Introduction
Invasive pneumococcal disease (IPD) caused by Streptococcus pneumoniae is an important cause of morbidity and mortality among young children and adults 65 years and older. S. pneumoniae is one of the most important causes of serious diseases, such as pneumonia, meningitis, and bacteremia (1). According to 2018 surveillance data from US Centers for Disease Control and Prevention (CDC), the incidence of IPD in the general population was 9.6/100,000, while the mortality rate was 1.06/100,000. The incidence rate of IPD (24/100,000) and the mortality rate were also much higher in people over 65 years than the general population (2). The IPD surveillance systems in Turkey and Middle East Region are largely inadequate, resulting in a lack of knowledge of the true incidence of IPD. Overall, studies generally observed a high burden of IPD among adults in this region (3).
Besides advanced age, the most common IPD risk factors include asplenia or splenic dysfunction, chronic lung diseases, chronic kidney diseases, chronic heart diseases, chronic liver diseases, and immunocompromised status (malignancies, acquired immune deficiency syndrome, receiving immunosuppressive therapy, solid organ transplantation.) (4). A nursing home residence is also an important risk factor for IPD and IPD-related short-term mortality (5).
Nasopharyngeal (NP) pneumococcal carriage is often asymptomatic, but also it is the first and most important stage in the disease’s pathogenesis. NP carriage varies between 20% and 50% in children and 2%-30% in healthy adults (6, 7). As the childhood pneumococcal vaccination effort has expanded, vaccine serotype pneumococcal carriage and disease have decreased in children and unvaccinated adults. The serotype distribution has also changed. In particular, the carriage prevalence of non-pneumococcal conjugate vaccine 13 (PCV13) serotypes has increased (8, 9). The epidemiology of pneumococcal disease and carriage should be followed to monitor the effectiveness of current pneumococcal vaccination programs. The World Health Organization (WHO) Pneumococcal Carriage Working Group recommends conducting NP carriage studies regularly (10).
In Turkey, the pneumococcal conjugate vaccine 7 (PCV7), which includes serotypes 4, 6B, 9V, 14, 18C, 19F, and 23F, was introduced in 2008 for immunization of children. In 2011 PCV7 was replaced by the PVC13, which includes serotypes 1, 3, 5, 6A, 7F, and 19A additionally, with the universal access as 3+1 schedule, to be given at 2,4, and 6 months with a booster dose provided at 12 months of age. Since 2016, the sequential use of PCV13 and pneumococcal polysaccharide vaccine (PPSV23) (containing serotypes 1, 2, 3, 4, 5, 6B, 7F, 8, 9N, 9V, 10A, 11A, 12F, 14, 15B, 17F, 18C, 19A, 19F, 20, 22F, 23F, and 33F) is recommended for adults ≥65 years old and 19-64 years old with IPD risk factors by the General Directorate of Public Health (11). IPD surveillance system in Turkey has recently been established, but there is no published information about the circulating pneumococcal serotype yet. There is a lack of data in the literature about pneumococcal carriage and serotype distribution in adults from Turkey.
In this study, we aimed to determine the prevalence of overall pneumococcal nasopharyngeal carriage and risk factors for carriage; the prevalence of PCV13 serotype carriage; and serotype distribution among adults ≥ 65 years of age who live in a nursing home in İzmir, Turkey.
Materials and Methods
Study Design
The research was planned as a cross-sectional study. The study was conducted at the Narlıdere Nursing Home Elderly Care and Rehabilitation Center, İzmir/Turkey. İzmir is the third most populous city of Turkey and located in western Turkey. Of the population, 9.6% is ≥ 65 years old in Turkey, 17.1% in İzmir (12). The nursing home is located in Narlıdere, western part of İzmir, and most of the residents are people with a high level of education, good socioeconomic status, low comorbid diseases, and high physical activity.
Inclusion criteria were current residence in the nursing home, age ≥ 65 years, and willingness to participate. We visited the facility on weekdays between March 9 and March 31, 2017. The residents were called from their rooms and informed about the study, and those who agreed to participate in the study were invited to the medical unit of the nursing home for sampling. Informed written consent was obtained from all participants. We interviewed all participants to obtain their sociodemographic and medical history data. This information was recorded using a standard questionnaire.
Sampling and Isolation
One NP sample was obtained using a swab with a flexible aluminum shaft for each adult. The samples were transported to the laboratory using Stuart transport medium and processed within 8 hours. The samples were inoculated on BD Columbia agar with 5% sheep blood and incubated for 18–24 h at 35–37ºC with 5% CO2 (in a candle jar). On a blood agar plate (BAP), colonies that were small, grey, and moist (sometimes mucoidal) with a zone of alpha-hemolysis (the colonies become flattened, and the central portion becomes depressed as the culture plate ages) were considered to be S. pneumoniae and were examined for further identification. The catalase test was applied to colonies showing pneumococcal morphology. Catalase-negative colonies were examined using Gram staining. Colonies with Gram-positive diplococci were inoculated on a BAP and incubated under the same conditions for purification. The optochin susceptibility test was used for S. pneumoniae identification with bile solubility as a confirmatory test. The S. pneumoniae ATCC 49619 strain was used as a positive control.
Capsular Typing
Capsule serotypes were determined by co-agglutination and capsule swelling (Quellung) reactions, which are accepted by the WHO as the reference methods (10). We used pneumotest plus (Serum Statens Institute, Denmark) containing capsule antisera against the serotypes included in PPSV23, which are known as the most common cause of IPD (13).
1. Bacterial suspension preparation: We used a sterile loop to prepare a light to moderate cell suspension (approximately equal to a 0.5 McFarland density standard) from overnight growth on BAP (18–24 h at 35–37°C in a candle jar) in 300 µl of phosphate-buffered saline (PBS).
2. Antisera preparation: A homogeneous suspension was prepared by mixing 100 μl of sterile PBS and 100 μl of antiserum. The mixture was stored at +4ºC and used for one week.
3. Application of a capsule-swelling reaction: The slides were wiped with 70% ethyl alcohol. The patient number, negative control, and serum codes were written on the slide. Then, 3 μl of 1% methylene blue, 3 μl of antiserum, and 3 μl of prepared bacterial suspension were added. As a negative control, the mixture prepared with 3 μl of PBS was used instead of the bacterial suspension. Both the test mix and the negative control were placed on the same slide during each test.
4. The test mixture and the negative control were covered with a coverslip, and the capsule-swelling reaction was evaluated with an x100 objective (x100 magnification) after 5 min.
5. We began testing with pooled antisera. Once a positive reaction was obtained, we proceeded with the individual group and serotype-specific antisera included in the pooled antisera that gave the positive reaction to determine the serogroup and serotype, according to the manufacturer’s chessboard system (13).
Ethical approval was obtained from the Noninvasive Research Ethics Committee of Dokuz Eylül University as Decision No. 2016/31–31. Permissions were obtained from the Narlıdere Nursing Home Elderly Care and Rehabilitation Center Directorate and the Republic of Turkey Ministry of Family, Labor, and Social Services.
Statistical Analysis
The statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) 22.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics (the median and interquartile range or percentages, as appropriate) were used to summarize each sociodemographic and clinical variable included in the questionnaire. A Mann-Whitney U test was used to compare the continuous variables. The carriage was evaluated in terms of categorical variables using a chi-square test or a 2-sided Fisher’s exact test when appropriate. A multivariate logistic model was applied to investigate demographic characteristics (age and sex) and clinical features (presence of comorbidities and other factors) associated with pneumococcal carriage. Statistical significance was set at p<0.05.
Results
Characteristics of the Study Population
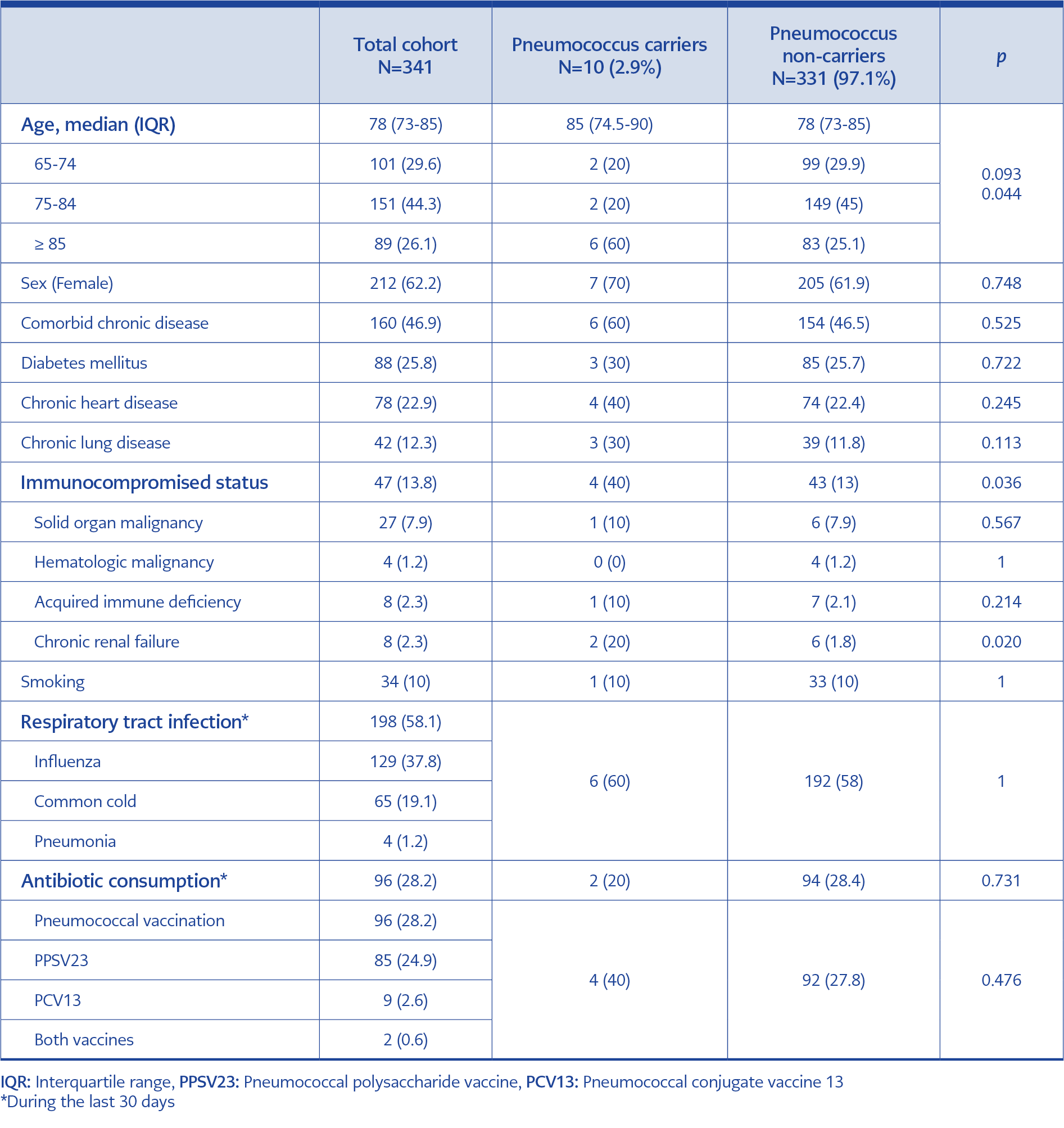
Table 1. Demographic characteristics and clinical features of total cohort, pneumococcus carriers and pneumococcus non-carriers.
In all, 341 (42.8%) of the 797 nursing home residents agreed to participate in the study. The median age of the participants was 78 (range: 65–97) years. Most of the participants were female (62.2%). In the group that did not participate in the study, the median age was 76 years, with 59.6% of females. Regarding medical history, 160 (46.9%) of the participants had at least one chronic disease, and 44 (12.9%) were immunocompromised. Smoking was uncommon, with only 10% of the participants being current smokers. Detailed demographic characteristics, comorbidities, respiratory tract infection, antibiotic consumption, and pneumococcal vaccination history of the participants are shown in Table 1. Of the participants, 24.9% had only PPSV23, 2.6% had only PCV13, and 0.6% had both vaccines.
Pneumococcal Carriage
Of the 341 elderly participants, 10 (2.9%) had a pneumococcal carriage. The median age of those with pneumococcal carriage was 85 (Range between 71 and 91 years old). Pneumococcal carriage increased with age and was found in 6.7% of those aged 85 years and over. Pneumococcal carriage prevalence was significantly higher in ≥ 85 years old participants than those between 65-74 and 75-84 years old (p=0.044). Having immunocompromised status and chronic renal failure were also associated with pneumococcal carriage (p=0.036, p=0.020). In multivariate regression model, having immunocompromised status was significantly associated with carriage (OR=4.7, 95 % Cl:1.25-17.95, p=0.022). The relationship between demographic characteristics, clinical features, and pneumococcal carriage is shown in Table 1.
Capsular Serotypes
Serotypes 7F, 23F, and 20 were isolated from three individual persons. Serogroups 10, 11, 15, 17, and 33, which are not included in PCV13, were isolated from other individuals, and their serotypes could not be determined because we did not have the factor sera required to determine their serotypes. The serotypes of pneumococci isolated from two people could not be determined using the Pneumotest kit. These strains were non-vaccine serotypes or unencapsulated pneumococci.
Of the determined carriage serotypes, 20% were included in PCV13 (serotypes 7F and 23F). PCV13 serotype coverage prevalence was 0.6% (2/341). Serotypes 7F, 20, 23F, and serogroups 10, 11, 15, 17, and 33 are included in PPSV23. However, because the serotypes of these strains could not be determined, clear information about the coverage of PPSV23 could not be given. None of the carriers had a history of PCV13, and four were vaccinated with PPSV23.
Discussion
This is the largest study investigating pneumococcal carriage in the elderly living in nursing homes in Turkey. Also, this is the first study to determine the serotype distribution of pneumococci colonizing adults in Turkey published in the literature. The pneumococcal carriage prevalence was 2.9% in this study. In another study from Turkey, Çetinkol et al. took NP samples from 92 nursing home residents older than 60 years old, and carriage prevalence was 16.3%. Although the carriage prevalence was higher than this study, the study had a small sample size and pneumococcal serotype distribution was not determined (14).
The pneumococcal carriage prevalence determined in this study was consistent with the studies from Europe and the USA conducted on people living in nursing homes. Flamaing et al. reported 4.1% of pneumococcal carriage among 296 people residing in a nursing home in Belgium (15). In Portugal, between 2010 and 2012, Almeida et al. found that pneumococcal carriage was found in 2.3% of 3361 people aged 60 and over. While the carriage prevalence was 1.9% in those living at home, it was significantly higher (5.7%) in those living in a nursing home (16). Becker-Dreps et al. took samples from 210 people from two nursing homes between December 2013 and April 2014 in the USA and reported a 2.9% pneumococcal carriage (17). Drayß et al. have tested 677 asymptomatic adults living at home or nursing homes, aged 65 years or more. S. pneumoniae was detected in none of the participants (18).
In this study, the pneumococcal carriage prevalence was found to be increased with age and was significantly higher in those aged 85 years and older. In the study by Ansaldi et al., carriage prevalence was 24.4% in those between 70 and 79 years old, and it was statistically higher than those aged 60–69 and ≥80 years (19). In the study by van Deursen et al., the pneumococcal carriage prevalence was 13.6% in those aged 75 years, less than half of the carriage prevalence (29.2%) of people aged 65–74 years (20). Esposito et al. reported that the carriage prevalence was higher (11.4%) in individuals <75 years old compared with those aged ≥75 years old (7.6%) (21). The reason for the different results in the relationship between age and pneumococcal carriage in the studies is thought to be the factors like the number of people living together, underlying chronic disease, physical activity, socioeconomic status, etc.
In this study, having a comorbid chronic disease, chronic lung disease, chronic heart disease, or diabetes mellitus (DM) was not detected as a statistically significant risk factor for pneumococcal carriage. Almedia et al. reported that having a chronic disease was not associated with a pneumococcal carriage. Chronic obstructive pulmonary disease and asthma were associated with pneumococcal colonization, whereas heart disease and, DM were not (16). In our study, immunocompromised status was a statistically significant risk factor for pneumococcal carriage. We could not find any study in the literature showing the relationship between immunocompromised conditions and pneumococcal carriage.
The percentage of pneumococcal vaccination among participants included in this study was low. While one-fourth of the participants had only PPSV23, only 2.6% had only PCV13, and 0.6% had both vaccines. The pneumococcal carriage was detected in 4.2% of those who had the pneumococcal vaccine, and it was not statistically significant. In similar studies, the percentage of pneumococcal vaccination varied between 3.6% and 87.3%; the lowest vaccination rate was in Portugal, and the highest was in the USA. There was no significant association between pneumococcal vaccination and overall pneumococcal carriage (15-21).
As PCV13 vaccination in adults increases in Turkey, it is expected to cause shifts in the distribution of serotypes that cause colonization and disease. According to our study results, PCV13 covers 20% of the serotypes isolated. In similar studies from Europe, the coverage of PCV13 varied between 9.1% and 51.4% (16, 19, 20, 22, 23). In a recent study from Japan, 18.2% of the participants carried a pneumococcal serotype included in PCV13 (24). These studies have been conducted in various countries, at different time periods, with different age groups, and using different methods. Therefore, serotypes that cause NP colonization and the percentage of vaccine coverage varied between studies. Geographical characteristics, socioeconomic and genetic factors, and vaccination policies of countries affect pneumococcal carriage. Each country must establish a national and regional surveillance system to monitor pneumococcal serotypes.
The strengths of our study: We investigated NP pneumococcal carriage and risk factors in older people residing in nursing homes, with a higher number of participants than other existing studies from Turkey. Previous studies conducted in Turkey focused more on pneumococcal carriage and antibiotic resistance among strains. In this study, we determined the serotypes of isolated pneumococcal strains and the serotype coverage of PCV13 for NP carriage was low.
The study was conducted in İzmir Narlıdere Elderly Care and Rehabilitation Center. People living in this center are generally a group of people with a high level of education, good socioeconomic status, low comorbid diseases, and high physical activity. For this reason, the results of our study may not reflect the public. In their population-based study, Almeida et al. showed that pneumococcal carriage percentages were different among the participants living in rural vs urban living areas and retirement home vs family home residents (16). The participants enrolled in the study could induce selection bias because this population tends to have better physical activity and be less care dependent. However, there was no difference in demographic characteristics between the study participants and the other residents.
We used conventional culture and serotyping methods recommended by the WHO. Molecular methods to investigate NP pneumococcal carriage have been recently developed. Recent studies report that conventional culture methods underestimate pneumococcal carriage prevalence and molecular methods are superior to conventional methods in detecting pneumococcal carriage (19, 20, 22, 23, 24). Because of the lack of specific anti-sera for further serotyping in pneumotest plus kit, the serotypes of serogroup 10, 11, 15, 17, and 33 could not be determined and the exact vaccine coverage of PPSV23 could not be found. Another limitation of the study was using capsule swelling reaction for serotyping.
Although this study was conducted at a single center in a limited geographic setting, a high number of subjects were included. Results showed that pneumococcal colonization in older people is uncommon and that most of the isolated serotypes are not PCV13 serotypes. The monitoring of pneumococcal carriage and serotypes can offer useful information about the circulation of this pathogen among older people as well as the potential protective effect of pneumococcal vaccines.