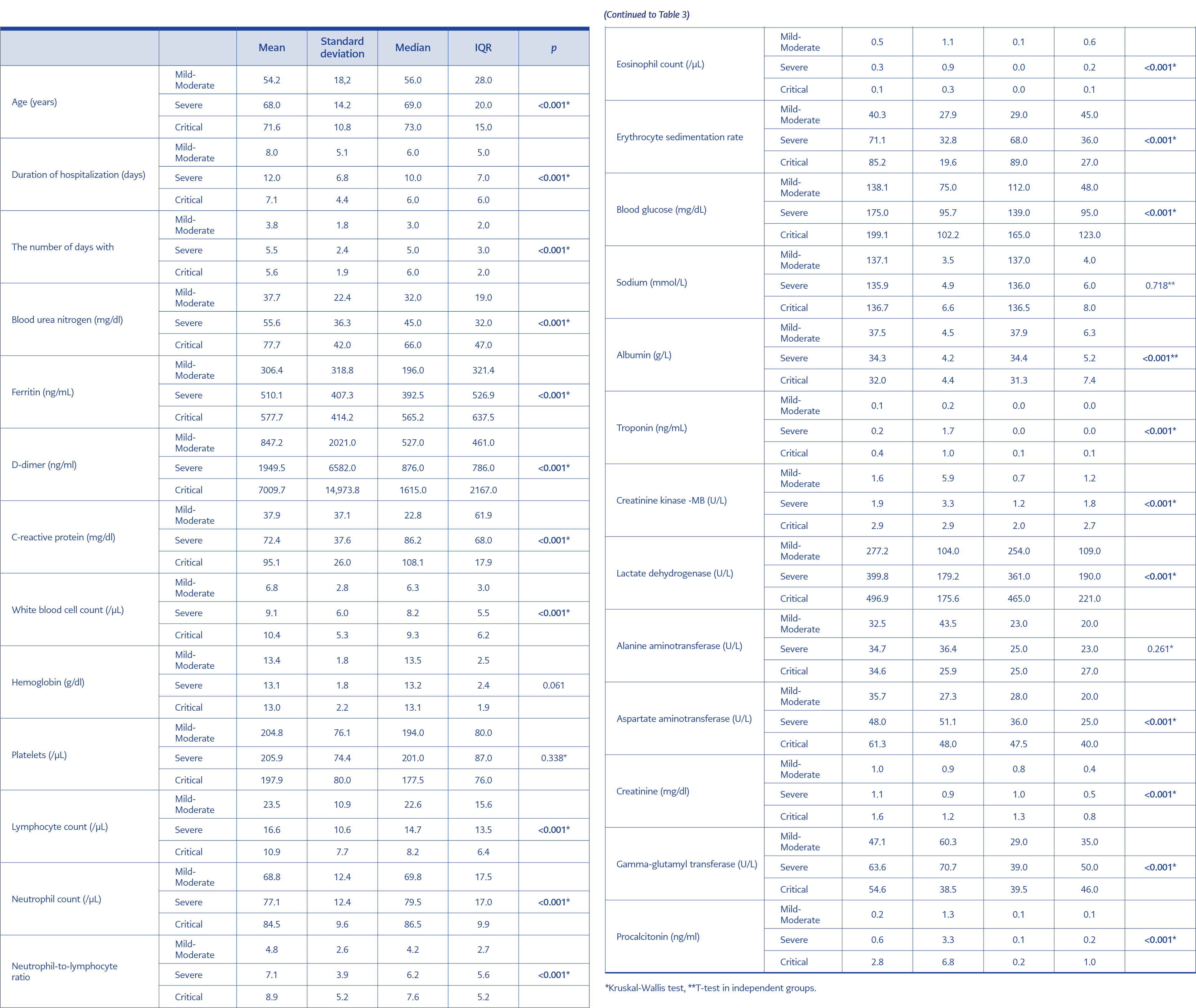
Table 3. Assessment of the mean age, duration of hospitalization and blood parameters according to the clinical severity of COVID-19.
In the comparison of mild-moderate, severe, and critically ill patient groups, a significant difference was found between the groups in terms of mean age, duration of hospitalization, and the number of days with symptoms on admission (p< 0.05) (Table 3). In blood tests performed on the day of hospital admission, blood parameters such as leukocytosis (mean value of significantly elevated white blood cells [WBC]), lymphopenia (mean value of significantly reduced lymphocytes) differed significantly among the three groups and were higher in the severe and critical patient groups than the mild-moderate patient group. While the mean values of NLR, ferritin, aspartate aminotransferase (AST), LDH, gamma-glutamyl transferase (GGT), procalcitonin, blood sedimentation, CRP, blood urea nitrogen (BUN), creatinine, D-dimer, glucose, creatinine kinase-MB (CK-MB) were significantly higher, the mean values of albumin were significantly lower. The difference between the three groups was statistically significant (p< 0.05) (Table 3).
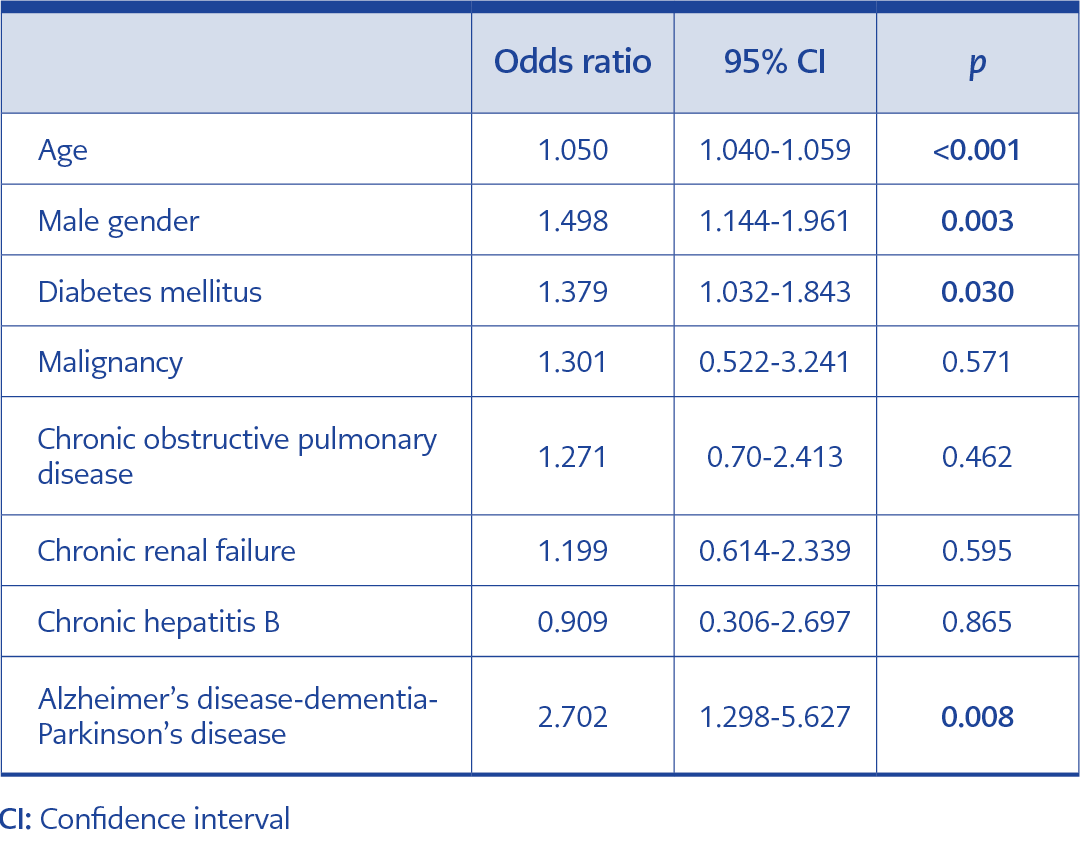
Table 4. Results from ordinal logistic regression analysis to identify risk factors independently associated severity.
Ordinal logistic regression analysis revealed that male gender (p:0.003, OR:1.498, 95% CI: 1.144-1.961), age (p<0.001, OR:1.050, 95% CI:1.040-1.059), DM (p:0.030, OR:1.379, 95% CI: 1.032-1.843), malignancy (p:0.571, OR:1.301, 95% CI:0.522-3.241), COPD (p:0.462, OR:1.271, 95% CI:0.670-2.413), CRF (p:0.595, OR:1.199, 95% CI:0.614-2.339), CHB (p:0.865, OR:0.909, 95% CI: 0.306-2.697), and Alzheimer’s disease/dementia/Parkinson’s disease (among neurological diseases) (p:0.008, OR:2.702, 95% CI: 1.298-5.627) were independently associated with the severity (Table 4).
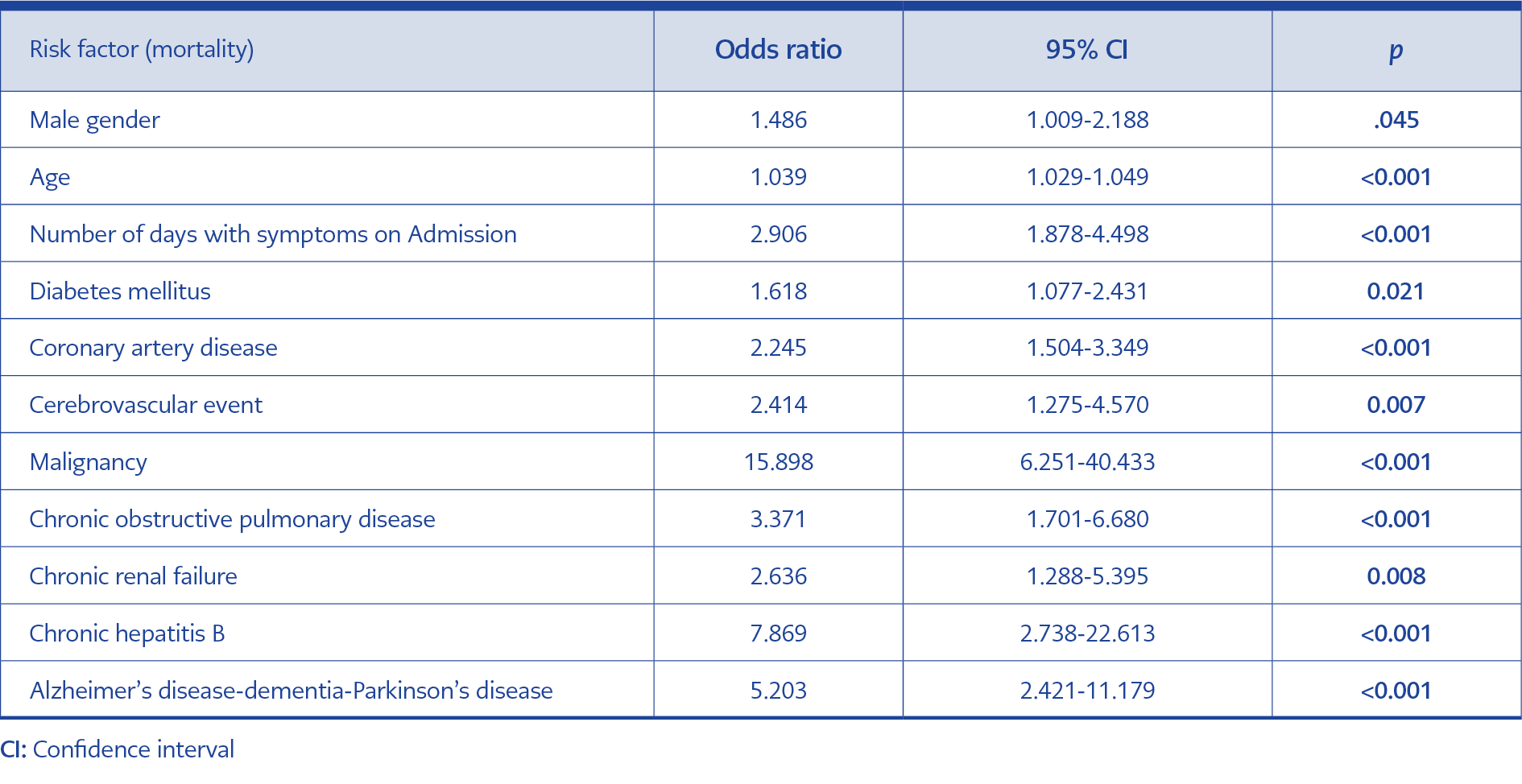
Table 5. Results from multivariate logistic regression analysis to identify risk factors independently
associated mortality.
In multivariate logistic regression analysis, male gender (p:0.045, OR:1.486, 95% CI:1.009-2.188), age (p<0.001, OR:1.039, 95% CI:1.029-1.049), number of days with symptoms on admission (p<0.001, OR:2.906, 95% CI:1.878-4.498), DM (p:0.021, OR:1.618, 95% CI: 1.077-2.431), CAD (p<0.001, OR:2.245, 95% CI:1.504-3.349), CVE (p:0.007, OR:2.414, 95% CI:1.275-4.570), malignancy (p<0.001, OR:15.898, 95% CI:6.251-40.433), COPD (p<0.001, OR:3.371, 95% CI:1.701-6.680), CRF (p:0,008, OR:2.636, 95% CI:1.288-5.395), CHB (p<0.001, OR:7.869, 95% CI:2.738-22.613) and Alzheimer’s disease/dementia/Parkinson’s disease (p<0.001, OR:5.203, 95% CI:2.421-11.179) were independently associated with mortality (Table 5).
Discussion
Our study investigated the factors predicting the severity and mortality of COVID-19. We followed up all patients with positive RT-PCR results in the first 7 months of the pandemic; especially in the first months by hospitalizing them. We assessed laboratory tests and clinical severity on the first day of hospital admission. Laboratory tests were performed mainly in patients who needed hospitalization for COVID-19. Each of the laboratory parameters played an important role in risk stratification and prediction of COVID-19 outcomes. In addition to the laboratory tests, other conditions were also important factors that influence the severity and prognosis of COVID-19. Only classifying the disease as mild-moderate, severe and critical based on the test results and other conditions on admission can indicate the prognosis of the disease, and help to hospitalize selected patients in the high-risk group earlier.
In our study, the mortality rate was high and the deaths were especially among male patients (p< 0.05). It was observed that as the mean age of the patients increased, the clinical severity also increased. Many studies demonstrating male gender and advanced age as critical factors on the severity and prognosis of COVID-19 supports our findings (5). An early study raised concerns about the gendered impacts of the COVID-19 outbreak and it was confirmed by many subsequent studies later on (6, 7, 8).
While males and females have the same prevalence of COVID-19 infection, males with COVID-19 appear to be at greater risk for worse outcomes and death regardless of age (9). In a study conducted in Denmark, 2281 (47.1%) of 4842 COVID-19 patients were male. However, it was found that males with COVID-19 infection had a 50% higher risk of severe events (such as ICU admission, intubation, or death) (10). In another study conducted in the United States, it was observed that 83.8% of patients receiving mechanical ventilation were male, and the proportion of younger patients was significantly higher in patients who were successfully weaned from mechanical ventilation (11). In a study from Wuhan, 49.1% of cases were classified as severe, and advanced age was also significantly associated with a severe clinical course (12).
Comorbidities such as HT and DM were noted more frequently in the severe-critical patient group than in the mild-moderate patient group in our study. This result is supported by many studies which have suggested that these additional diseases are risk factors for fatal COVID-19 cases (8, 13). Our study involving 1298 cases also revealed comorbidities such as DM, CAD, HT, COPD, CRF, and CVE as significant risk factors for severe infections and mortality in both groups (p< 0.05). Studies have reported that the prevalence of HT and DM is significantly higher in COVID-19 patients requiring ICU monitoring than in patients not admitted to the ICU (14, 15). In addition, the Centers for Disease Control and Prevention (CDC) states that individuals with HT are at increased risk for severe COVID-19 (16). Chronic renal failure was addressed in a study of 10,482 individuals with COVID-19. In that study, 419 participants had a CRF diagnosis with a mortality rate of 31.7%, while non-CRF patients had a mortality rate of 25.4% (17). In our study, CRF was also found to be a risk factor for mortality. In general, patients with CRF have a high prevalence of comorbidities such as HT, CAD and DM, leading to worse outcomes in COVID-19 patients (18). Another parameter associated with mortality and severe infections in our study was the diagnosis of malignancy. The presence of malignancy in the patients proved to be a risk factor for mortality. In a study conducted using the data from the database of the Republic of Turkey Ministry of Health, COVID-19 patients with hematologic malignancies were shown to be at increased risk for severe events compared to COVID-19 patients without malignancy; confirming the vulnerability of the immune system of patients with malignancies (19).
A study conducted on 534 patients from Turkey concluded that laboratory findings, underlying diseases and clinical classification of patients as mild-moderate and severe-critical were good predictors of disease prognosis. In this study, RT-PCR results of 296 patients were positive and the overall mortality rate was 9.92% during the surveillance of patients, while it was 35.3% in the severe patient group and 1.3% in the mild-moderate patient group (20). The results of our study agree with this study. The mortality rate in our study was 23.9% in the severe patient group, 100% in the critical patient group, and 4.71% in the mild to moderate group (p < 0.001). On the recommendation of the provincial pandemic committee, we monitored the patients with positive RT-PCR results and comorbidities in the first months of the pandemic in the hospital as much as possible. Most patients had mild to moderate clinical outcomes on admission. However, during follow-up, we found that the condition of these patients might worsen and they might require intensive care. Therefore, the mortality rate and ICU admission rate were higher than expected for patients with mild to moderate clinical outcomes. Mortality rates reported among countries and age groups vary widely, ranging from approximately 0.5% to 10%. There are also studies in the literature that report mortality rates of more than 20% in hospitalized patients (7). In our study, the mortality rate was as high as 11.8% as it included the hospitalized population.
The tissue damage caused by SARS-Cov-2 can induce the over activation of macrophages and granulocytes and the overproduction of proinflammatory cytokines. This event leads to a cytokine storm called secondary hemophagocytic syndrome and is referred to as macrophage activation syndrome (MAS) (21). In the diagnosis of MAS, persistent fever, high CRP, ferritin, D-dimer, alanine aminotransferase (ALT), AST, LDH values and many laboratory tests such as lymphopenia, thrombocytopenia, neutrophilia are informative. These laboratory findings of MAS can be seen especially in severe-critical patients (22). Therefore, these laboratory findings have an essential role in the poor prognosis of COVID-19. LDH, one of the laboratory findings of MAS, is an enzyme that plays a role in the conversion of lactate to pyruvate in the cells of most body tissues and increases after tissue destruction. Many studies have concluded that LDH is a potentially useful surveillance parameter for COVID-19 pneumonia and can help in detecting disease progression (12, 20). In our study, we also determined that LDH statistically significantly predicted the clinical severity of the disease (p< 0.01).
In our study, NLR, leukocytosis and lymphopenia in hemogram examination were found to significantly predict the clinical severity of COVID-19 (p< 0.001). As shown in a current study, lymphopenia was associated with prognosis in many studies since the first days of COVID-19 detection (12, 23). In another study, NLR was also the best predictor of severe COVID-19 (24). From this point of view, the hemogram findings of our study are compatible with the literature.
The present study established that high levels of procalcitonin, CRP and ferritin, which are positive acute phase markers, and low levels of albumin, which are negative acute phase markers, were influential in indicating severe disease and predicting the prognosis of it. Many studies in the literature are similar to our study in this regard. In a study, CRP levels of 28.7 μg/mL in non-severe disease and 47.6 μg/mL in severe disease were detected in 140 hospitalized COVID-19 patients (p < 0.01) (25). The results of many studies suggest that high procalcitonin, CRP, and decreased albumin levels can be used to predict severe courses in COVID -19 (19, 23, 25). The results of many studies suggest that high procalcitonin, CRP, and decreased albumin levels can be used to predict COVID-19 severity (19, 23, 25). Ferritin, one of the other acute-phase proteins, is known to be an important mediator of immune dysregulation, especially under extreme hyperferritinemia, through direct immunosuppressive and proinflammatory effects that contribute to cytokine storm. We showed the effects of ferritin on COVID-19 severity and mortality in our study. As in our study, the literature indicates that ferritin levels increase as a result of macrophage and hepatocyte activation in COVID-19 (1). Our study also determined D-dimer level to be associated with the severity and mortality of the disease. It is known that coagulopathy and embolic events in COVID-19 result from the binding of the virus to angiotensin-converting enzyme-2 receptors and various mechanisms such as endothelial damage and complement activation. In association, there is an increase in D-dimer levels. Studies have indicated that increases in D-dimer levels are an important marker to detect critical disease early (26, 27). In this regard, our study supports the literature.
The findings of our study demonstrated that early diagnosis and treatment were of great importance in COVID-19. We found an association between the number of days with symptoms on admission (for four and more days) and mortality (p< 0.05). This was attributed to the delayed diagnosis and treatment of patients.
Conclusion
As confirmed by the findings of our study, to predict the risk of progression to severe disease, laboratory parameters such as serum procalcitonin, CRP, D-dimer, ferritin, NLR, LDH and albumin values, and the presence of comorbidities such as DM, HT, CAD, CVE and CRF are critical. Therefore, patients with a high risk of developing severe and critical diseases should be monitored closely.
Our study has some limitations. It was designed as a single-center retrospective study. Our patients who resulted in mortality were not screened for secondary bacterial infections. We did not identify patients with MAS, and treatments given to the patients were not analyzed. Additional limitations include home steroid use, which may affect the results of blood tests performed during hospitalization, whether a secondary bacterial infection was present during hospitalization, and not questioning the use of antibiotics.
As inequalities in vaccination persist worldwide and the desired level of vaccination has not been achieved, it seems the number of infected and fatal cases will not decrease to the desired level. Therefore, it is increasingly important to identify the characteristics of severe and critical COVID-19 patients, investigate risk factors associated with mortality, and classify patients at the time of diagnosis to determine appropriate treatment protocols and provide access to early treatment.