Introduction
Fungal infections account for about 20% of all microbiological infections in critically ill patients (1). Candida species are the most common fungi in intensive care units (ICU) and mostly C. albicans (1-3). However, the rate of non-albicans Candida species such as C. tropicalis, C. krusei, C. parapsilosis, C. glabrata which are more resistant to antifungal therapy, has been increasing rapidly in recent studies (1, 2, 4, 5).
The rise of fungal infections in critically ill patients can be related to the presence of advanced age, prolonged length of ICU stay, glucocorticoid or immunosuppressive drug use, diabetes mellitus (DM), multiple invasive procedures, renal replacement therapy (RRT), and the wide use of invasive devices (central venous catheter [CVC]). In addition, many ICU patients are treated with prolonged antibiotic therapies for real or presumed bacterial infections, favouring the onset of fungal infections (2, 4-6).
Candida species are found in the normal flora of the mucous membranes of the skin, gastrointestinal tract (GIS), genitourinary system (GUS) and respiratory system in humans. Colonization is most common in the oesophagus and can become an invasive disease in 3-25% (1). In order to distinguish between colonization and infection, tissue biopsy is required under ideal conditions. However, since the procedure is invasive, it is suggested that the Candida score may also be used to estimate the presence of infection (2,4-6). This study aimed to evaluate the fungal strains cultivated from samples taken in a medical ICU and detect risk factors for invasive Candida infections.
Material and Method
Patients and Data Extraction
Patients admitted to the third level ICU of a university hospital between January 1, 2016, and December 1, 2018, were included in the study if at least one of their cultures was positive for fungal infection during ICU stay.
Routine surveillance culture screening is not performed in our ICU. Microbiological sampling is performed only in the presence of clinical or laboratory signs of infection. These signs include fever, clinical deterioration, elevated acute phase reactants, hemodynamic instability, presence of purulent secretions, redness/pus around any catheter entry area, and development of symptoms suggesting sepsis.
If patients with candiduria have an indwelling Foley catheter, repeat urine cultures are required after changing the catheter. Antifungal therapy is initiated in patients with positive urine cultures if clinical findings of urinary tract infection are present or repeat urine cultures are positive. Antifungal treatment is also administered in patients as an empirical/pre-emptive therapy if they have a high Candida score or are septic and have risk factors.
If a patient has a suspected catheter infection or a positive culture, the catheter is removed as soon as possible. The catheter tip is sent for microbiological examination if there is no contraindication to remove the catheter. Echocardiography, hepatobiliary ultrasonography and ocular examination are performed as soon as possible for all patients with candidemia.
In the case of a positive culture, the patient is evaluated by his doctor, an intensivist and the consulting infectious disease specialist to plan treatment in a consensus.
The patients’ data included in the study were obtained by examining the intensive care files and laboratory data via the hospital’s electronic database. Patient’s age, admission diagnoses, Charlson comorbidity index (CCI), ICU and hospitalization admission and exit dates, presence of a solid tumor or hematological cancer, radiotherapy or chemotherapy treatment, immunosuppressive therapy, steroid use before or during ICU, presence of central venous catheter (CVC) or urinary catheter, invasive mechanical ventilation (IMV) requirement, need for renal replacement therapy (RRT), total parenteral nutrition (TPN) administration, presence of abdominal surgery, APACHE II score, sequential organ failure assessment (SOFA) score, and Candida score were recorded. Moreover, the types of fungal strains, sampling site and presence of multifocal colonisation, results of antibiotic susceptibility testing and the antifungal agents administered and their adequacy were recorded to the database. In addition, C-reactive protein (CRP) and procalcitonin levels, body temperature and antibiotic therapy when samples were obtained were recorded in the pre-prepared patient registration form.
Microbiology
The samples taken from patients were plated on blood agar (Oxoid, UK), MacConkey agar (Oxoid, UK) for bacterial growth and incubated at 37oC for 24-48 hours in ambient air and chocolate agar at 37oC for 24-48 hours in CO2 air. In addition, sabauraud dextrose agar (Oxoid, UK) and potato dextrose agar (Oxoid, UK) were used for fungal growth, incubated both at room temperature and 37oC at least for five days.
Yeast identification was made with cornmeal agar with Tween® 80 (Oxoid, UK) for determining the Candida chlamydospore morphology and MALDI-TOF MS system (Bruker Daltonics, Germany).
Candida albicans ATCC 90028 and Candida parapsilosis ATCC 22019 were used as the quality control strains.
Antifungal susceptibility testing was performed by broth dilution method according to European Committee on Antimicrobial Susceptibility Testing (EUCAST) recommendation.
Statistical Analysis
The research data was evaluated through the Statistical Package for Social Sciences (SPSS) 22.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were presented as mean ± standard deviation, median (interquartile range [IQR]), frequency distribution and percentage. IQR are calculated by subtracting the 25th percentile from the 75th percentile. Pearson’s chi-square test and Fisher’s exact test were used to evaluating categorical variables. Visual (histogram and probability plots) and analytical methods (Kolmogorov-Smirnov test and Shapiro-Wilk test) were used to examine if the variables were distributed normally. Mann-Whitney U Test was used among the two independent groups for the variables that were not normally distributed, and the Kruskal Wallis test was used among the four independent groups. When a significant difference was detected, the Mann-Whitney U test with Bonferroni correction was used in the post-hoc binary comparisons for the source of the difference. If variables conformed to the normal distribution, we used Student’s T-test between two independent groups and one-way ANOVA between four independent groups for statistical significance. When there was a significant difference, Tukey’s HSD test was used in post-hoc binary comparisons.
Independent predictors in predicting candidemia development status and pre-emptive treatment status using possible factors determined in previous univariate analyses were analyzed using logistic regression analysis. In addition, we evaluated model fit by the Hosmer-Lemeshow test. Statistical significance was set as p <0.05.
Results
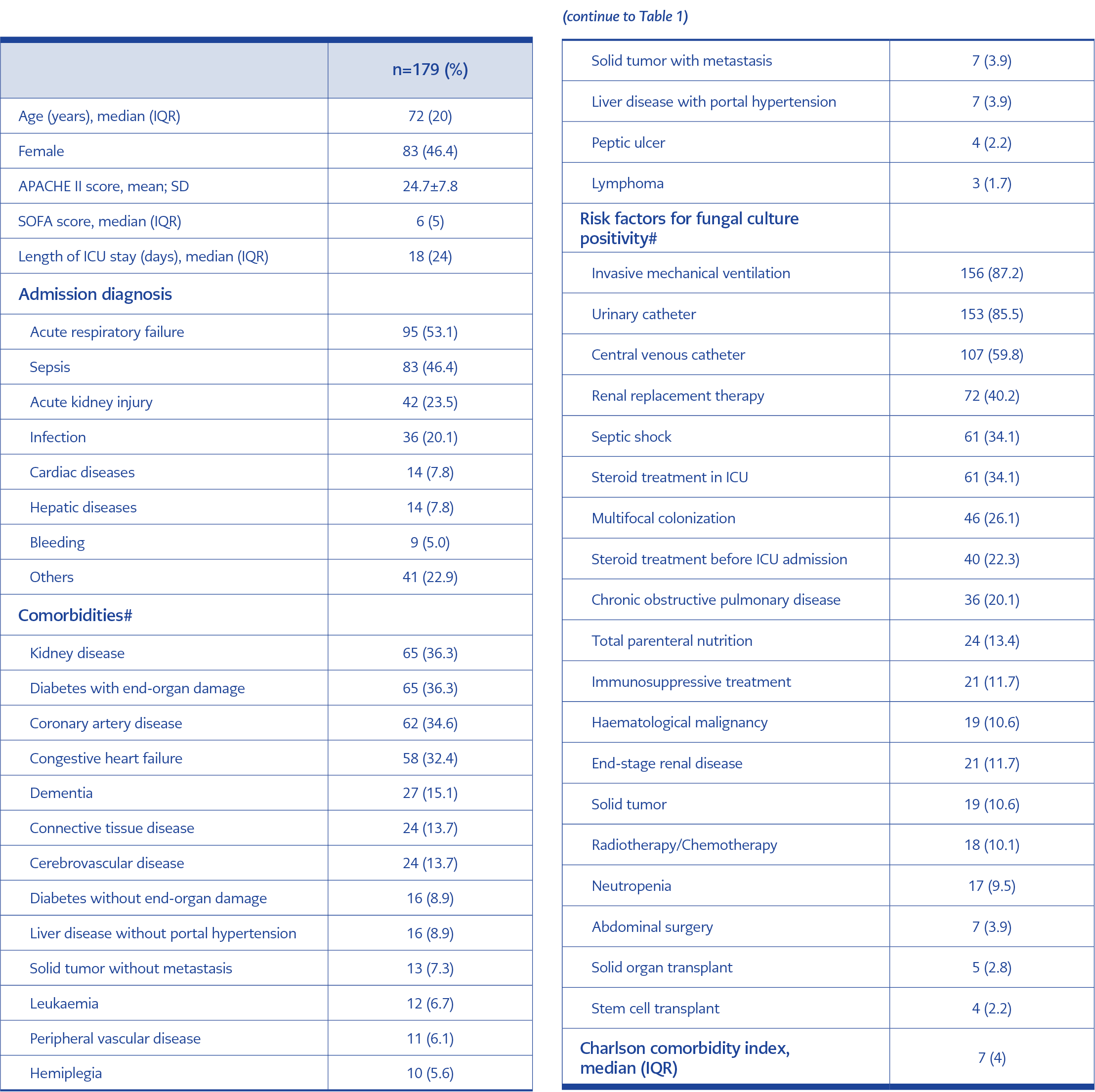
During the study period, a total of 384 fungal growths were reported for 179 patients. Samples were collected from urine, endotracheal aspirate, peripheral blood and blood drawn from a catheter, catheter tip, sputum, wound, abscess and cerebrospinal fluid. The median age was 72 years (IQR:20), and 46.4% were women. The mean APACHE II score was 24.7 ± 7.8, and the median SOFA score was 6 (IQR: 5). The median length of ICU stay was 18 (IQR: 24) days.
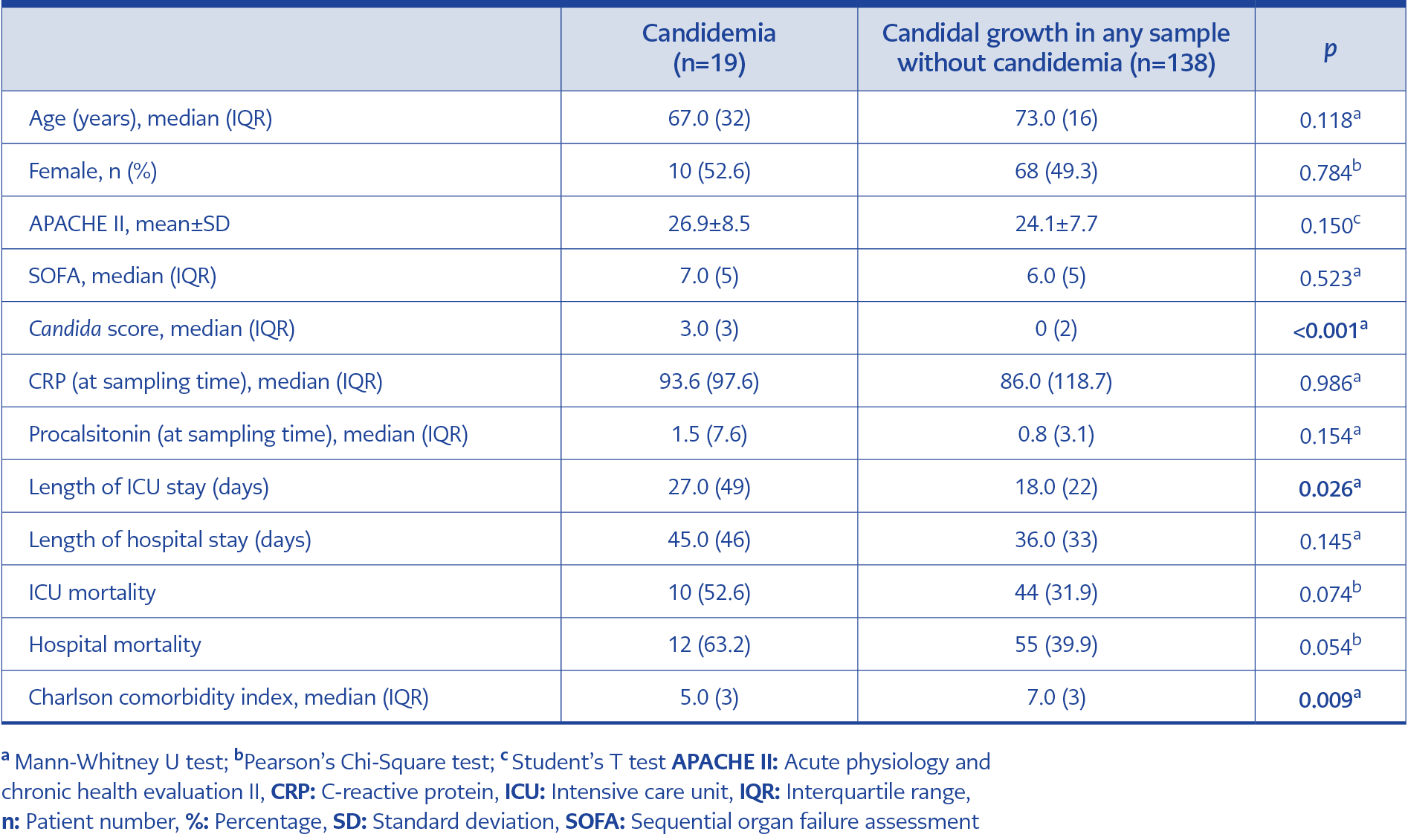
Table 4. Distribution of some descriptive and clinical features among patients with and without candidemia.
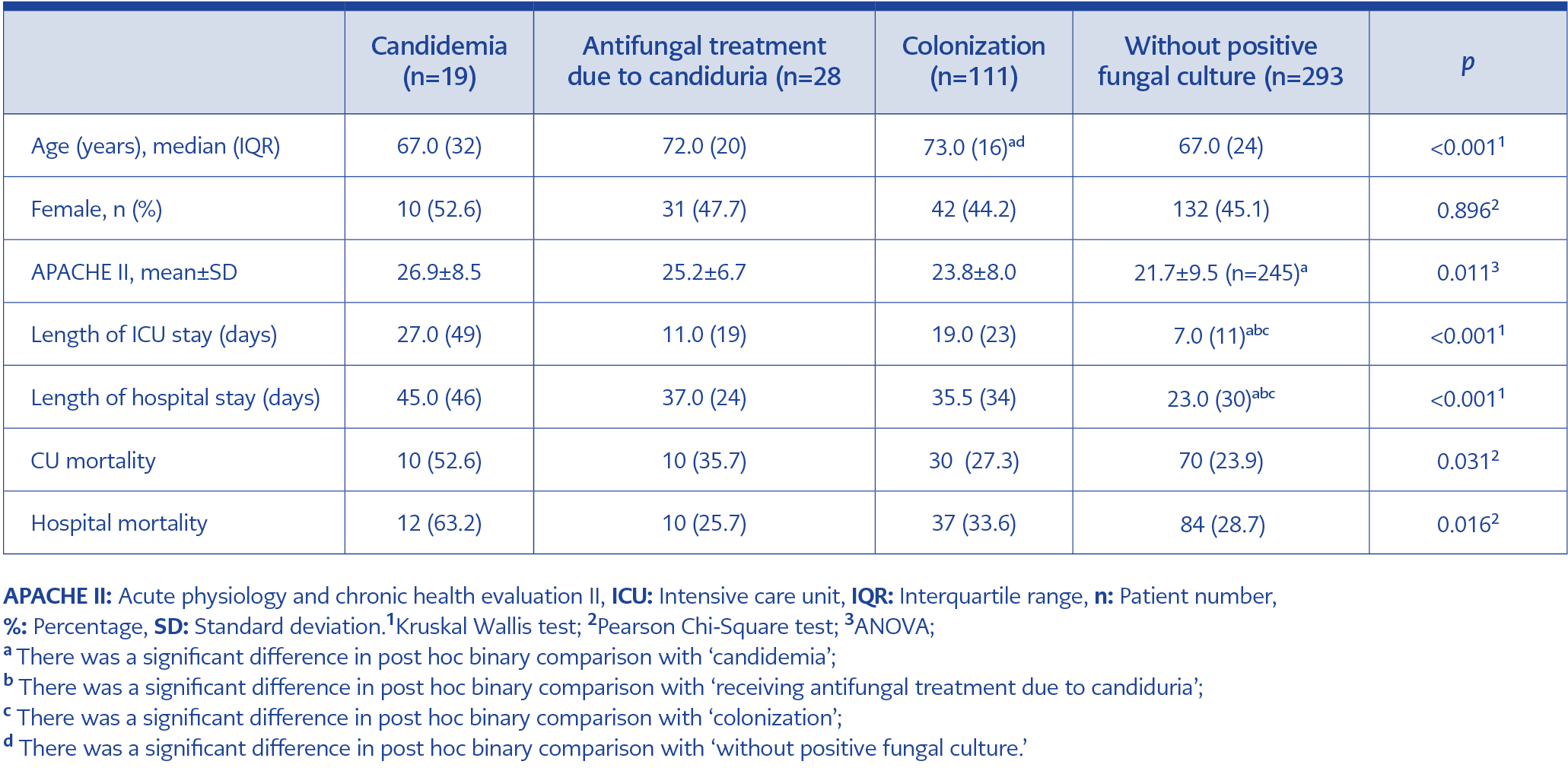
Table 5. Distribution of some descriptive and clinical features of patients with candidemia, candida urinary tract infection, candidal colonization and patients without any fungal growth.
Patients’ admission diagnoses included acute respiratory failure (53.1%), sepsis (46.4%), and acute kidney injury (23.5%). The most common comorbid disease was diabetes with end-organ damage (36.3%). It was followed by coronary artery disease (34.6%), congestive heart failure (32.4%) and dementia (15.1%). The median CCI was 7 (IQR: 4). As an infection risk factor, 87.2% of the patients were on invasive mechanical ventilation, 85.5% had an indwelling urinary catheter, and 59.8% had a central venous catheter (Table 1).
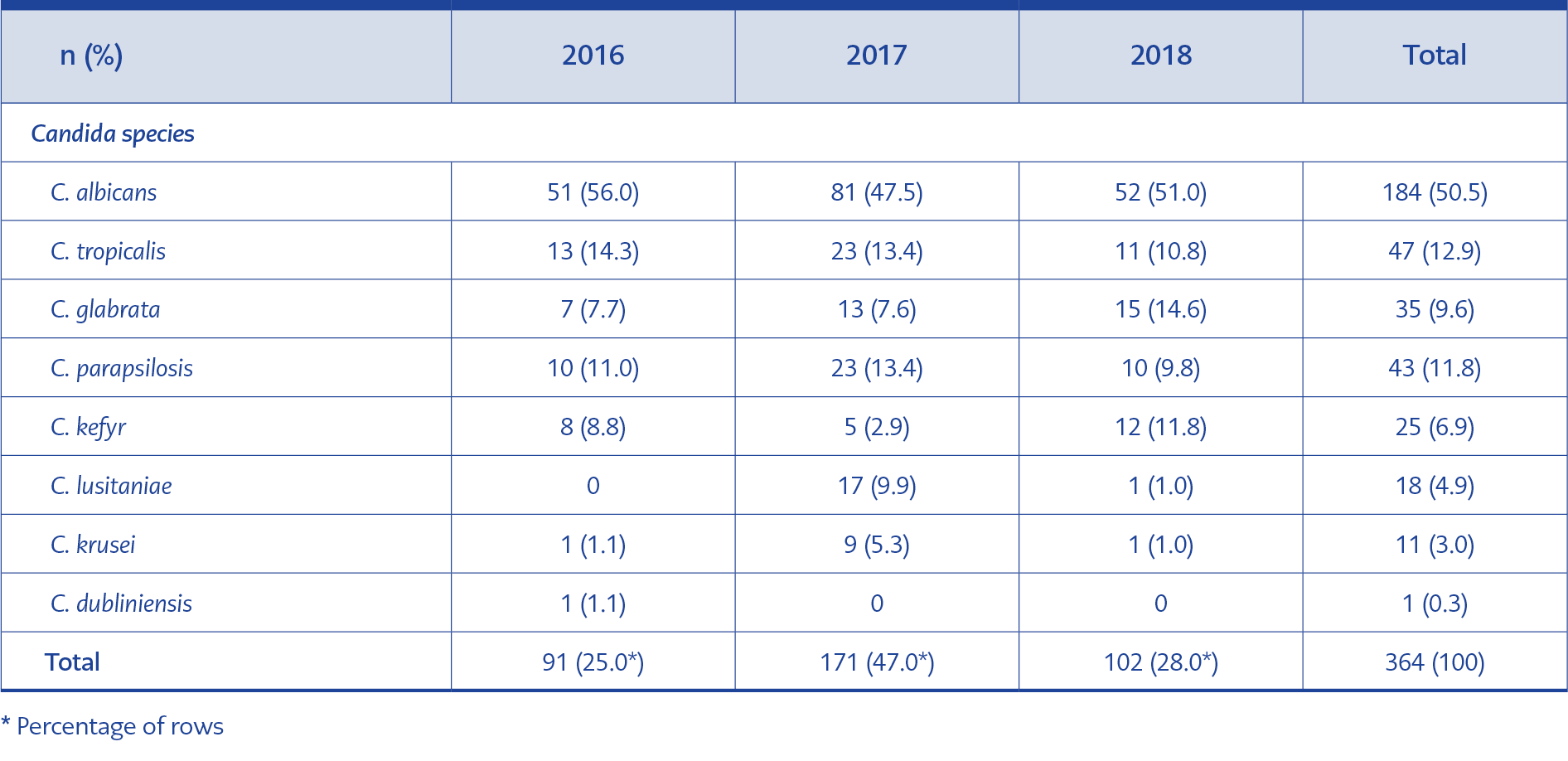
In 384 samples examined, C. albicans (184-47.9%) were the most common strain revealed. It was followed by other Candida species (46.8%) and Aspergillus species and, in several cases, Trichosporon, Saccharomyces, Fusarium and Rhizopus (5.3%). The frequency of Candida spp., which were the most common agents, by years is presented in Table 2.
Antibiotic susceptibility testing was performed in 170 (46.7%) of 364 Candida species. In these tests, apart from five C. krusei samples naturally resistant to fluconazole, fluconazole resistance was detected in 1 of 184 samples for C. albicans and 3 of 35 samples for C. glabrata. In addition, fluconazole susceptibility testing was performed in 14 of 19 patients with candidemia and no resistance to fluconazole was detected.
Amphotericin B resistance was detected in 7% of Candida species. Candidemia was detected in 19 (10.6%) of 179 patients. In these 19 patients, the most common causative agent was C. parapsilosis (57.9%). Echinocandin resistance could not be evaluated during the study period because the antimicrobial agent was not available in our hospital.
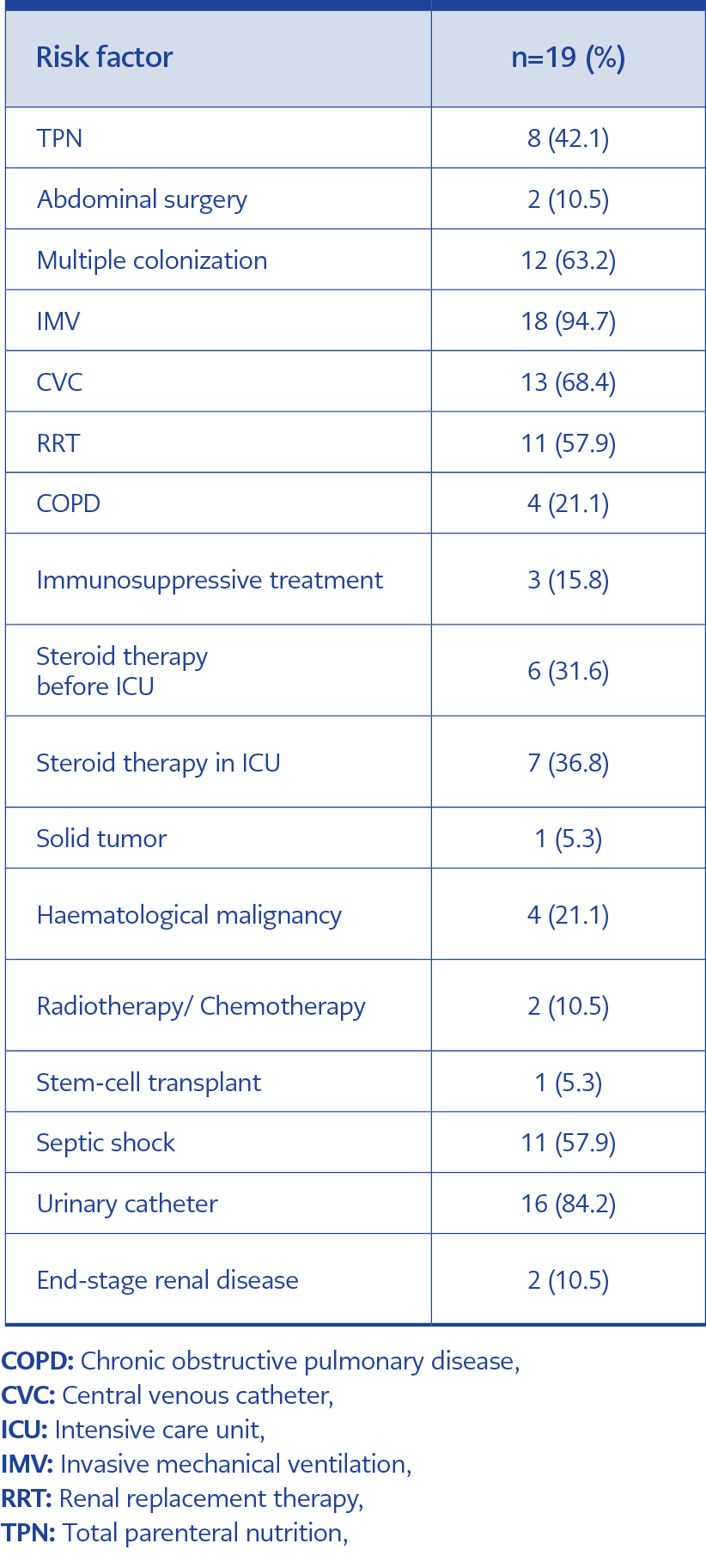
Nineteen patients with candidemia were examined for the relationship between risk factors and Candida type. Most common risk factors noted were IMV (n=18), CVC (n=13), RRT (n=11) and TPN (n=8). Septic shock accompanied candidemia in 11 patients, and C. parapsilosis positivity seemed more likely to present with septic shock.
When patients with and without candidemia were compared Candida score, CCI and length of ICU stay were determined as possible risk factors for the development of candidemia (Table 4). We performed multivariate logistic regression analysis to evaluate the independent effects of probable predictors determined as a result of univariate analysis in predicting the development of candidemia. As a result of this analysis, we found that length of ICU stay did not increase the risk of candidemia, and patients with higher candidemia scores and lower CCI were more likely to develop candidemia (Candidemia score: OR:1.93, 95% CI:1.27-2.94, p:0.002; CCI: OR:0.77, 95% CI:0.62-0.95, p:0.016).
When patients, who were admitted to ICU during the same period, with (either colonization or infection) and without fungal growth were compared, a significant difference was found between the groups in terms of age, APACHE II score, length of ICU and hospital stay, ICU and hospital mortality (p <0.05) (Table 5).
Discussion
Fungal infections have increased rapidly in the last few decades (7). Infections related to Candida species are seen more frequently in ICUs. These infections are associated with 47% mortality (8). Many risk factors have been identified for the development of candida infections, such as broad-spectrum antibiotic treatment, hemodialysis, pancreatitis, TPN and treatment with steroids or other immunosuppressive agents (9). Despite new antifungal agents and advanced care, candidemia is associated with increased medical cost, mortality and long-term hospitalization (10). A change in the pattern of infecting strains from C. albicans to non-albicans Candida species, such as C. glabrata, C. tropicalis, C. parapsilosis and C. krusei, is noted (11).
In the EPCAN observational study evaluating 956 positive fungal cultures, the most common strain was Candida spp. (94.1%) (12). In a thesis study evaluating candidemia in non-neutropenic ICU patients in our university, C. albicans was observed at a rate of 50.7% (13). In our study, C. albicans was the most common strain, followed by C. tropicalis and C. parapsilosis. The increasing frequency of C. glabrata over the years was also remarkable.
Patients with a high probability of invasive candidiasis, that is, patients with more comorbidities and who are clinically septic, are empirically administered antifungal treatment while waiting for culture results. For some patients, fungal growth was reported only from tracheal aspirate or urine samples, and antifungal treatment was discontinued in those patients who were not considered to have a fungal infection. Positivity of the tracheal aspirate sample was not considered a diagnostic feature alone for fungal infection for any of the patients. These patients were considered to have fungal colonization.
In a recent multi-center study conducted in our countries ICU’s, candidemia developed in 11.9% of colonized patients (14). In our study, the rate of development of candidemia after colonization was determined as 12%. Therefore, colonized patients in the ICU should be carefully monitored, considering the risks of possible invasive candidiasis in the future.
Recent epidemiological studies show an increase in candidemia because of C. parapsilosis (15,16).
In our study, C. parapsilosis was observed as the most common agent in candidemia. A positive culture was detected in the first 72 hours in 5 of these patients. The incidence of increasing C. parapsilosis infections belongs to the organism’s ability to grow in parenteral nutrition solutions and its tendency to form biofilms that lead to colonization of intravascular devices and prosthetic materials (17,18). In our study, except for one patient, all of the patients who developed candidemia by C. parapsilosis had a CVC, which is considered a risk factor for positive C. parapsilosis culture.
Seventy-two percent of patients who developed candidemia due to C. parapsilosis were in a septic shock when samples were obtained. It was thought that septic shock may have developed early during the infection since it was related to an intravascular device.
Procalcitonin value below <2.00 ng/ml is one of the findings that may help in the early differential diagnosis between candidemia and bacteremia (19). In the current study, the mean procalcitonin value in patients with candidemia was 1.5 ng/ml accordingly.
In the study conducted by Al-Dorzi et al. in 2018, it was found that 42.9% of patients with suspected candidemia were administered empirically antifungal treatment and caspofungin was the most frequently used agent (20). In our study, 68.4% of the patients with candidemia were administered antifungal treatment considering the clinical and laboratory data before the blood culture result was reported. No significant difference was found in the APACHE II and SOFA scores, mortalities and length of stay of the patients administered empirical or targeted therapy. It was known that early antifungal treatment decreases mortality and shortens the length of stay in patients with candidemia. However, we did not find a significant difference in this study due to the small number of patients with candidemia.
The European Society of Clinical Microbiology and Infectious Diseases (ESCMID) and the Infectious Diseases Society of American (IDSA) 2016 guidelines no longer recommend the use of fluconazole in first-line therapy for invasive candidiasis and recommends the use of echinocandins as primary care empirical therapy (4,21). In the present study, we observed that caspofungin was preferentially administered in septic patients with candidemia in accordance with the literature and current guidelines. However, the validity of this recommendation should be reviewed regarding local resistance patterns because strains’ resistance against fluconazole was lower than the literature indicated.
It is stated that de-escalation is a safe choice in ICU, whereas improper treatment at the time of diagnosis prolongs ICU stay and increases mortality (22). Yet, in our study only one patient’s systemic antifungal therapy was de-escalated to fluconazole.
In another study conducted by Jordà – Marcosve et al., the ICU mortality rate was 54% in candidemic patients, while 29.4% in patients without candidemia (23). In our study, the ICU mortality rate was 52.6% in patients with candidemia and 23.8% in patients without any fungal growth. APACHE II scores and length of ICU stay were significantly higher in patients with candidemia than patients without fungal growth. The higher mortality rate, APACHE II score and length of ICU stay in patients with candidemia may be due to high-risk factors and comorbid conditions as well as disease severity. A significant difference was found in multivariate analysis between Candida scores of patients with candidemia and without candidemia. It is known that Candida score can be an important parameter in determining the risk of invasive candidiasis. In our study, Candida score was found to be higher in patients with candidemia.
Our study had some limitations. Firstly, since it was a retrospective study, some patients’ data could not be accessed. Secondly, the contemporaneous positive bacterial culture of patients was not evaluated. Thirdly, the antibiotic susceptibility test was not performed in every fungal positivity, and the echinocandin susceptibility of the strains could not be evaluated. As a result, a complete susceptibility and resistance pattern of the strains could not be evaluated.
Conclusions
Despite increasing rates of fungal infections in ICUs, diagnosis is still difficult due to the lack of specific symptoms, late positivity in cultures and the common occurrence of contemporaneous bacterial infections. Delayed treatment is directly related to increased mortality. Therefore, it is important to know the risk factors to identify high-risk patients and administer early antifungal treatment. The use of the Candida score for this purpose is relevant in critically ill medical patients. In the face of increasing rates of non-albicans Candida infections, knowing the local susceptibility patterns when planning preemptive treatment of candida infections is crucial.