Introduction
Visceral Leishmaniasis (VL) is a widespread infectious disease of the reticuloendothelial system which is mostly caused by Leishmania infantum (L. infantum) and L. donovani in the Old World. The disease is endemic in many parts of the world, and estimated cases of VL have reached 300,000 globally with a high rate of HIV co-infection, and over 20,000–40,000 deaths annually (1). Visceral leishmaniasis, also known as kala-azar, is a zoonotic infection; recent reports point to the zoonotic involvement of L. donovani and dogs act as the main animal reservoirs of the disease while the vectors are the female Phlebotomus (sandflies) (2).
Turkey is situated on the crossroads between continents and acts as a bridge for people crossing to Europe from Asian and African countries like Turkmenistan, Pakistan, Afghanistan, Sudan, Nigeria and Ethiopia where VL is endemic (3).
With a total of 14,587 Cutaneous Leishmaniasis (CL) and 207 VL cases reported between the years 2005-2014, Leishmaniasis is an important public health issue in Turkey (4). Several species of Phlebotomus are present in Turkey. Until now 22 species of Phlebotomus and Sergentomyia have been reported from the country (5), and some of these Phlebotomus are proven vectors of leishmaniasis ( e.g. Phlebotomus neglectus, P. papatasi, P. perfiliewi, P. sergenti, P. similis, and P. tobbi) (6).
The genetic exchange and hybridization have been demonstrated in trypanosomatid parasites. (7) Although asexual reproduction has been attributed to Leishmania spp., the genetic exchange has recently been demonstrated in laboratory studies (8) which was followed by other publications exploring mechanisms of hybridization in Leishmania spp. (9,10). Both by molecular findings and analysis, it has recently been demonstrated that Leishmania spp. can form hybrids and Leishmania spp. show a high genetic variation. These findings led to the discussion that the currently identified strains are invalid, and the taxonomy should be restructured (11).
The genetic exchange seen in Leishmania spp. has considerable effects on epidemiology, biology, pathology, diagnosis, and treatment of Leishmaniasis (12). Naturally occurring hybrid forms of Leishmania spp. were also published, but the focus of these studies was mainly the parasite and not the clinical propagation of the disease caused by the hybrid strain. (13,14)
In this study, we aimed to investigate the clinical propagation of the naturally occurring disease in the human and dog which are infected by the hybrid strain of L. infantum/donovani and to analyze the vector Phlebotomus spp. using molecular methods, for the first time in Turkey.
Materials and Methods
Study Population
The patient was a 27-year-old male security officer working in Manisa. He was living in Manisa in a 200 m2 house made of stones and sand bricks with a garden that has white mulberry and blackberry trees. It was observed that the family was using some part of the garden as living grounds and that they kept a four-year-old female dog at the same place. He was sleeping in the garden next to the dog in warm summer nights, without using a mosquito net (Figure 1).

Figure 1. The photo of the garden where the infection cycle was enabled. A: The stone wall where the phlebotomus were caught B: The dog’s open air residence under the mulberry tree C: The sofa where the host was using to sleep in warm summer nights.
Before his visit to a general practitioner, the patient had symptoms like fever, shivers, night sweating, fatigue, myalgia and weight loss (12 kg in 6 weeks) for six weeks. He was prescribed symptomatic medication for viral upper respiratory tract infection and was externalized. Without any relief after using the medication, the patient applied to a secondary level hospital and was prescribed antibiotics. His fever was still high even after the antibiotic regimen, so he was hospitalized in the same hospital and was given moxifloxacin and ceftriaxone empirically. The blood tests could not maintain any solid diagnosis, and the patient was transferred to Celal Bayar University Medical Faculty Hospital with the fever of unknown origin pre-diagnosis.
Laboratory tests for the infectious and non-infectious causes of fever, such as non-specific bacteriologic and mycobacteriology cultures, viral serologic tests, auto-immune markers, thyroid function tests were performed. Besides, radiologic tests, such as chest x-ray, abdominal ultrasound, abdominal and thoracal CT, echocardiography, were performed to detect the cause of fever of unknown origin. The patient having fever, splenomegaly, and pancytopenia was assessed for malaria using thin and thick blood smears.
Consultations were made from haematology and rheumatology departments because of splenomegaly and pancytopenia. Bone marrow samples were sent to the haematology department for possible malignant causes and parasitology department for parasitological examination.
Canine Leishmaniasis (CanL) Case History Analysis
The physical examination of the CanL case revealed conjunctivitis, blepharitis, weight loss, fatigue, loss of appetite, skin ulceration, onychogryphosis, epistaxis, alopecia, lymphadenopathy (LAP) and splenomegaly. Most of the symptoms were present for the last six months.
Sample Collection
Blood and bone marrow samples were obtained from the patient using intravenous catheterization and fine-needle aspiration, respectively. Five ml of blood was drawn from the dog via the brachial vein and serum was separated (15) followed by lymph node aspiration for the parasitological examination.
Sandflies were collected using Centers for Disease Control and Prevention (CDC) light traps. Light traps were set in the garden, the patient’s and dog’s living grounds as well as 100 meters radius around the house (Figure 2). The light traps were operated for three nights between 6.00 p.m. -11.00 a.m., and the sandflies were collected every morning between 09.00-11.00. Following the collection, they were morphologically identified and dissected. The female flies’ tissues were crushed for DNA isolation.
Direct Microscopy and Culture Methods
Bone marrow and lymph node aspirates were inspected for Leishmania spp. amastigotes under light microscopy (x1000 augmentation) from Giemsa stained slides after fixation with methyl alcohol. Furthermore, the samples were cultured in biphasic enriched Novy- MacNeal- Nicolle (NNN) medium containing saline solution, defibrinated rabbit blood and enriched agar using beef infusion, liver, and yeast extracts (16). The obtained mixture was then distributed to sterile tubes, each consisting of four millilitres of the mix. After keeping the tubes in room temperature until solidification, the tubes were transferred to +4 °C fridges. For the liquid compartment of the medium, 1 ml of RPMI-1640 medium containing 15% fetal calf serum was used. The media were kept in ovens at 25 °C and were inspected for the existence of promastigotes every day. Following detection, the liquid content of the media was transferred to flasks containing RPMI-1640 for the rapid production of promastigotes. The promastigotes were then collected from the RPMI-1640 medium and planted in a petri dish containing the solid phase of the EM to obtain a single colony for molecular purposes (17).
Serological and Molecular Analysis
The blood samples drawn from the patient were centrifuged to separate the serum. The patient sera were used for the Indirect Fluorescent Antibody Test (IFAT). An rK39 dipstick test was also performed for both the human and dog samples.
Real-time ITS-1 PCR
Primers and probes used for the real-time ITS1-PCR method were Old World species-specific (L. tropica, L. major, L. infantum, L. donovani). The procedure which was described previously by Töz et al. was followed (18). In short, the 25 µl final mixture for the real-time PCR analysis consisted of; 12,5 µl QuantiTect Probe PCR kit master (Qiagen), 10 Mm of each primer (forward primer 5′-CTGGATCATTT-TCCGATG, reverse primer 5′- GAAGCCAAGTCATCCATCGC -3′) 4,5 µl PCR Grade water, 4 μM of each probe and 20 ng genomic DNA. The protocol that was used in the Real-time PCR device (Rotor-Gene® Q) is; 95˚C forward denaturation for 15 minutes followed by 40 cycles (denaturation at 95˚C for 15 seconds, binding at 50˚C for 30 seconds and 20 seconds at 72˚C for elongation) and termination at 95˚C for one minute and 1 minute at 40˚C. Melting curve analysis was employed for species discrimination. Species identification was made by comparing the positive controls with the melting curve results of the specimens.
We used four Leishmania strains reference of The World Health Organisation for controls: L. major (MHOM/SU/1973/5ASKH), L. tropica (MHOM/AZ/1974/SAF-K27, MHOM/IL/1990/LRC-L590), L. donovani (MHOM/IN/80/DD8) and L. infantum (MHOM/TN/80/IPT1).
Cysteine Protease b (CPB) Gene PCR
A PCR of the CPB gene region that discerns differences between L. infantum and L. donovani was also performed as described previously by Hide and Bañuls (19). In brief, a real-time PCR was carried out using primers that were previously designed (unpublished data, provided as Supplementary Material) and PCR was run on Rotor-Gene® device (Qiagen, Germany) according to manufacturer’s instructions. The PCR amplification products are dyed with SYBR green nucleic acid dye and visualized under UV light following gel electrophoresis with 2% agarose gel.
Results
Patient Findings
The patient’s physical examination revealed; traube was closed, the right costovertebral angle was tender and splenomegaly was observed. His blood investigations showed pancytopenia with a leucocyte count of 2600/mm3, haemoglobin level of 9.7 g/dl and thrombocyte count of 62000/mm3. In the abdominal computed tomography (CT), the liver was noted as 18 cm and spleen as 18,5 cm with normal parenchymal echogenicity. There were several lymph nodes in para-aortic and para-iliac regions not exceeding one cm. Not being able to find the cause of the fever with the tests, empiric piperacillin-tazobactam was administered to him for pancytopenia.
Parasitological and Molecular Evaluation
The peripheric and thick blood smears were evaluated as negative for malaria. Leishmania spp. amastigotes were detected in the slides prepared from the bone marrow and lymph node aspirates while promastigotes were present in the EM. For patient serum, IFAT IgG was 1/256 positive, and rK39 was positive for both dog and human samples.
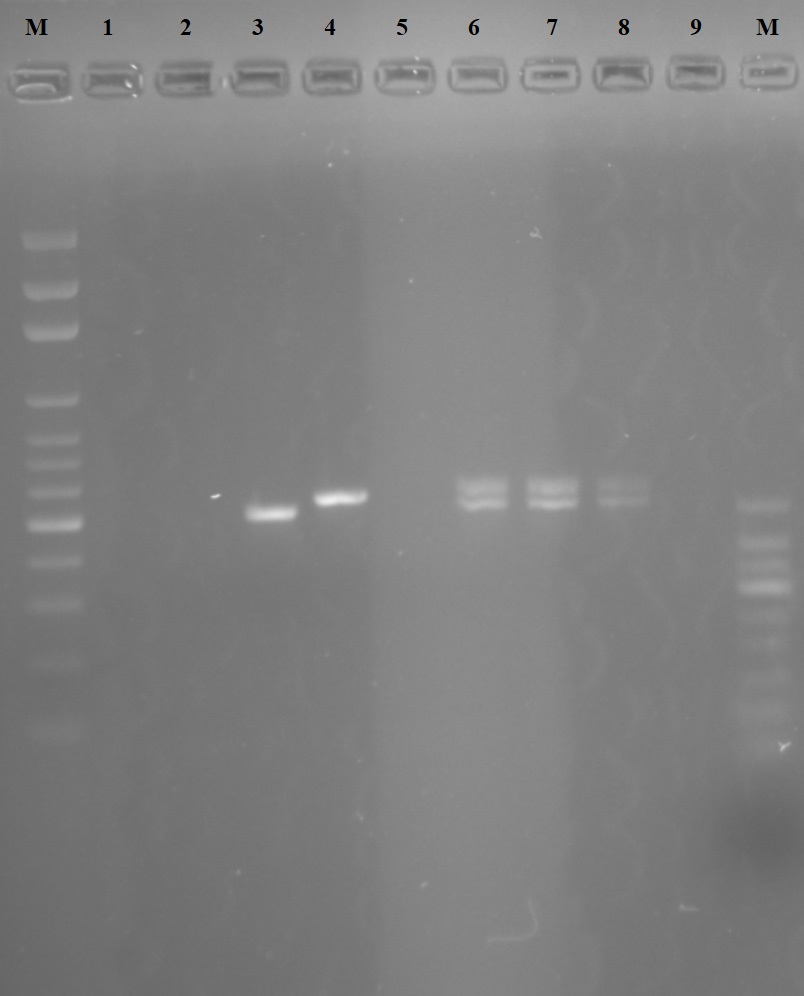
Figure 3. Gel image of the cpb gene region PCR. M. 100 bp Marker
1. Reference L. tropica (MHOM/AZ/1974/SAF-K27)<
2. Reference L. major (MHOM/SU/1973/5ASKH)
3. Reference L. infantum (MHOM/TN/1980/IPT1)
4. Reference L. donovani (MHOM/IN/1980/DD8)
5. Reference L. aethopica (MHOM/ET/1972/L100)
6. Sample 1 L. infantum/donovani hybrid (human)
7. Sample 2 L. infantum/donovani hybrid (dog)
8. Sample 3 L. infantum/donovani hybrid (phelobotomus)
9. Negative Control
10. 50 bp Marker”
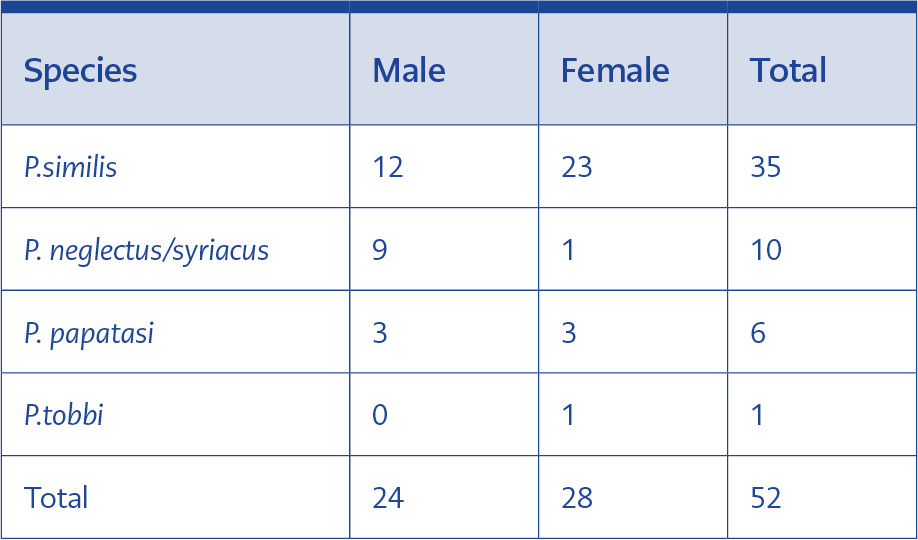
Of 52 Phlebotomus (24 male and 28 female), 35 were P.similis (12 male, 23 female), ten were P .neglectus/syriacus (nine male, one female), six were P.papatasi (three male, three female) and one was P. tobbi (female) (Table 1). Molecular analysis showed an identical Leishmania strain in both the patient, dog, and P.tobbi. Melting curve analysis for the Real-time PCR revealed two peaks in all three samples and gel electrophoresis revealed two bands both compatible with L. infantum and L. donovani regions in all three samples, thus leading to the conclusion that the infecting strain was a hybrid of L. infantum and L. donovani (Figure 3).
Treatment
After assessing the patient’s severe clinical situation besides the unpredictable treatment response of the hybrid strain against pentavalent antimonials because of the reported antimonial resistance in the region (20), Liposomal amphotericin B was chosen as the drug of choice. Evaluating the patient as young and without any serious organic functional disorder and considering the high cure rates by liposomal amphotericin B therapy (21) he was administered liposomal amphotericin B with the dosage of 3mg/kg/day for ten days with the total dose of 30 mg/kg.
After the liposomal amphotericin B therapy, the patient’s fever resolved, clinical symptoms regressed, and the blood tests returned to normal. He was externalized and was screened every 15 days for three months in the clinic. During these 3 months, his appetite was going better, and he started gaining weight, his liver and spleen returned to normal size, and his fever resolved. Because of the patient’s refusal of a control bone marrow sampling, his peripheric blood smear was controlled for Leishmania spp. amastigotes and was evaluated negatively.
As for the CanL case, the dog was evaluated by a veterinarian and assessed as severe canine leishmaniasis (CanL). The dog was subjected to euthanasia by its owner, considering the clinical severity of its situation and the environment suitable for the infection chain with abundant vector sand flies in the surroundings,. It was recommended that the garden should be sprayed with insecticides.
Discussion
The detection of the hybrid strain can be attributed to the developing molecular methods which enable us to differentiate Leishmania spp. through genomic and proteomic approaches. There is no possible way to show when or where the hybridization occurred with the present data, but there is good reason to suggest recent development of the L. infantum/donovani hybrid for various reasons. Firstly the hybrid strain was found in the Manisa region, which is in the western part of Turkey. Manisa is in the Mediterranean climatic zone and bears the rainfall characteristics of the Mediterranean climate. The rains are concentrated in winter while summers are dry. The warmest months are June, July, and August with temperatures rising above 30 °C, the average temperature in July is 34.4 °C and the average temperature all around the year is 16.8 °C. The minimum temperatures in winter do not go below 6 °C. It is identified that Manisa has 162 summer days and an average of 107.5 days in a year the temperature is above 30 °C. The city is situated on the foot of Spil mountain, and a big part of the Gediz river flows through Manisa, very close to the ancient Lydian Empire’s capital city Sardis (22).
Manisa city is an industrial and agricultural centre in the region, housing a population of 1 380 366 people in 2015 (23). It is also a neighbour city of Izmir, the 3rd most populous city in Turkey. Because of its location and job opportunities, Manisa attracts lots of immigration from all around Turkey (24). With the ongoing refugee crisis in the region, there are also lots of Syrian refugees immigrating to Manisa (25). Around 4 million registered Syrian refugees are now residing in Turkey. In Syria, 46 398 new CL cases were reported in 2009 and 42 165 in 2010, VL cases are sporadic (26,27). The Syrian CL patients are also crossing the border to Turkey and settling in the cities. These patients’ conditions are monitored by the Turkish Ministry of Health and documented. It is suspected that a large proportion of the refugees are CL patients and can easily carry the disease to Turkey (28).
With the rising population in the region and immigration from Leishmaniasis endemic regions, Leishmaniasis cases are increasing. This increase in the number of human and animal infections in the region coupled with the infections caused by different strains carried in by the human population movements may have generated a suitable environment for hybrid formation (29).
Interspecific hybrid formation among trypanosomatids has long been debated (30), but the mechanisms of hybridization have not been fully elucidated. Only recently, intraspecific hybridization has been demonstrated (7). Furthermore, the genetic diversity and structure of L. infantum populations and ways to discriminate them is under debate (31). Hybridization among different strains of Leishmania spp. may stand as a powerful tool for Leishmania genomic exchange and thus strain differentiation. In this study, we show a hybrid strain of L. donovani/L. infantum acquired from the sand fly P. tobbi midgut as well as the human and dog samples. These data strongly suggest that the L. infantum/donovani hybrid strain is zoonotic and causes VL in humans and CanL in dogs. As to where the hybrid formation occurred remains unanswered, the previous studies suggest the permissive vector P. tobbi may play a role in hybrid formation (8,32).
A study also shows a VL causing hybrid strain obtained from the Kuşadası region, which is also in western Turkey in the year 2001 (33). In that study, the hybrid strain was characterized by Multilocus Microsatellite Typing (MLMT) analysis, which is mostly used for population genetic studies. Also, in the same study, the hybridizing populations of Leishmania spp. were attributed to L. infantum MON-1, which is the prevalent zymodeme causing VL in Turkey and a non-MON-1 Turkish-Cypriotic population (33). No other study was published referring to a hybrid strain of Leishmania spp. from the region. Applying advanced population genetics and sequencing methods to those two hybrid strains may provide insights into the hybridization process. The only proven vector for L. infantum/donovani hybrid strain is P. tobbi.
Another issue that is open to debate is that this strain may not be a hybrid or a variant but an original species in itself. Recent literature shows increasing numbers L. infantum and L. donovani infections, and some of these cases are CL causing strains that are detected in Turkey (34). Further investigations of the exact molecular make-up of the genome of this L. infantum /donovani hybrid strain, as well as its pathological properties, needs to be carried out.
Conclusions
According to our findings, it is assumed that L. infantum and L. donovani strains have formed a hybrid strain and the resulting hybrid strain is capable of causing severe VL in humans and CanL in dogs, and the natural vector of this strain is P. tobbi. These findings are attributed to the mass population movements from Leishmania spp. endemic countries into our country. It is also assumed that hybrid Leishmania strains can pose a threat as emerging infectious agents for the region and our country and may also present itself with different clinical propagations and affect much larger populations.