Introduction
The first case of COVID-19 was reported on 10 March 2020 in Turkey (1). Various treatment options have been used in all countries because of the lack of proven treatment (2). Among these options, hydroxychloroquine (HQN) and azithromycin (AZ) combination is expected to treat the disease (3). These two drugs have been used among the patients with moderate levelslevel of disease in many countries since the first day of the diagnosis of symptomatic disease.
HQN is used to treat autoimmune diseases and malaria; AZ is used in the treatment of various infections (4,5). When used in their indications, the common side effect is QTc prolongation in electrocardiography (ECG) (6,7). Development of torsades de pointes, which is a form of polymorphic ventricular tachycardia, is the most undesirable situation in QTc prolongation.
Adverse cardiac events related to the use of HQN was reported (8, 9). We aimed to describe the adverse cardiac events associated with using both HQNhqn and azithromycinAZ for COVID-19.
Materials and Methods
We included 93 hospitalized COVID-19 patients who had positive polymerase chain reaction (PCR) test results or clinical and radiological findings . The patients who had QTc interval over 460 msec were excluded. The HQNhqn dose was 200 mg twice a day for five days with or without concomitant AZ 500 mg once a day. On the first day and fifth day of hospitalization, ECG findings of the patients were reviewed. In ECG records, PR interval, QRS duration, QT interval, QTc interval, and JTc interval were calculated. Bazett’s formula was used when calculating the QTc interval. The JTc interval was obtained by subtracting QRS duration from the QTc interval. The ECG values of the first day and fifth day were compared. QTc prolongation >40 msec or exceeding 470 msec was considered moderate, and prolongation > 60 msec or exceeding 500 msec erewas considered critical prolongation. In patients with QRS duration >120 msec, JTc duration was considered. JTc prolongation > 60 msec or exceeding 410 msec were considered as critical prolongation.
Statistical Analysis
Continuous variables were presented by mean and standard deviation, whereas categorical variables by percentages. The paired t-test was used in comparison ofto compare paired mean values;valuses, otherwise, the Mann-Whitney U test was used. Categorical variables were compared with a chi-square and Fisher exact test. Statistical analysis was performed using IBM SPSS Statistics 22. Statistical significance was set as p < 0.05.
Results
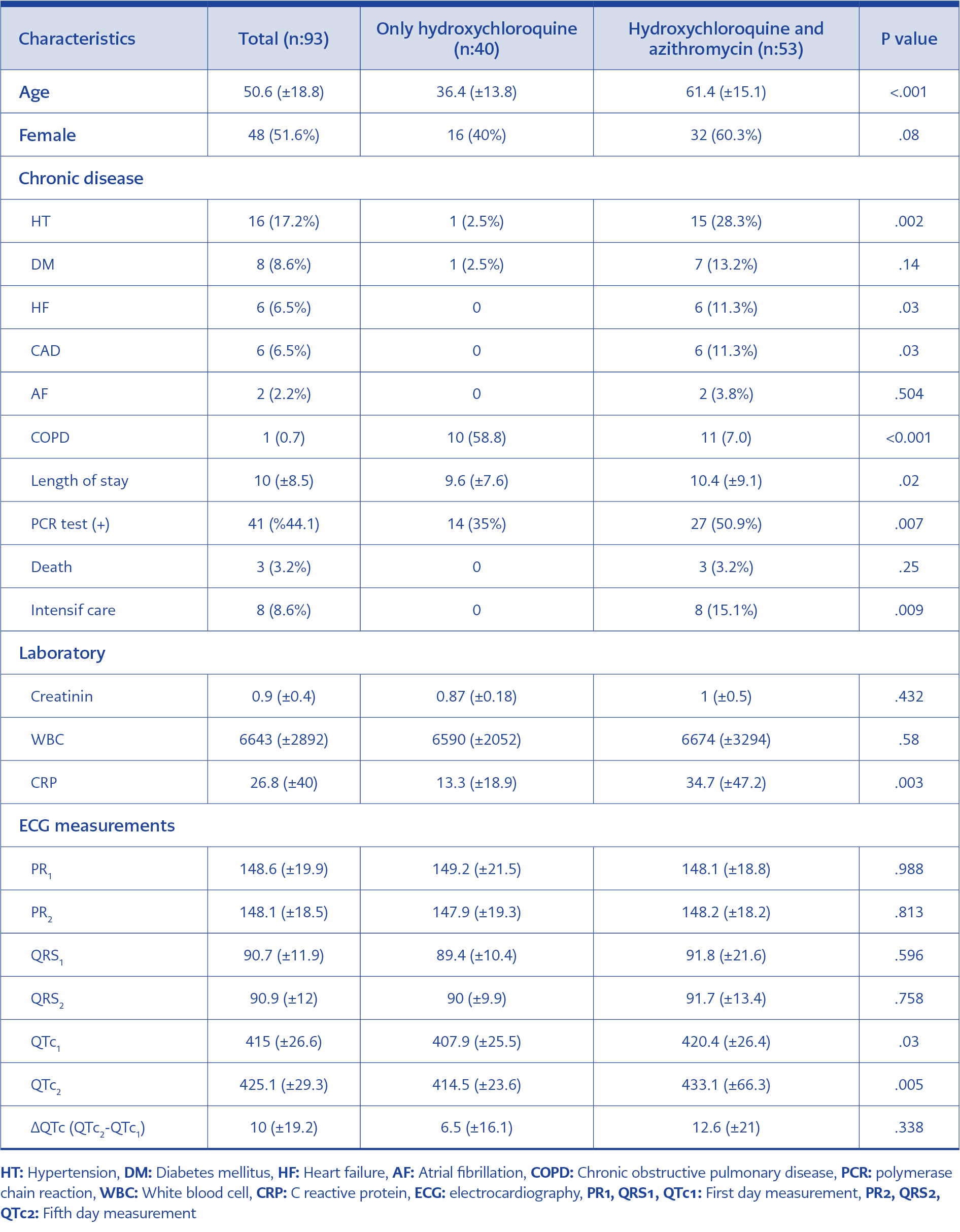
Among 93 patients, 40 (43%) patients received HQN monotherapy 53 (57%) patients received HQN and azithromycinAZ therapy. The mean age of the patients in HQN+AZ group was older than HQN group (Table 1). The proportion of comorbidities among HQN+AZ group was significantly higher than HQN group (Table 1). The PCR test positivity, being in intensive care, elevated C reactive protein (CRP) and prolongation ofprolonged hospital stay were higher in HQN + AZ group (Table 1).
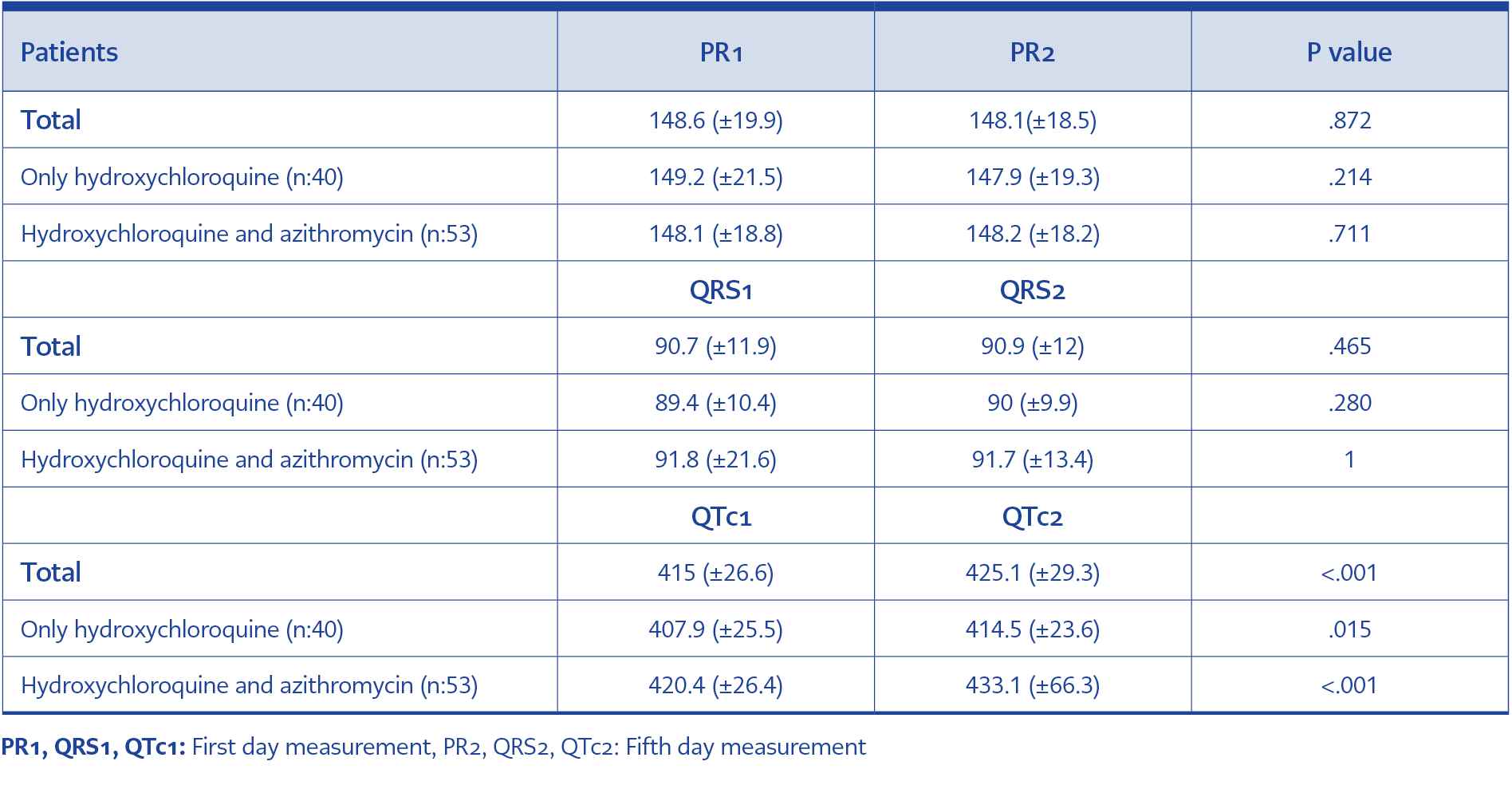
When the first day and fifth day ECG records are compared, no significant difference was found in terms of PR intervals (p=0.872) and QRS duration (p=465), however in the fifth day, QTc intervals were found to be significantly prolonged in HQN+AZ group ( 12.6+21) than the HQN group (6.5±16.1 ms), (p=0.05, Table 1).
The finding of ECG werewas detailed in Table 2. The QTc interval exceeded 470 msec in four patients, and in two patients, the QTc was prolonged > 40 msec. Moderate prolongation was detected in six patients (6.45%). QTc interval exceeded 500 msec in three patients and all of them were female in the combination therapy group. In two of these three patients, the QTc prolonged > 60 msec. The QRSother patient’s QRS duration was calculated as 144 msec on the first ECG and 142 msec on the second ECG. Therefore, JTc interval was evaluated on this patient. The first JTc interval was 346 ms, and the second JTc distance was 364 ms. This patient was not evaluated as critical QTc prolongation. Critical QTc prolongation was detected in two patients (2.15). Also, in the combination therapy group, this ratio was 3.7. One of the patients was admitted with acute atrial fibrillation and a beta-blocker was started, and then a sinus rhythm was provided by cardioversion.
In total, 13 patients received favipiravir treatment. One of them received both convalescent plasma and tocilizumab; one patient received only convalescent plasma, the other one only tocilizumab. None of these patients had prolonged QTc.
In total, three patients died (3.22%). The cause of death was reported as acute respiratory distress syndrome and no cardiac complications, including QTc prolongation, were detected in these patients.
Discussion
Since there was no specific drug for treatment, a variety of drugs were used in COVID-19 infection. Turkish Ministry of Health suggested HQN or HQN+AZ. Despite the frequent use of HQN for autoimmune diseases, reported cardiac side effects were limited in the literatüreiterature (10). However, it is known that it has effects on the conduction system by blocking potassium ion channels in the heart. Although side effects such as PR interval prolongation, QRS duration widening and atrioventricular block are also observed, QTc prolongation seems to be a more serious side effect recently (11). While this side effect was known to be rare by using for more than one year or acutely with eroverdose, recently the rate of side effects was reported up to 21% in using short-term and therapeutic dose (9, 11,12). In our study, no detection of QTc prolongation with HQN dose, was400 mg daily (total 2 gr), seems to be more compatible with literature and clinical practice.
AZ is a macrolide group antibiotic that is still used in respiratory and urinary tract infections (5). It is known that the drug affects multi-ion channels and causes heart conduction problems, especially QTc prolongation. Therefore, it is avoided to be used in elder ages, especially >60 years of age, because of the increased ratio of this side effect (13). Two drugs were previously used for three days in malaria treatment, and QTc prolongation was detected with no critical side effects.
Almost all the hospitalized patients in Turkey received treatment for at least five days, and this period was extended to 10 days as a result of clinical evaluations. At the end of the fifth day, we found critical QT prolongation in two (3.7%) patients receiving HQN and AZ combined therapy. One of the patients was on antiarrhythmic therapy, and the other one was 82 years old.
In HQN monotherapy treatmentHQN for five days, QTc prolongation was observed while no PR interval prolongation, no QRS widening was observed. However, the QTc prolongation did not reach the critical level after five days of treatment. Critical QTc prolongation in patients receiving dual therapy was observed in two of 53 patients. ECG monitorization can be more frequent in patients taking HQN and AZ together. Especially in patients with dysrhythmia, received antiarrhythmic therapy, with organic heart disease and > 80 years old, daily ECG monitorization seems to be appropriate.
In conclusion, close ECG monitoring is necessary, and the frequency of ECG monitorization should be individualized in patients receiving azithromycin with hydroxychloroquine.