Introduction
Many parts of the world are affected by Echinococcus granulosus (1). This helminthic zoonosis leads to important public health and economic burdens in endemic regions (2, 3). E. granulosus can even lead to death if complications occur (4). Dogs are the definitive hosts of E. granulosus, where the larval state, called protoscoleces, develop into adult worms in their intestines. Among the intermediate hosts are cattle and sheep, while humans are only accidental intermediate hosts where protoscoleces from hydatid cysts (1, 2) cause hydatid disease (HD) (4).
The main treatment options for HD are percutaneous aspiration and surgery (5). The most preferred therapeutic option in humans is surgery (6). In surgical management, the fundamental steps are intracystic protoscoleces neutralization, followed by evacuation and residual cavity management (7). Spilled protoscoleces during surgery have the ability to create new cysts by implantation (2, 3). Therefore, it is obligatory to use scolicidal agents to inactivate protoscoleces (4). To prevent intraperitoneal dissemination surgical site is walled off with scolicidal agent-soaked gauze (7). Povidone-iodine (PI) and chlorhexidine-gluconate (Chx-Glu) are among many used scolicidal agents in this context (4, 7). Although many scolicidal agents have proven in vitro efficacy, toxicities and side effects limit their in vivo usage (3, 4). All scolicidal agents used in history have adverse effects, which are mostly unacceptable, and prevent their safe use (8). Besides all this, sodium chloride is electrolyzed to produce hypochlorous acid (HOCl) (9). Hypochlorous acid is synthesized by immune cells, is part of reactive oxygen species (ROS), and has antimicrobial properties (10).
The search for the most effective and physiological scolicidal agent suitable to be used during surgery continues today. There is no way to calculate the applied scolicidal agents’ concentration in the cyst. Thus, knowing the efficacity of an agent at different concentrations is important. Therefore, in this study, we aimed to determine whether;
- HOCl, an agent which can be used on mucosal surfaces (11), has a scolicidal effect,
- to determine the scolicidal efficiency of HOCl at different concentrations,
- to compare HOCl’s efficiency with PI 10%, a well-established scolicidal agent in Turkey (4), and with 0.04% Chx-Glu, which has been claimed to be an ideal scolicidal agent (12), and
- to test HOCl’s scolicidal effectiveness on sterile gauze at different concentrations.
Materials and Methods
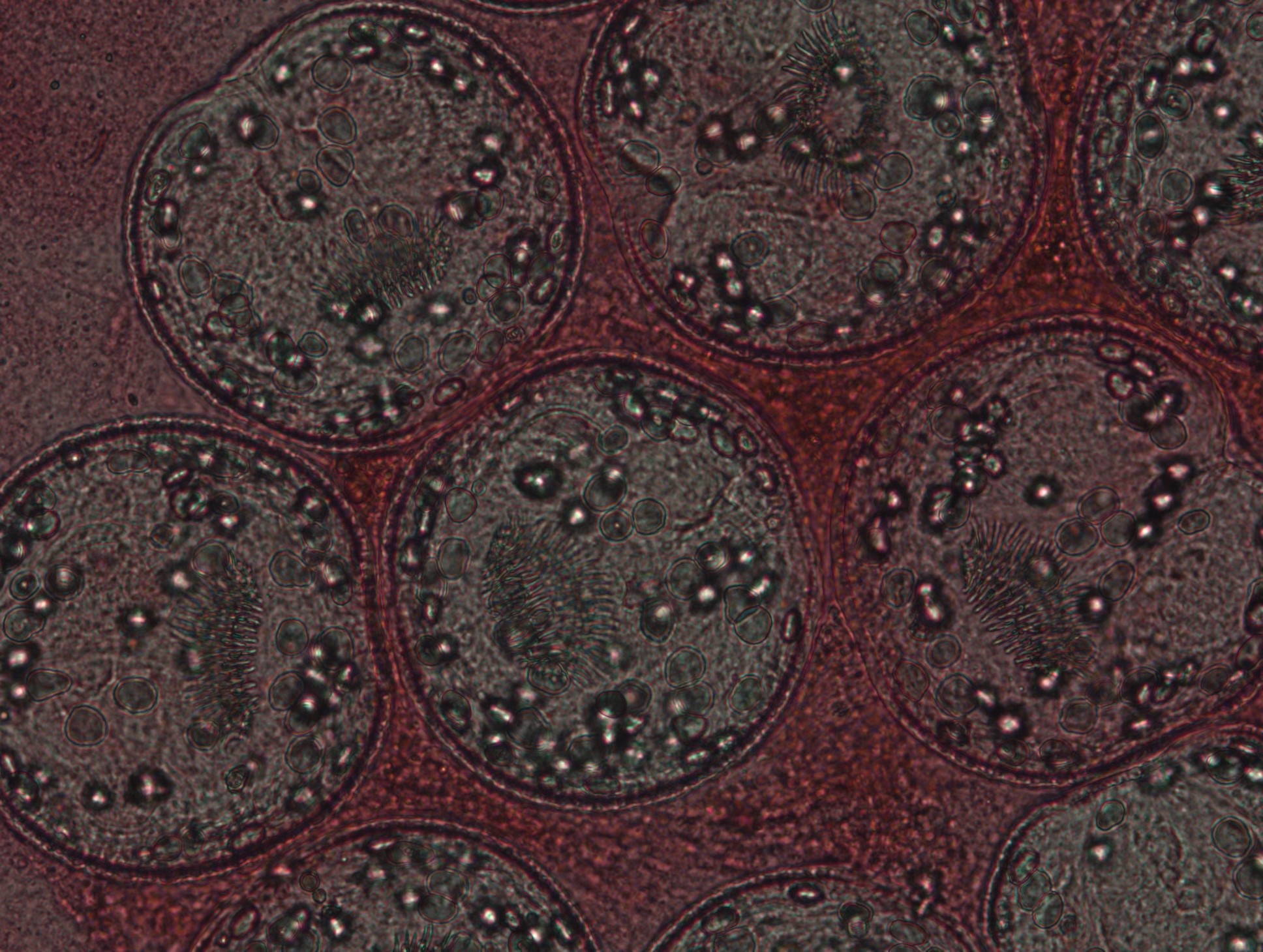
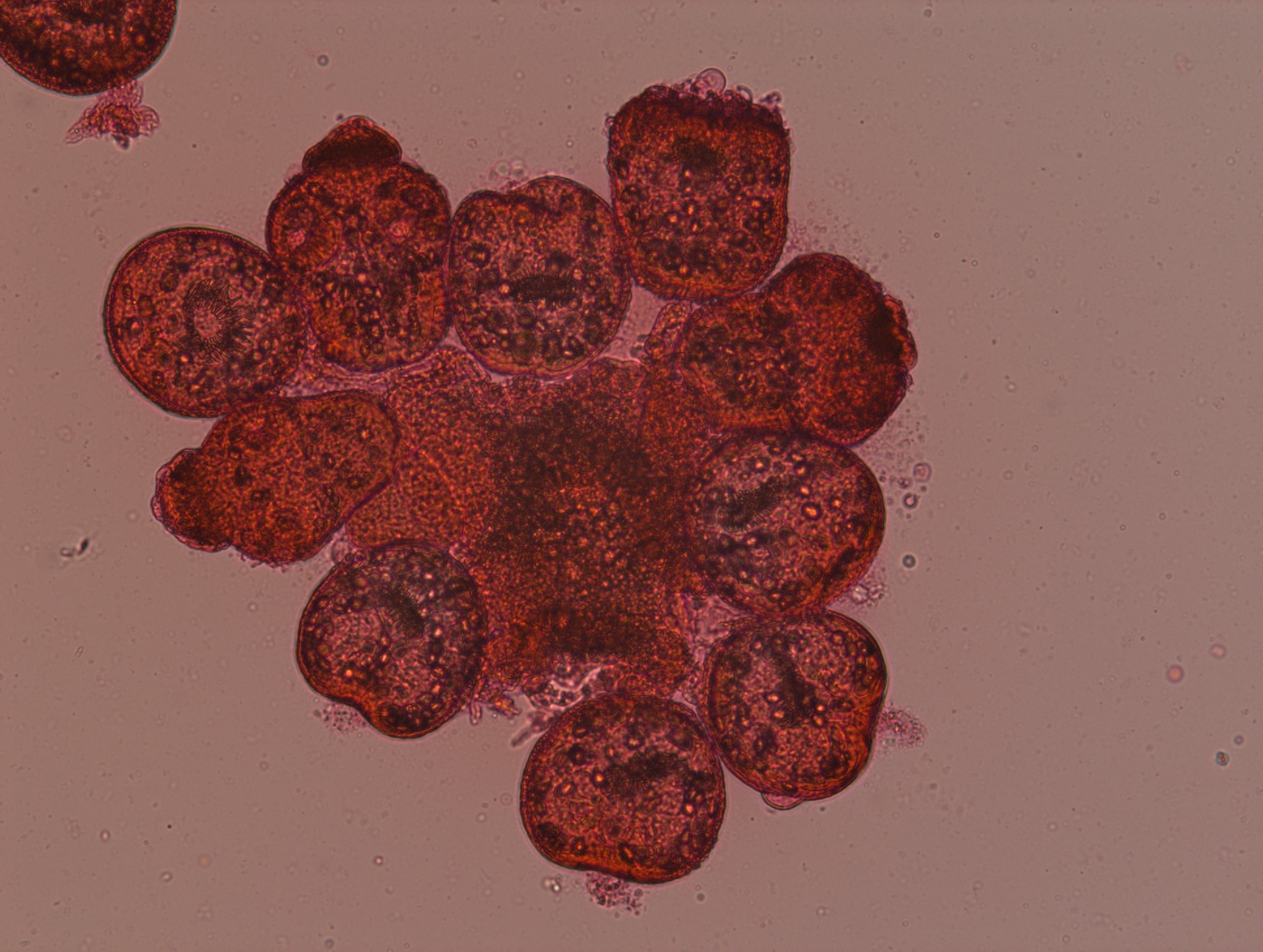
Livers with cysts from sheep were obtained from a local slaughterhouse in Çanakkale province, Turkey. Aseptic methods were used to collect hydatid fluid in sterile tubes. The supernatant was removed after the protoscoleces were sedimented and formed hydatid sand. The obtained suspension had 4000±100 protoscoleces/mL, and viability was 95%. A microbiologist with 25 years of experience in parasitology evaluated viability by flame cell motility and 0.1% eosin dye exclusion test using a light microscope at X40 magnification. Live protoscoleces excluded the eosin dye (Figure 1), were colorless, and had flame cell activity, while dead protoscoleces had no flame cell activity and were colored red (Figure 2).
Our study consisted of two parts. First, the effectivity of HOCl (Crystalin; NPS Biosidal, İstanbul, Turkey) at different concentrations was tested and compared to PI 10% (Batticon; Adeka, Samsun, Turkey) and Chx-Glu (Hibitanol, Kimpa, İstanbul, Turkey).
Study groups with scolicidal agents in tubes were as follows: Group I (negative control): Saline (0.9% NaCl) (Adeka, Samsun, Turkey), Group II: 200 ppm/mL HOCl, Group III 20 ppm/mL HOCl, Group IV: 2 ppm/mL HOCl, Group V: 0.2 ppm/mL HOCl, Group VI (positive control): PI, 10%, Group VII (positive control): Chx-Glu 0.04%. In each group, 0.1 mL of study solution was added to 0,1 mL of protoscoleces-rich solution, and after waiting for 5, 10, and 15 minutes examined for flame cell activity and dye exclusion.
The effectiveness of the HOCl on the gauze was tested in the second part of the experiment, as described by Besim et al. (7). Briefly, 1×1 cm gauze was absorbed in different concentrations of HOCl, and one drop of protoscoleces-rich solution was dropped on each gauze. After 5, 10, and 15 minutes, gauze was immersed into different tubes filled with 0.9% NaCl and shaken vigorously. The gauze was removed, and the remaining solutions were examined microscopically for viability by flame cell activity and 0.1% eosin dye exclusion test. Study groups were defined as; Group A: 200 ppm/mL HOCl, Group B: 20 ppm/mL HOCl, Group C: 2 ppm/mL HOCl, and Group D: 0.2 ppm/mL HOCl. All experiments in both study groups were repeated three times.
Results
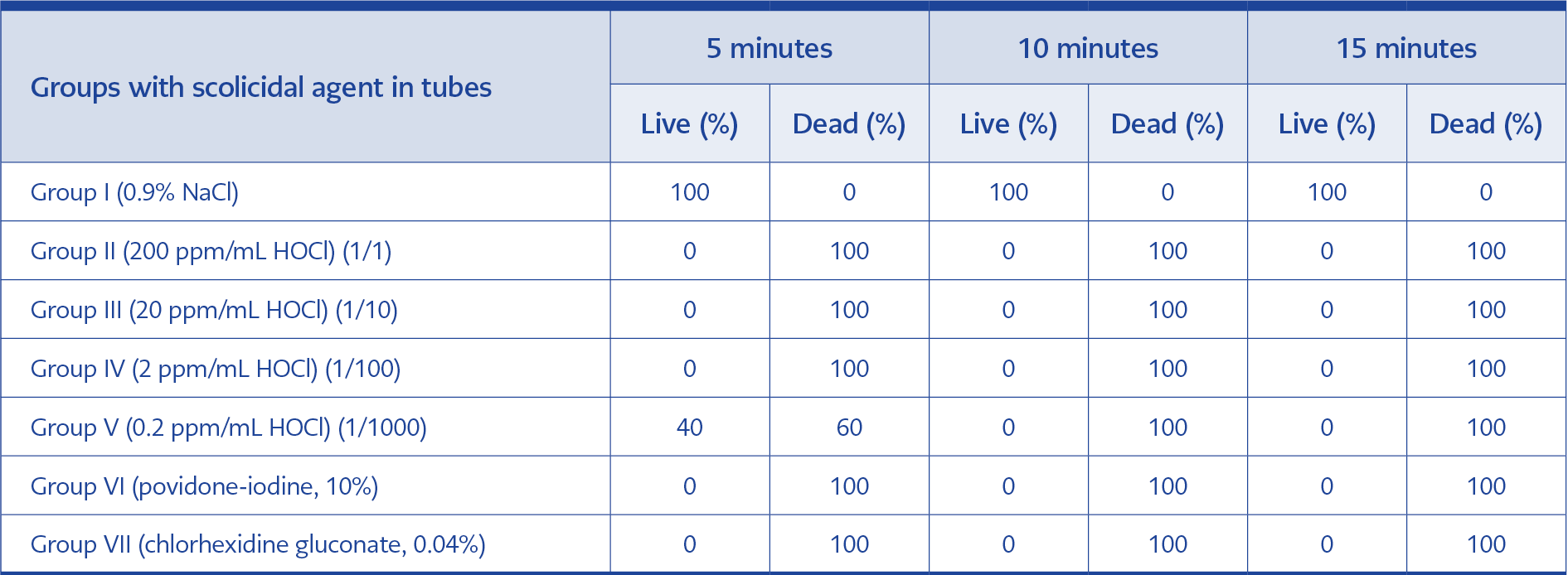
The scolicidal agent/concentration, acting times, and scolicidal activity results are given in Table 1. All scoleces survived all test durations in the 0.9% NaCl group (negative control group). HOCl was 100% effective at 1/1 (200 ppm/mL), 1/10 (20 ppm/mL), and 1/100 (2 ppm/mL) concentration in all tested time periods. HOCl at 1/1000 (0.2 ppm/mL) concentration in 5 minutes was 60% effective and 100% effective after 10 and 15 minutes. Povidone-iodine and Chx-Glu both had a 100% scolicidal effect at all three tested periods (positive control groups).
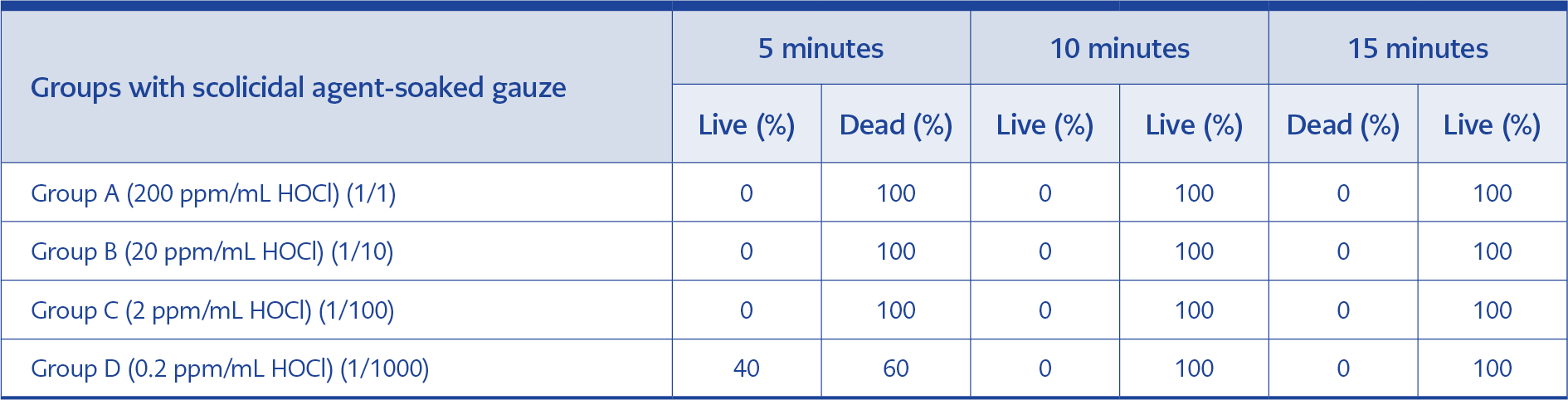
In the second part, the scolicidal efficiency of HOCl on gauze was tested after 5, 10, and 15 minutes and the results were similar to the first part of the study, as shown in Table 2.
Discussion
Hydatid disease is an important public health problem, especially in developing countries. Although many clinical and experimental studies about the treatment have been published, an ideal scolicidal agent has not been defined (4). Surgery continues to be the most common treatment for HD (13). Protection of the surgical site with scolicidal agent-soaked gauze to prevent inoculation of spilled protoscoleces and cyst content aspiration, followed by delivering a scolicidal agent into the cyst long enough to act, are vital steps in surgery (3, 14). Thus, we also evaluated the scolicidal effectiveness of HOCl on gauze.
We observed no scolicidal activity after 15 minutes of saline exposure, which was concordant with 0.9% NaCl being used as the negative control in some studies (15, 16). Povidone-iodine and Chx-Glu are among many scolicidal agents used to inactivate protoscoleces (2). Their effectiveness is concentration-dependent, and their end concentration in the cyst content cannot be exactly known (7). An ideal scolicidal should act in a short time, be effective at low concentrations, be easy to apply, and have minimal toxicity (14).
The effectiveness of PI 10%, a preferred scolicidal agent, is dose-dependent (4, 7). Serious side effects have been described, such as renal failure, even at low concentrations (17). Peritoneal lavage with PI can lead to death caused by peritoneal fibrosis (18). Another undesired factor is that tissue proteins or blood, which are part of the operative field, have the potential to inactivate PI (19). Thus, it has been proposed that PI should not be used under any circumstances (20). Our in vitro study, where we used undiluted PI and no interfering particles or substances were present, confirmed its known in vitro scolicidal activity and served as a positive control group.
Topçu et al. proposed that 0.04% Chx-Glu is the most potent scolicidal agent (21) and is effective in 5 minutes (12). The Chx-Glu solution used in their study was not pure; the form used in the study had additives that may have potentialized its scolicidal activity. It must also be prepared preoperatively to the desired effective concentration and non-toxic dilution. In our study, we used 0.04% Chx-Glu (Hibitanol; Kimpa, İstanbul, Turkey) because Klorhex solution (Drogsan, Ankara, Turkey) was not available anymore. Hibitanol must also be prepared preoperatively; it is not pure, has additives, and has been effective at all tested times. Chlorhexidine is also reported to be one of the most common perioperative allergenic agents (22), and 0.05% and higher concentrations of Chx-Glu have been reported to cause peritonitis (3). Thus, caution when using Chx-Glu should be emphasized.
To our best knowledge, there are no studies conducted on the scolicidal effect and efficiency of HOCl. The hypochlorous acid solution, a weak acid, is used as a disinfectant in hospitals, animal facilities, and food packaging plants (23). HOCl is appropriate for the disinfection of endoscopes that get in contact with mucous membranes (24). It has been widely used as a disinfectant for medical instruments and inanimate surfaces and could potentially be used as a disinfecting agent for nanostructured implant biomaterials without compromising their performance (25). HOCl is endogenously produced by the myeloperoxidase system (26); it has powerful oxidative properties and is non-irritating and non-sensitizing to mammalian cells within its effective antimicrobial range (27).
Hypochlorous acid is highly active against all bacterial, viral, and fungal human pathogens, and a small amount of HOCl can kill spore-forming and non-spore bacteria in a short period (28). HOCl also has an anti-biofilm and killing effect on microorganisms within the biofilm (11). It has a significant protective effect on tissues by inhibiting the production of inflammatory mediators and thereby contributes to the processes of tissue protection (10). Moreover, it is non-toxic, and its favorable effects on human cells promote its use on mucosal surfaces. With these properties, stabilized HOCl is widely used in infected or non-infected wounds and parasites (11). HOCl has been authorized in the United States to be used in food since 2017 and in China to be used for human mucous membranes since 2020 (9). So far, no resistance has been observed against hypochlorous acid (29). Thus, we concluded that HOCl could have a scolicidal effect on EG protoscoleces and maintain its scolicidal properties on gauze. Both assumptions have been proven to be correct in the first and second parts of our experiment.
HOCl is generated from sodium chloride by electrolysis. The concentration used in this study was 200 ppm, pH 7.1, oxidation-reduction potential (ORP) 871 MV, and its stability was 24 months (NPS Biosidal, İstanbul, Turkey). For this study, we used its 1/1, 1/10, 1/100, and 1/1000 concentrations. Most in vitro cytotoxicity studies have found if the sodium hypochlorite concentration is kept to 0.025% or less, effects on cultured cells are minimal or nonexistent (30). When converting units, 0.0001% is 1 ppm, and 0.025% is 250 ppm. The highest HOCl concentration in our study was 200 ppm/mL, significantly lower than the mentioned 250 ppm/mL. These values imply HOCl to be a safe and non-toxic scolicidal agent, even at a cellular level.
All studied scolicidal agents were concentration-dependent, and their degree of dilution in the cyst content was unpredictable (7). A dilution up to 2 ppm/mL HOCl (1/100 dilution of 200 ppm/mL) was 100% effective in 5 minutes, while 0.2 ppm/mL (1/1000 dilution of 200 ppm/mL) was 60% effective in 5 minutes, and 100% effective in 10 minutes. The knowledge of a cut-off dilution was valuable additional information for our study and indicated a wide action range at unintended dilutions.
In conclusion, hypochlorous acid is a commercially available product without further preparation needed before use and has a broad spectrum. Hypochlorous acid maintains 100% efficiency in 5 minutes at 1/100 dilution and %100 efficiency in 10 minutes at 1/1000 dilution, which are positive attributions during surgery. These positive attributions were also shown on gauze, which shields surgical sites against spillage-caused hidatosis. Our in vitro study will be the way for in vivo studies.