Introduction
In December 2019, Wuhan city, the capital of Hubei province in China, became the center of an outbreak of pneumonia of unknown cause. By Jan 7, 2020, Chinese scientists had isolated a novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2; previously known as 2019-nCoV), from these patients (1, 2). World Health Organization designated the disease as coronavirus disease 2019 (COVID-19) in February 2020 (3). The first case has been reported on March 11th in Turkey. The outpatient clinics, emergency departments, clinics and intensive care units (ICU) were reorganized according to the follow-up of patients diagnosed with COVID-19.
According to the data obtained that far, it was known that the symptoms of the disease began to appear within 4-6 days after contact, but the incubation period could extend up to 14 days (4). Fever, weakness, dry cough and anorexia were the most common symptoms in symptomatic patients (5). Advanced age, diabetes mellitus (DM), hypertension (HT), cardiovascular disease, chronic and kidney disease increases the risk of severe infection (6). After March 15, 2020, patients diagnosed with possible and definite COVID-19 were treated in our hospitals clinics and intensive care units.
This study aimed to investigate the differences in the patients treated ICU in terms of demographic, epidemiological, clinical, laboratory, and treatment protocols.
Method
Study design and participants
The study was designed retrospectively at Erciyes University Medical School. Patients with definite COVID-19 diagnoses treated in pandemic patient follow-up clinics and ICUs between March 15, 2020, and May 30, 2020, were included in the study.
Patients who met the definition of a possible case and had a hospitalization criterion were admitted to the clinics or intensive care units. Combined throat and nose swab samples were taken from the patients at the time of hospitalization.
The Republic of Turkey Ministry of Health guideline made case definitions (7). According to this guide, patients with one of the following criteria were classified as possible cases:
A: At least one of the signs and symptoms of fever or acute respiratory disease (cough and respiratory distress), AND if the diagnosis of COVID-19 cannot be ruled out with the current clinical presentation, AND a history of being abroad of himself or his relative within 14 days before the onset of symptoms.
B: At least one of the signs and symptoms of fever or acute respiratory disease (cough and respiratory distress) AND close contact with the confirmed COVID-19 case within 14 days before the symptoms.
C: At least one of the signs and symptoms of fever and severe acute respiratory infection (cough and respiratory distress), AND presence of hospitalization due to severe pneumonia AND failure to explain the clinical picture with another cause/disease.
D: Cough or shortness of breath with a sudden fever and no runny nose.
Definitive Case: SARS-CoV-2, for the cases meeting the possible case definition, was identified using a real-time RT-PCR method.
Data collection
Epidemiological, demographic, clinical, laboratory, treatment, and outcome data were extracted from the hospital’s electronic medical records.
Laboratory and radiological imaging procedures
A chest x-ray or tomographic examination was performed to the patients with respiratory symptoms. Laboratory diagnoses of COVID-19 cases were made in respiratory specimens by real-time RT-PCR method. The second COVID-19 PCR sample was sent 24 hours after the first negative result of the patient who clinically, laboratory, or radiologically likely to be COVID-19. Control RT-PCR samples were taken from patients with stable vital signs for 72 hours during treatment. Periodic respiratory tract samples were taken from COVID- 19 PCR-positive patients in 48 hours. Patients who were COVID-19 PCR-negative in the combined nasal and throat swab samples were discharged. Routine blood examinations were complete blood count, coagulation profile (prothrombin time, active thromboplastin time, INR, D-dimer, and fibrinogen) serum biochemical tests (including renal and liver function, creatine kinase, lactate dehydrogenase, and electrolytes), myocardial enzymes (Creatine phosphokinase, troponin, and Pro-BNP). Also, serum ferritin, CRP, and procalcitonin were monitored periodically.
Patients with at least one of the following criteria were defined as severe cases: (1) Respiratory rate >30/min. (2) Oxygen saturation of ≤93%. (3) PaO2/FiO2≤300 mm Hg. They were transferred to the intensive care unit (7).
Demographic data, risk factors, comorbidity, symptoms, physical examination, and laboratory findings on admission, follow-up, and the treatments were evaluated. We compared ICU and non-ICU patients and investigated whether these factors were risk factors for admission to intensive care units or not.
Statistical analysis
Histogram, q-q plots, and Shapiro-Wilk’s test were applied to evaluate the data normality. Levene’s test was used to test variance homogeneity. While comparing the differences between non-ICU and ICU groups, independent-samples t-test or Mann-Whitney U test was applied for continuous variables, and Pearson’s chi-square analysis or Fisher’s exact test for categorical variables. Kaplan-Meier plots were generated to compare the survival probabilities of patient groups. Moreover, univariate and multiple Cox proportional hazards regression analyses were conducted to identify the risk factors of survival time. Significant variables at p<0.05 were included in multiple models, and forward elimination was performed using the Wald test. The proportional hazards assumption was checked using the Schoenfeld residuals. Hazard ratios were calculated with 95% confidence intervals. Hosmer-Lemeshow test was used to assess the goodness of fit of the built model. All p values were adjusted using the Benjamini-Hochberg procedure to control for multiple testing. Statistical significance was set as p<0.05. All analyses were conducted using R 3.5.1 (www.r-project.org) and TURCOSA (Turcosa Analytics Ltd. Co., Turkey, www.turcosa.com.tr) software.
Results
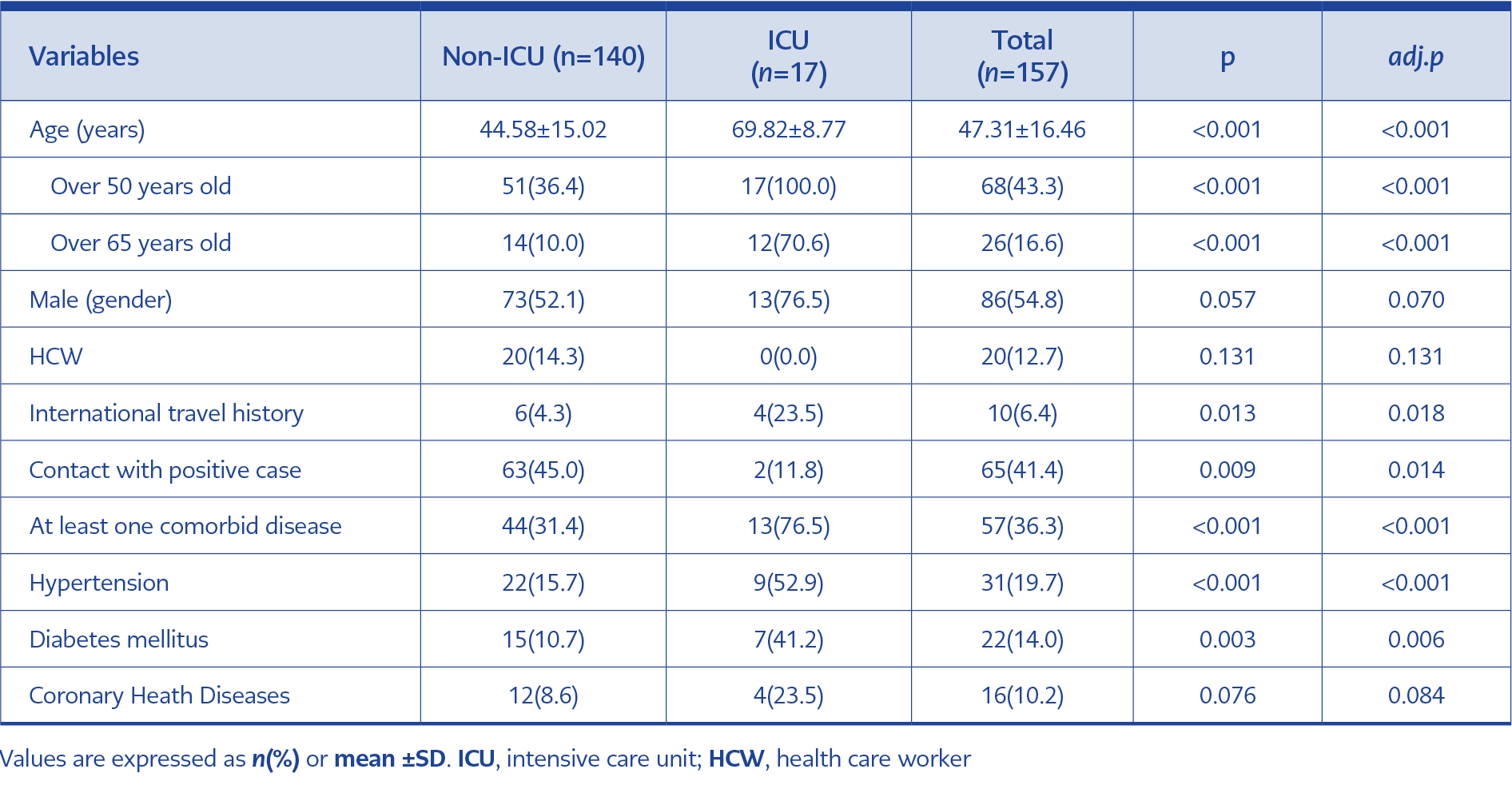
During the study period, 870 patients were hospitalized with the possible case definition, and 157 with definitive diagnoses were included. Seventeen patients needed ICU. The percentage of healthcare workers was 13%. Ten (6%) of the patients had a history of traveling abroad in the past 14 days, and four needed ICU during treatment. Of the patients, 41% had a history of contact with confirmed COVID-19 cases in the family or at work. The time from onset of symptoms to hospital admission ranged from one to 15 days (median three days). The comparison of patient characteristics between ICU and non-ICU groups are shown in Table 1.
The median age was 47, and 55% were male. Of the patients, 43% were over 50 years old, and 16% were over 65. All ICU patients were over 50 years old, and 70% were over 65. The percentage of patients over 65 and 50 years old was higher in ICU patients than the non-ICU group. Of the patients, 36% had at least one comorbid disease, the rate in ICU patients (76%) were significantly higher than those who did not need ICU (31%) (p <0.001). The most common comorbid diseases were HT (20%), DM (14%), coronary artery disease (10%), and respiratory diseases (8%). The rate of HT and DM was significantly higher in ICU patients (p <0.001 and p = 0.003, respectively).
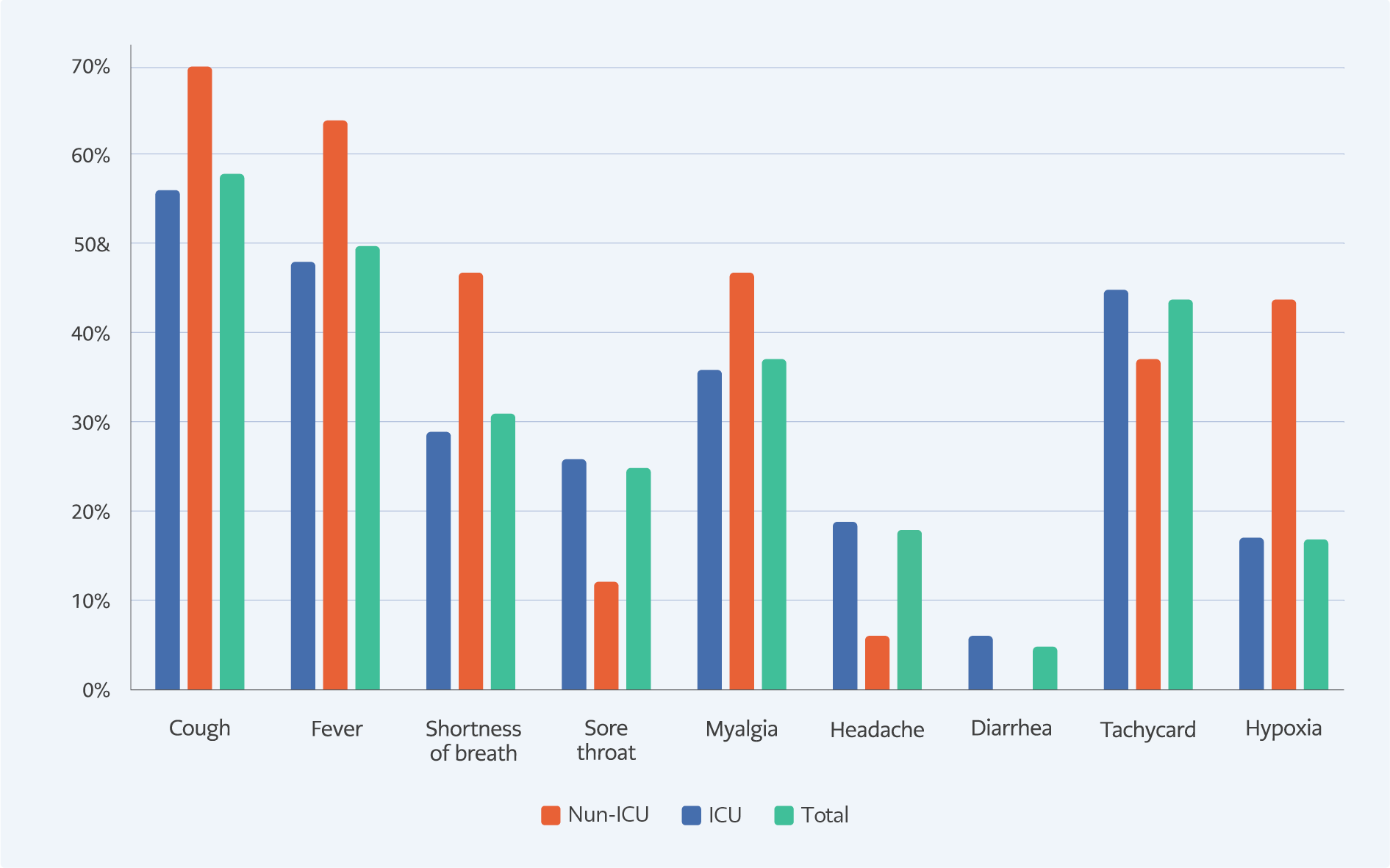
Figure 1. Comparison of patient symptoms and physical examination results between ICU and non-ICU groups
On admission to the hospital, the most reported symptom was cough (58%). Other symptoms were high fever (50%), myalgia (37%), shortness of breath (30%), sore throat (25%), and headache (18%). 40% of the patients had tachycardia, 24% high fever, and 17% hypoxia. The symptoms and physical examination results of the patients are shown in Figure 1. Fever, cough, dyspnea, myalgia and hypoxia presence on physical examination were higher in ICU patients than in non-ICU patients. The high rate of hypoxia on admission was statistically significant in ICU patients (p = 0.007).
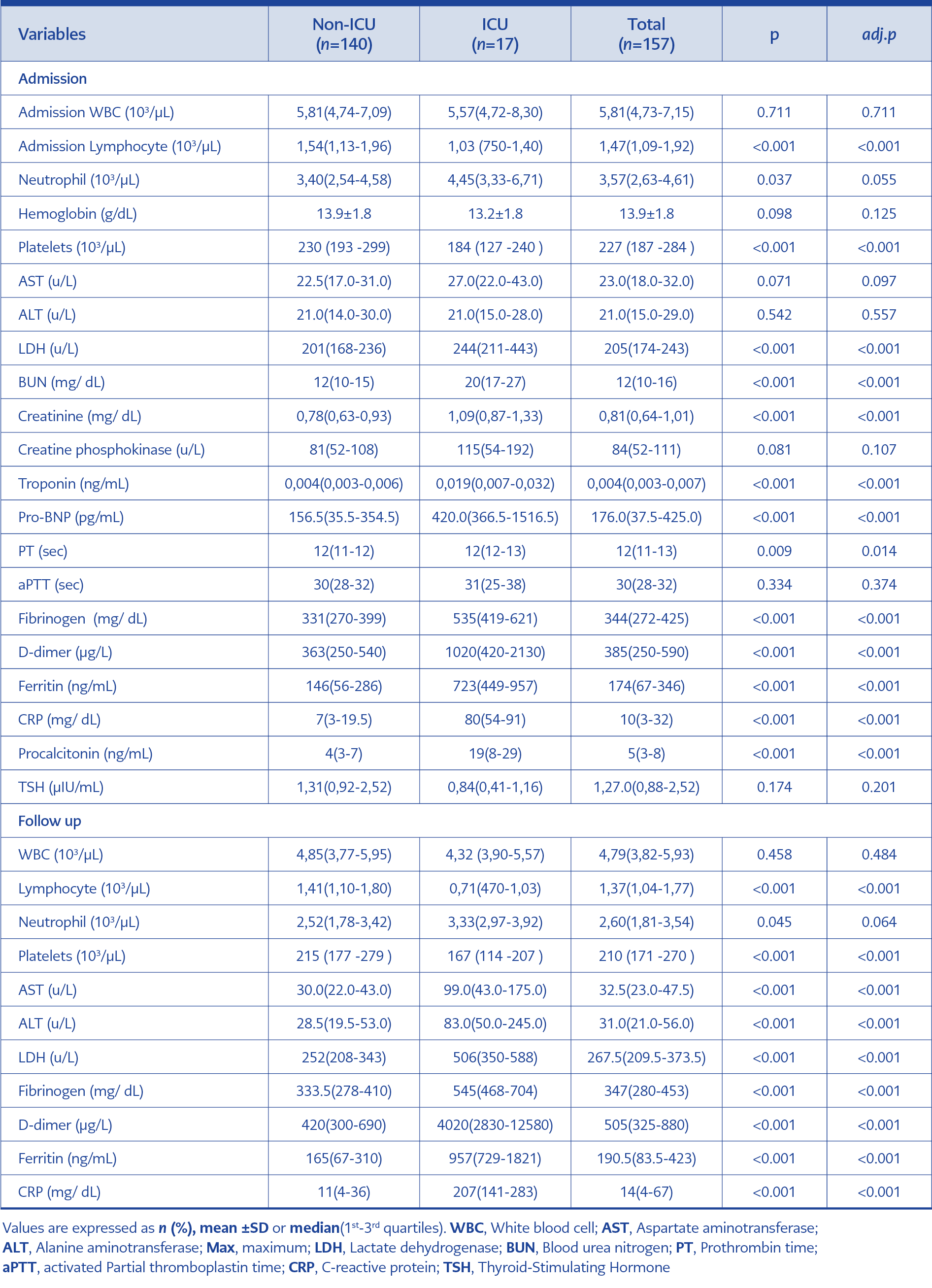
A comparison between admission and follow-up laboratory findings of ICU and non-ICU groups are shown in Table 2. A decrease was observed in leukocyte, neutrophil, lymphocyte, and platelet levels during treatment. Also, lymphocyte and thrombocyte levels were lower in ICU patients than those in clinics. The differences between the two groups were found statistically significant (p <0.001). Median aspartate aminotransferase (AST), alanine aminotransferase (ALT) and lactate dehydrogenase (LDH) levels were increased during clinical follow-up and treatment. Median AST, ALT, blood urea nitrogen (BUN), creatinine and LDH levels of ICU patients were significantly higher than those followed in the clinics (p <0.001). Median troponin and pro-brain natriuretic peptide (pro-BNP) levels of ICU patients were significantly higher than non-ICU patients (p <0.001). An increase in fibrinogen, D-dimer, Ferritin, and CRP levels from acute phase reactants was observed during clinical follow-up. Besides, the median level of acute-phase reactants in patients followed up at the ICU was higher both on admission and during treatment (p <0.001).
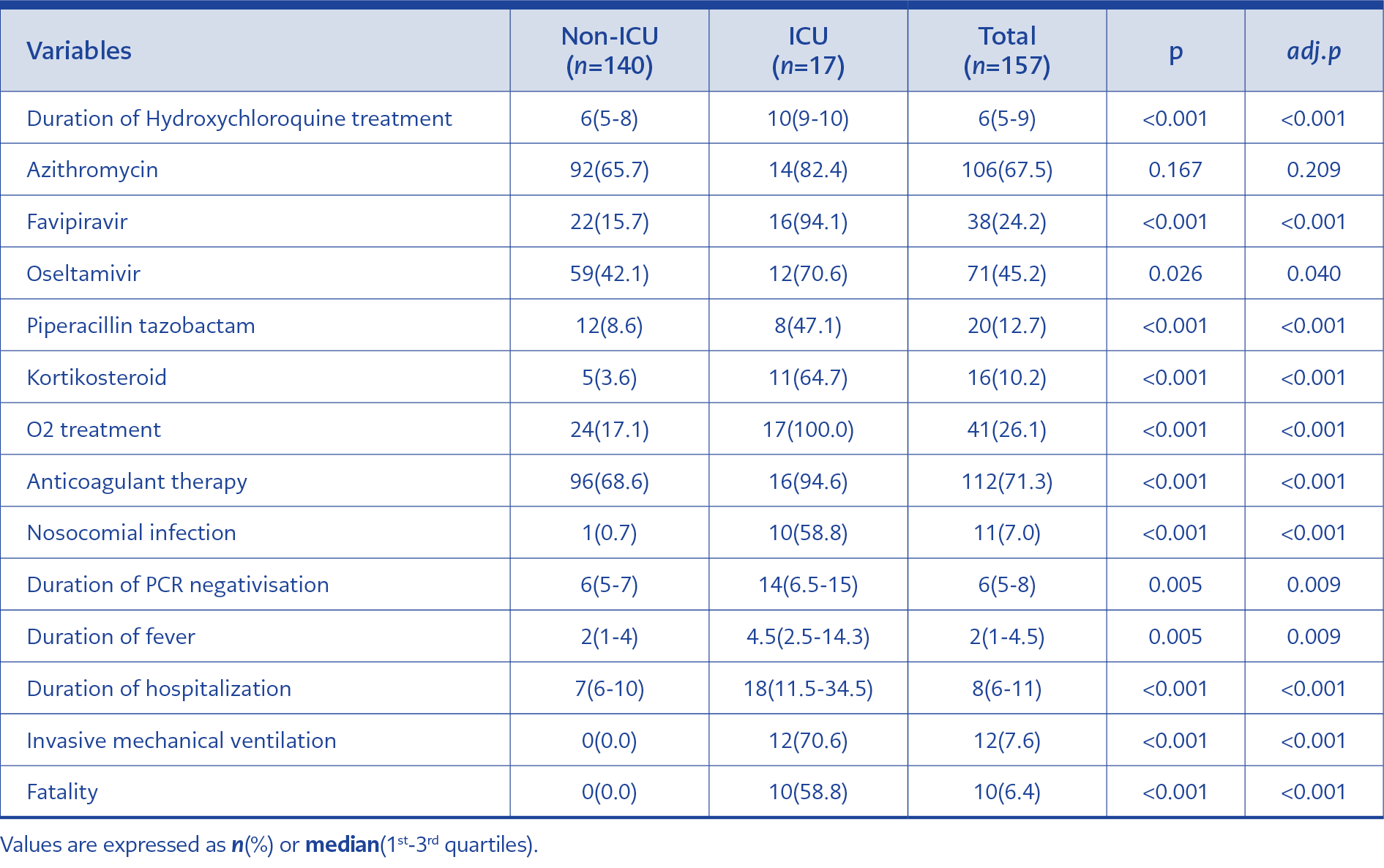
Treatment and prognosis related variables between ICU and non-ICU groups are shown in Table 3. Hydroxychloroquine treatment was initiated to all patients, and the treatment duration was longer in patients in the ICU (p <0.001). Azithromycin treatment was started to 67% of the patients with hydroxychloroquine. Favipravir treatment was initiated in 94% of ICU patients, while 15% of the non-ICU patients (p <0.001). Oseltamivir and piperacillin-tazobactam treatment rates were also significantly higher in ICU patients than in non-ICU patients (p = 0.04 and p <0.001, respectively). During the treatment, 10% of the patients needed corticosteroids, and this rate was higher in ICU patients (65% vs. 5%; p <0.001). While 17% of the patients in the pandemic clinics needed O2 support, all ICU patients needed O2 support (p <0.001). A nosocomial infection developed in 58% of ICU patients. Anticoagulant treatment was started in 112 patients according to risk factors. ICU patients had a higher treatment rate of anticoagulant prophylaxis (95%; p <0.001). Invasive mechanical ventilation was needed in 12 (71%) ICU patients, and nine died. The intensive care fatality rate was 59%, while the total fatality rate was 6%.
Discussion
COVID-19 has a wide range of clinical presentations, from mild disease to death. In this study, we aimed to evaluate the risk factors of ICU admission. There are advantages and disadvantages of treating patients in the ICU. In the early period, close monitoring and timely mechanical ventilation support in ICU decreases mortality (8). However, invasive procedures and long-term ICU hospitalizations may cause collateral damages and nosocomial infections (9). Thus, it is important for each center to know its patient population and determine clinical monitoring and treatment protocols.
In this study, the mean age of the patients was found to be 47 ±16.46. All patients admitted to the ICU were over 50 years old, and 70% were over 65. A study in which 13 clinical studies and meta-analysis of 3027 patients were performed reported that being over 65 years old increased the risk of disease progression six times (10). Italy, where the number of cases and mortality is high, reported that the mortality rate was 21% higher in patients aged 64 and over (11). As with many viral infections, typical disease symptoms could be masked with older age in COVID-19. Fever response could be suppressed, and atypical presentations could be observed (12). Such disadvantages caused difficulties in diagnosis and early treatment.
The frequency of comorbid diseases increases with age; drugs used in treatment can suppress the disease symptoms and poorly affect the prognosis (13). In our study, 36% of patients had at least one comorbid disease, but this rate increased to 76% in ICU patients. A retrospective study that examined the relationship between disease prognosis and comorbidity, and included 1530 confirmed COVID-19 cases reported that 25% of patients had at least one comorbid disease (14). In this study, it was reported that the presence of two comorbid diseases increased bad prognoses such as admission to intensive care, invasive mechanical ventilation, and mortality 2.59 times.
In our study, the presence of HT and DM was significantly higher in ICU patients. In a meta-analysis involving 21 retrospective clinical studies, the presence of DM and HT was reported to be closely related to severity and mortality (15). A prospective study, including 193 confirmed COVID-19 patients, 48 of whom were diabetic, compared diabetic and non-diabetic patients. As a result, a statistically significant increase was observed in diabetic patients’ acute phase reactants such as leukocyte count, CRP, procalcitonin, and proinflammatory cytokine levels such as IL-2, IL-6, IL-8 and TNF alpha (16). In a meta-analysis in which six clinical studies and 1527 patients were compiled, the frequency of DM and HT was reported to be two-fold higher in the severe patient group in need of intensive care (17). In patients with uncontrolled diabetes, the cellular immune system, the first line of defense against COVID-19 was damaged (13). In hypertensive patients, it was suggested that after binding of the COVID-19 virus to the ACE-2 receptor, it might cause an increase in angiotensin 2 level and an increased risk of adverse drug reactions (18). Although physiopathology could not be clearly explained for both comorbid factors, it was obvious that it posed a risk for severity and mortality. It was important to monitor these patients more closely.
Of the patients, 6% had a history of traveling abroad. 80% of these patients who had been to Saudi Arabia for worship were elderly and had comorbidities. Their advanced age, close contact during worship, accommodation, and travel increased the risk of viral spread. Therefore, the treatment rate and mortality were higher in these patients. All of these cases were treated at the beginning of the pandemic. Following the travel restriction measure, the number of such patients decreased both in our hospital and country. Modeling studies on this issue concluded that travel restrictions were significant in preventing disease spread (19, 20).
When comparing laboratory parameters on admission and clinical follow-up, lymphocyte and platelet counts were lower, and LDH, ferritin, fibrinogen, D-dimer, CRP, and procalcitonin values were higher in ICU patients. Among the biochemical parameters, AST, ALT and kidney function tests increased during clinical follow-up; the increase was higher in ICU patients. A meta-analysis included 21 studies, 3377 patients and 33 laboratory parameters compared laboratory values in patients with severe and mortal courses. Patients with the severe and fatal disease had significantly increased white blood cell (WBC) count, and decreased lymphocyte and platelet counts compared to non-severe patients and survivors. Biomarkers of inflammation, cardiac and muscle injury, liver and kidney function, and coagulation measures were also significantly elevated in patients with severe and fatal COVID-19 (21). A study reported from China prospectively followed 179 patients; while WBC and lymphocyte levels were lower in the severe patient group, CRP, procalcitonin, creatinine, and D-dimer levels were higher (22). Another study in which 191 patients were retrospectively analyzed reported that the level of D-dimer was associated with mortality, and mortality was 28 times higher in patients with a D-dimer level of above 1 μg/mL (23). Cytokines released with increasing ACE-2 suppression in the severely ill group cause microangiopathies with oxidative stress and endothelial dysfunction. Microangiopathies tended to have thrombosis and vasculitis-like findings. Myocardial damage occurred when the virus was attached to the myocardial ACE-2 receptor. As a result of both conditions, the risk of myocardial infarction increased (23).
For this reason, cardiac biomarkers could be elevated. In a meta-analysis including 14 clinical studies and 4659 patient data, pro-BNP and troponin levels were reported to be significantly higher in the deceased patient group (24). In our study, troponin and pro-BNP levels were higher in ICU patients. Parameters such as troponin and D-dimer might also be guided in terms of anticoagulant treatments to prevent thrombotic complications. In this study, considering the patients’ comorbid factors and laboratory values, almost all ICU patients were given anticoagulant therapy.
In the current study, there is no data about the viral load. Thus, longer positivity of the virus in ICU patients could not be related to viral load. It was known that sensitivity in lower respiratory tract samples was higher than in nasopharyngeal samples (25). PCR positivity was confirmed in all patients included. However, when periodic samples were taken, the PCR positive period was longer in ICU patients. Two important factors could be effective in this situation. Firstly, the rate of taking lower respiratory tract samples was higher in ICU patients who needed mechanical ventilation. The second important factor was viral replication and higher viral load. In an observational study examining the dynamics of RT-PCR findings of 301 patients, the duration of PCR positivity was higher in patients over 65 (26). The fact that the patients treated at the ICU were old can be considered as another factor. Nosocomial infections caused by excessive viral load and mechanical ventilator and other invasive procedures in ICU patients caused prolonged fever duration (27). For this reason, antipseudomonal antibiotic treatment rates, such as piperacillin-tazobactam, were also found higher in this patient group.
The fatality rate in 157 patients included in this study was 6%. The fatality rates reported in the literature ranged from 1% to 32.5% (28, 29). It is expected that the fatality rate will decrease both locally and globally by clarifying the physiopathology of the disease, knowing the risk factors, developing new drugs, and supportive treatments.
Conclusion
The patients with a confirmed diagnosis of COVID-19 who needed ICU had advanced age and more comorbidities. Lymphopenia, thrombocytopenia, liver function test disorder, and high D-dimer, CRP, and troponin levels may be significant for the severity of the disease. Duration of the fever, hospitalization, and duration to negative reverse-transcriptase PCR tests were longer and, mortality rates were higher in ICU patients. Therefore, patients with risk factors as demographic, clinical, and laboratory findings in terms of poor prognosis should be followed up more closely.