Introduction
Hip and knee arthroplasties stand out as the most prevalent surgical interventions employed to address joint deformities, predominantly stemming from osteoarthritis associated with an aging population. In Turkey, a total of 34,240 hip arthroplasties and 81,109 knee arthroplasties were conducted in 2022. Meanwhile, in the United States of America (USA), the annual tally of hip and knee arthroplasties is estimated at around 1,000,000, with projections indicating a surge to 4,000,000 by the year 2030 (1). This substantial increase in arthroplasty procedures accentuates the growing significance of periprosthetic joint infections (PJI), recognized as the most prevalent, devastating, and high financial burden complication arising from arthroplasty (2).
The incidence of PJI following hip arthroplasty ranges from 0.5% to 1%, while for knee arthroplasty, the incidence was reported to vary between 0.5% and 2% (3, 4). In Turkey, the reported incidence of PJI after knee and hip arthroplasties in 2022 was 0.43% and 1.44%, respectively (5). However, difficulties in actively surveilling patients after discharge suggest that the true incidence of PJI may be higher than reported (6). Studies indicate that approximately one-fifth of PJIs may go undetected, not being reflected in surveillance data (6).
Many risk factors and risk prediction models for the development of PJI have been identified in the literature. Body mass index, diabetes mellitus (DM), male sex, immunosuppression, hypoalbuminemia, history of rheumatoid arthritis, National Nosocomial Infections Surveillance Score (NNIS), American Society of Anesthesiologists (ASA) score are among the most commonly reported patient-associated risk factors, and blood transfusion, prolonged operative time, previous surgery are the most frequently identified among surgery/hospital-associated risk factors (7-10). Hence, it is imperative to accurately determine the true incidence of PJI, identify associated risk factors, develop suitable infection control programs, target high-risk patient groups, and establish postoperative care recommendations.
This study primarily aimed to ascertain the incidence of early-stage knee and hip PJI within our center and identify the associated risk factors specific to knee and hip PJI.
Materials and Methods
This study was designed to be monocentric, prospective, and descriptive, spanning the period from June 15, 2018, to June 15, 2019. All patients aged 18 years and above who underwent knee and hip arthroplasties were included. An average of 400-450 knee arthroplasties and 200-250 hip arthroplasties were performed annually in our center. The operating rooms dedicated to arthroplasty were equipped with vertical laminar flow, and a HEPA filter was present. The surgical team involved in the operations wore masks and disposable surgical aprons. Skin preparation for patients involved the application of povidone-iodine solution and 3M™ Ioban™ 2 antimicrobial incise adhesive drape. Knee arthroplasties were executed using the medial parapatellar arthrotomy approach, while hip arthroplasties were conducted employing the modified anterolateral approach for all cases.
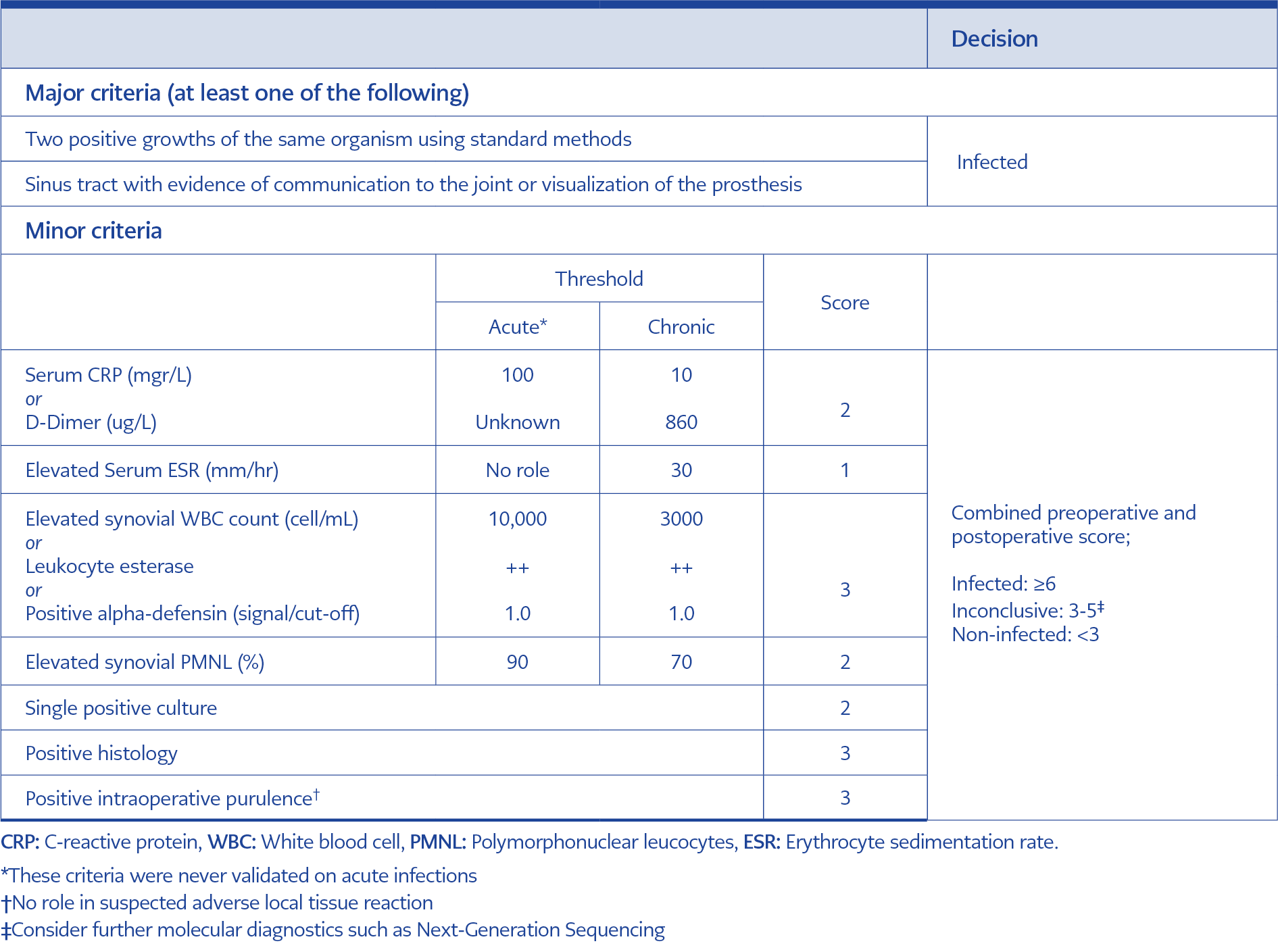
Cases of PJI following shoulder and ankle arthroplasties, revisions performed after PJI, and patients lacking postoperative follow-up for a 90-day period were excluded. Shoulder and ankle arthroplasty were excluded because of the lack of comparable national surveillance data in our country, and risk factors were different than knee and hip arthroplasty. Patients undergoing revision arthroplasty for PJI were excluded because the incidence of PJI was higher than that of primary arthroplasties. Early-onset PJI was defined as complications arising within the first 90 days postoperatively (11). The diagnosis of prosthetic joint infection adhered to the criteria outlined in the International Consensus Meeting on Periprosthetic Joint Infections, published in 2018 (Table 1) (12).
The Clinical Research Ethics Committee of Gazi University approved the study on November 26, 2018 with decision number 875. Informed consent was obtained from all individual participants included in the study.
Study Protocol
This study included consecutive patients who underwent hip and knee prosthesis surgeries. The recorded and evaluated demographic information included age, gender, body mass index (BMI), comorbid diseases, the existence of a secondary prosthesis, prior history of prosthetic joint infection, perioperative hospitalization periods, preoperative anemia (serum hemoglobin levels below 13 g/dL in men, 12 g/dL in women), presence of hypoalbuminemia (serum albumin level below 3.5 g/dL), surgical prophylaxis, operation time, type of operation (emergency-elective, primary-secondary), type of anesthesia (general or spinal/epidural), and transfusion history during hospitalization.
Threshold hemoglobin values for allogeneic blood transfusion were determined as 8 g/dL in asymptomatic patients and 10 g/dL in symptomatic patients (13). The study employed the NNIS and the ASA score systems to calculate and assess the risk of PJI (14-15). Continuous evaluation for PJI development was conducted daily during the patient’s hospitalization. In the post-discharge period, patients were prospectively monitored for PJI development in the Infectious Diseases and Clinical Microbiology or Orthopedics and Traumatology clinics. Clinical and laboratory data were extracted from patient files, the NUCLEUS v9.23.3 hospital database, and patient medical records.
Power Analysis
The incidence in the community typically ranges from 0.5% to 2% (3-4). In our study, the determined PJI incidence was 2.88%. The study’s statistical power, with a 95% confidence interval (CI), was calculated to be 95.07%, considering a type-1 error of less than 0.05 for the sample size of 590 within the study’s scope. The power analysis for the study was conducted using the OpenEpi version 3.1 publicly available statistics software.
Statistical Analysis
The statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) 20.0 (IBM Corp., Armonk, NY, USA). The normality of data distribution was assessed with histogram/Q-Q plots and Shapiro-Wilk tests. Categorical variables are reported as frequency and percentage and analyzed with a chi-square test. Continuous variables were presented as mean and standard deviation (SD) or median values and an interquartile range (IQR) of 25%-75%. Non-parametric values were analyzed using the Mann-Whitney U test.
The statistical significance was set as p<0.05. For determining early-onset PJI risk factors, multivariate models included variables such as comorbid disease, preoperative hospitalization time, NNIS score, and postoperative erythrocyte replacement, with a p-value of <0.2 in univariate analysis. Although the p-value was <0.2 in univariate analysis, venous insufficiency, anemia, chronic kidney disease, osteoporosis, prosthetic infection history, surgical site, and anesthesia type were excluded from the multivariate models due to the number of cases in the groups being less than 10. Additionally, operation time was excluded from the model due to its high correlation with the NNIS score included in the model.
Results
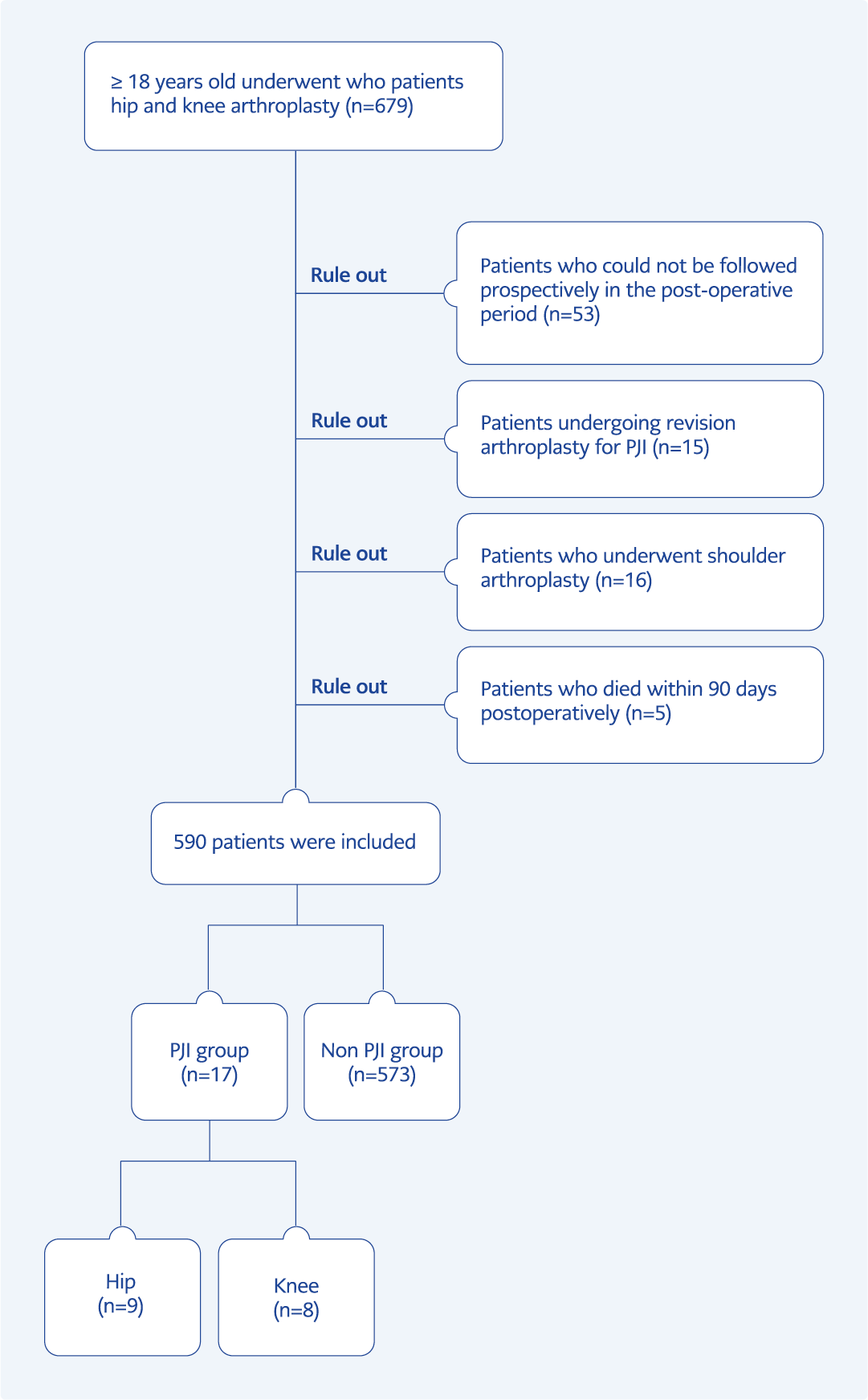
Although 679 patients who underwent arthroplasty were initially evaluated, 590 were ultimately included in the study. Hip arthroplasty was performed on 185 (31.4%) patients, while knee arthroplasty was performed on 405 (68.6%) patients. Of all arthroplasties, 553 (93.7%) were performed electively, while 37 (6.3%) were done under emergency conditions. The study’s flowchart is illustrated in Figure 1.
Primary arthroplasty was performed on 532 (90.2%) patients, while 58 (9.8%) underwent revision arthroplasty for mechanical reasons. The average age of the patients was 65.2 years (ranging from 22 to 99). Among the patients, 473 (80.2%) were female, and 117 (19.8%) were male. The average operation time was 99.9±32.6 minutes. Antimicrobial prophylaxis was administered to all patients within 30-60 minutes before arthroplasty, with cefazolin sodium being the most commonly used (95.4%). All patients received a single dose of antimicrobial prophylaxis within 30-60 minutes before arthroplasty. For prolonged procedures (>3 hours) or major blood loss (>1500 mL), intraoperative doses were repeated at intervals of one to two times during the procedure. Thromboembolism prophylaxis for patients undergoing arthroplasty included enoxaparin 40 mg/day for three weeks. Tranexamic acid was not administered to the patients. The reasons for this were the lack of a standard approach in the literature, the use of drains for perioperative bleeding control, and the risk of thromboembolism for elderly patients. No hematoma was observed during the hospital follow-up. Drains were used in 442 patients (74.9%) and removed on the second postoperative day. Prolonged drainage of fat necrosis was not observed. Autologous blood transfusion was not administered, and all blood transfusions were allogeneic.
None of the five patients who died within 90 days postoperatively and were excluded from our study developed PJI. The most common cause of death was pulmonary thromboembolism (three patients). The incidence of PJI was reported as 2.88%. Among the affected patients, 9 (52.9%) developed an infection after hip arthroplasty, while 8 (47.1%) developed it after knee arthroplasty. The incidence of PJI after hip arthroplasty and knee arthroplasty was 4.86% and 1.9%, respectively.
The three most frequently detected pathogens were methicillin-sensitive Staphylococcus aureus (n=6, 35.2%), Klebsiella pneumoniae (n=4, 23.5%), and Pseudomonas aeruginosa (n=2, 11.7%) respectively. Four of the PJIs were hematogenous.
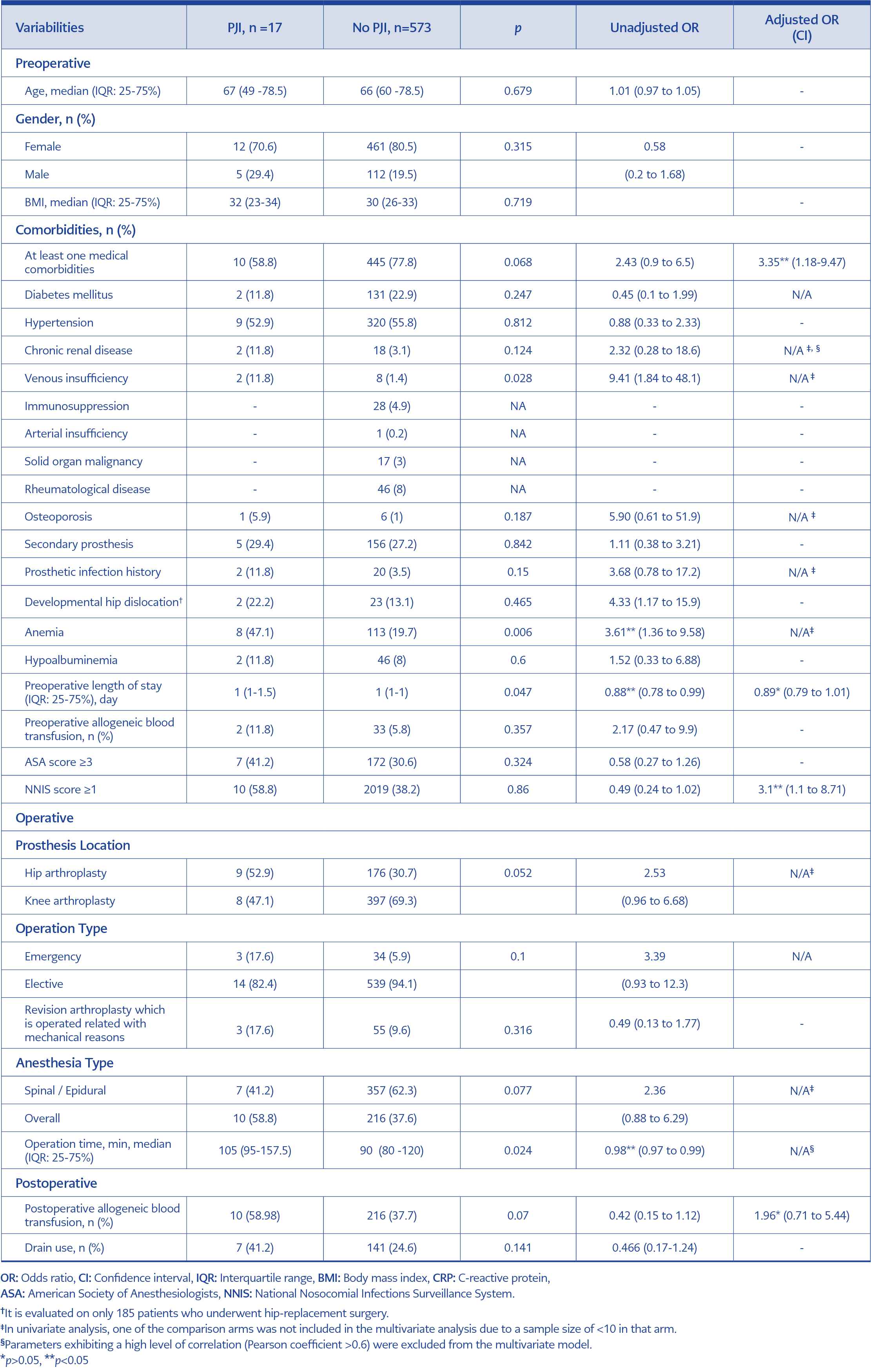
Patients with and without PJI were compared to identify potential risk factors. Various preoperative, perioperative, and postoperative surgical variables deemed significant predictors in the literature were evaluated. The results of both univariate and multivariate analyses are presented in Table 2.
Discussion
In our study, a comparison of the incidence of early-onset knee and hip PJI with national and local surveillance data revealed a higher PJI incidence, particularly after hip arthroplasty. We identified the presence of at least one comorbid disease, preoperative length of stay, and postoperative erythrocyte replacement as potential risk factors for PJI development. Additionally, NNIS score proved effective in predicting the development of PJI.
This study indicated a higher incidence of knee and hip PJI compared to national and international literature (3-5, 16). In a recent single-center study from Turkey, the incidence of total PJI was 1.1% and 2.4% in total hip and knee arthroplasties, respectively (17). Existing research suggests that national surveillance data might not accurately reflect the actual PJI incidence. Studies, such as those of Zhu et al. in New Zealand and Gundtoft et al., showed discrepancies between local and national data, with the true incidence being higher than reported (11, 18). Similarly, in our center, despite active surveillance, it was observed that not all PJIs were captured in the surveillance data. This highlights challenges in post-discharge follow-up, underreporting by diagnosing physicians, and insufficient bacteriological examinations in outpatients, contributing to an underestimated PJI incidence. The study proposes that addressing these surveillance issues is crucial for accurate identification and reporting of PJI cases, serving as a guide for future studies aiming to rectify deficiencies in local and national PJI surveillance.
Our study identified medical comorbidities as a risk factor for the development of PJI. This finding aligns with previous research that has also demonstrated an increased risk of PJI associated with medical comorbidities (19-21). Notably, DM is frequently linked to the risk of PJI. Lai et al. conducted a study evaluating the effects of single and cumulative medical comorbidities on PJI risk, revealing associations between DM, the number of medical comorbidities, and infection risk (19). Other frequently mentioned comorbidities include obesity, rheumatoid arthritis, cardiovascular diseases, chronic renal failure, chronic pulmonary disease, peripheral vascular disease, and immunosuppression (8, 22). However, due to limitations in the PJI sample size in this study, a detailed evaluation of which medical comorbidities specifically increase the risk of PJI was not possible.
NNIS is a scoring system designed to predict the risk of surgical site infection in the postoperative period. It incorporates three parameters: wound class of the operation, ASA score (≥3), and operation time (≥120 min) (15). Several studies have indicated that an NNIS risk index of 1 or higher can effectively predict the risk of periprosthetic joint infection (PJI) (23-26). In this study, patients with an NNIS score of 1 or higher exhibited an approximately three-fold increase in the risk of PJI. Despite efforts to enhance the NNIS risk score by incorporating additional parameters associated with PJI risk, studies have shown that the predictive value of NNIS remains robust. For instance, the inclusion of variables such as age, emergency surgery, trauma, general anesthesia, number of hospital beds, and type of surgery (primary arthroplasty, revision arthroplasty, partial arthroplasty) did not significantly improve the ability of NNIS to predict PJI risk (27, 28). These results suggest that NNIS continues to be an effective and valuable scoring system for clinicians to predict PJI.
Allogeneic blood transfusion is identified as a known risk factor for various infections, including PJI (29, 30). The detrimental impact of allogeneic leukocytes on neutrophil chemotaxis, suppression of T-helper 1 cells, and T-cell dysregulation resulting from the relative increase in T-helper 2 cells are the mechanisms responsible for this association (31). The PJI group exhibited a higher frequency of postoperative allogeneic blood transfusion within our study. It is suggested that practices aimed at reducing the need for transfusions in the postoperative period may contribute to a decrease in the development of PJI. However, the impact of this effect could not be conclusively defined in the multivariate analysis due to the small sample size in the PJI group.
In this study, patients with PJI were observed to have a longer preoperative hospital stay than those without PJI. However, this effect could not be conclusively demonstrated in the multivariate analysis due to the small number of patients. Previous studies have indicated that prolonged hospital stays are associated with increased exposure to nosocomial and virulent organisms, potentially contributing to the development of PJI. Hence, the literature recognizes hospitalization as one of the risk factors for PJI (32).
This study has several limitations. Firstly, the sample size for knee and hip PJI is relatively small, as the research was conducted for only one year at a single center. Consequently, it was challenging to obtain a sufficient number of patients for meaningful statistical comparisons to assess risk factors. In addition, the insufficient number of cases prevented the distinction between total, partial, and trauma-related arthroplasties in terms of PJI risk factors. Secondly, the absence of prior PJI data from the same center did not allow a comparison, making it difficult to determine whether the high incidence observed was periodic and identify the risk factors influencing PJI. In our center, there was no standard approach for patient-related risk factors such as diabetes mellitus, a high BMI, and active smoking. These factors were not included in the design and data of our study. Another limitation is tranexamic acid, which has been shown to reduce hematoma formation and therefore affects surgical site infection risk factors such as the need for drainage and blood transfusion, was not used. In addition, standard masks were used in our study instead of sterile closed-air circuit helmet systems, which have become important in arthroplasty operations. These factors may be important for future developments in infection control prevention.
In conclusion, the observed high incidence of knee and hip PJI highlights potential deficiencies in PJI surveillance at both local and national levels. Awareness of PJI risk factors, including NNIS scores and comorbidities, can serve as a guide for identifying high-risk patients before and after surgery. Addressing modifiable factors and implementing an effective infection control program is crucial for mitigating the risk of PJI in orthopedic arthroplasty procedures.