Introduction
Healthcare workers were on the front line of the global effort against COVID-19. Thousands of physicians died from COVID-19 in many countries (1). In addition, difficulties in accessing personal protective equipment (PPE), lack of training, and workforce shortages have exposed the vulnerability of healthcare workers who desperately sought other options to protect themselves (2).
There were efforts to discover an effective treatment and prophylaxis for coronavirus disease. At the same time, hydroxychloroquine (HQN), an antimalarial drug mainly used in treating immune-mediated diseases, was proposed as an option. However, while in vitro studies showed the ability of HQN to inhibit SARS-CoV-2 activity (3, 4), consequent clinical trials have not yielded promising results for the effectiveness of HQN in the treatment and prophylaxis of COVID-19 (5-7). Nevertheless, some countries included HQN in their national case management and prophylaxis guidelines, including Turkey, until recently (8, 9).
In March 2020, the Turkish Ministry of Health declared that pandemic hospitals were responsible for managing COVID-19 cases. Pandemic hospitals had to have at least two physicians specialized in infectious diseases and clinical microbiology, pulmonology, or internal medicine and had a level three adult intensive care bed. Istanbul, having one-fifth of the population in Turkey and being the center of commerce and international travel, was soon reported as the epicenter of the pandemic in Turkey. While physicians working in pandemic hospitals in Istanbul faced the surge of COVID-19 patients, they were also suggested to use HQN as a prophylaxis against COVID-19. Therefore, this study mainly aimed to describe the patterns of HQN use among physicians working in pandemic hospitals in Istanbul. In addition, we aimed to illustrate the factors contributing to HQN use among physicians and investigate the association of HQN use with the acquisition of COVID-19.
Materials and Methods
This cross-sectional study targeted physicians working in pandemic hospitals assigned to respond to the COVID-19 outbreak in İstanbul, Turkey. The study was approved by The Koç University Ethics Committee on Human Research (IRB No: 2020.183.IRB1.051) and the Turkish Ministry of Health (-2020-05-06T16-18-41).
We invited participants from seven pandemic hospitals. To ensure including a socioeconomically diverse group of hospitals, we chose three private university hospitals and four public hospitals. The total number of physicians working in these hospitals was 4722. We calculated the sample size in OpenEpi (10) program by taking the anticipated frequency of HQN use as 50% as recommended for unknown frequencies, 5% margin of error with a design effect of 1.7. The sample size at a 95% confidence interval (CI) was 607 participants.
The inclusion criteria for the participants were having a minimum medical doctor degree and actively working with patients in pandemic hospitals since March 11, 2020. We excluded interns and non-physician healthcare workers, including nurses, paramedics, caregivers, and administrative staff. We used a non-probabilistic method of convenient sampling approach to select participants by invitation sent via email from hospital administration to the physicians. We sought permission from each hospital to send an invitation email to their eligible physician staff. Hospital administrations sent two reminders in a weekly period after the first invitation email. We collected the data using an online survey, including 57 questions prepared according to the scientific literature. Physicians could proceed to the survey upon providing consent. Data collection was limited to one month between May 14, 2020, and June 13, 2020.
The survey assessed the socio-demographic characteristics of the participants (such as age, gender, and medical specialty), use of HQN since the pandemic’s beginning, dose, timing, and duration of HQN use, and use of any other supplements. In addition, it assessed the physicians’ working conditions during the pandemic, frequency of contact with COVID-19 patients, use of PPE, and whether they used regular medication or had any chronic health condition. We accepted the departments of infectious diseases, emergency, pulmonology, internal medicine, intensive care, ear-nose-throat, and pediatrics as the front-line departments. Based on the preliminary data analysis we conducted regarding the association between COVID-19 infection and the reported duration of weekly COVID-19 patient care, we dichotomized the variable by defining a cut-off for the duration of COVID-19 patient care as more than 12 hours per week, which was named as “prolonged COVID-19 patient care”.
Statistical Analysis
We provided a mean, median, and standard deviation (SD) for continuous variables and percentages for the categorical variables. We dichotomized the categorical variables and used the Chi-square test for comparisons in univariate analysis. We conducted two separate multivariable analyses with logistic regression. The significant variables in univariate analysis and the potential confounders were included in the models. One multivariable analysis for the predictors of HQN use was performed, including independent variables of age, gender, comorbidities, unprotected COVID-19 exposure, and working in the front-line departments. The second multivariable analysis was performed for the infection predictors, including age, gender, comorbidities, working in the front-line departments, and prophylactic HQN usage. Statistical significance was set as p<0.05 to reject the null hypothesis in a 2-sided equation. STATA software version 8.0 (Stata Corp., College Station, USA) was used in the statistical analysis.
Results
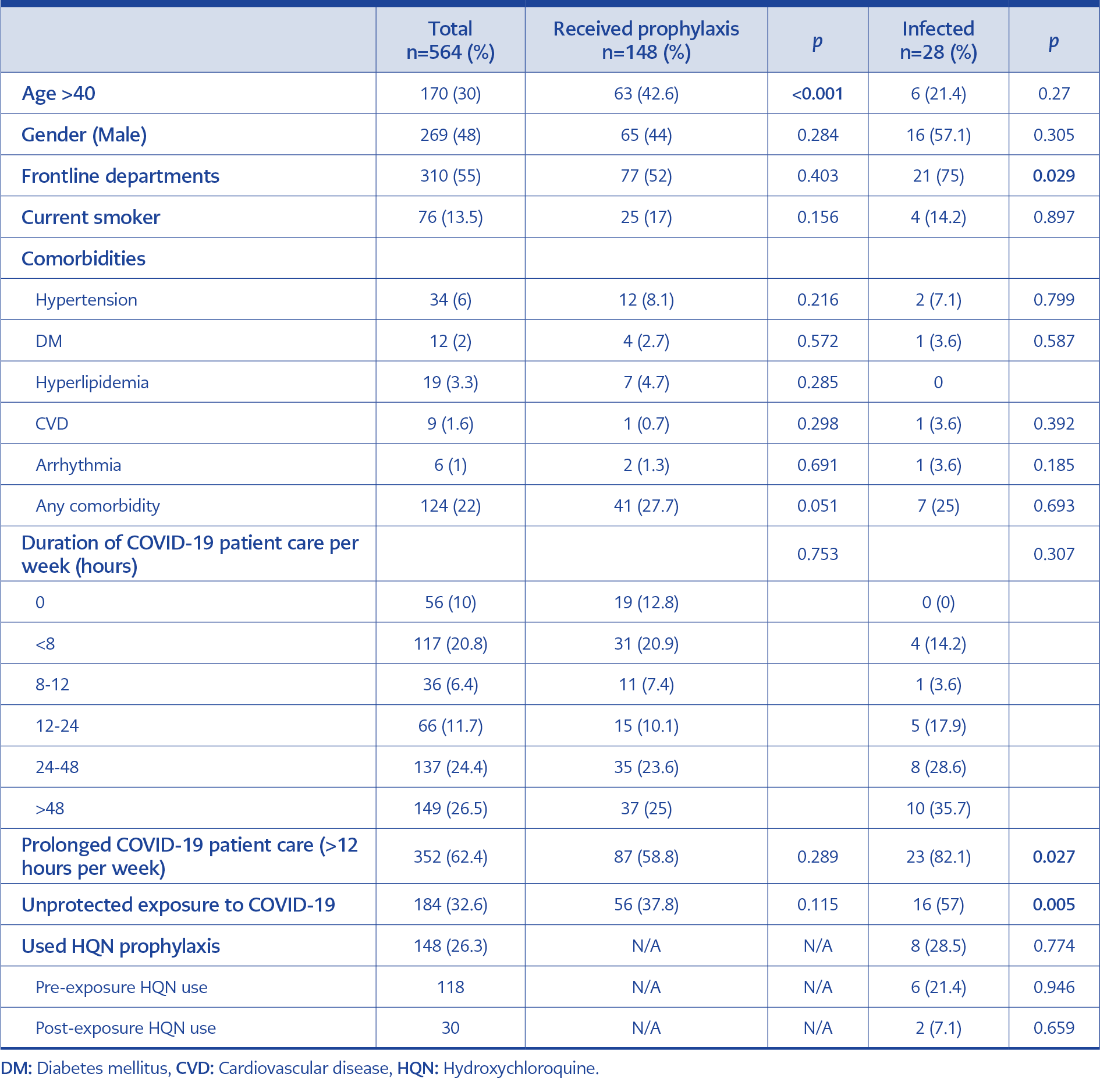
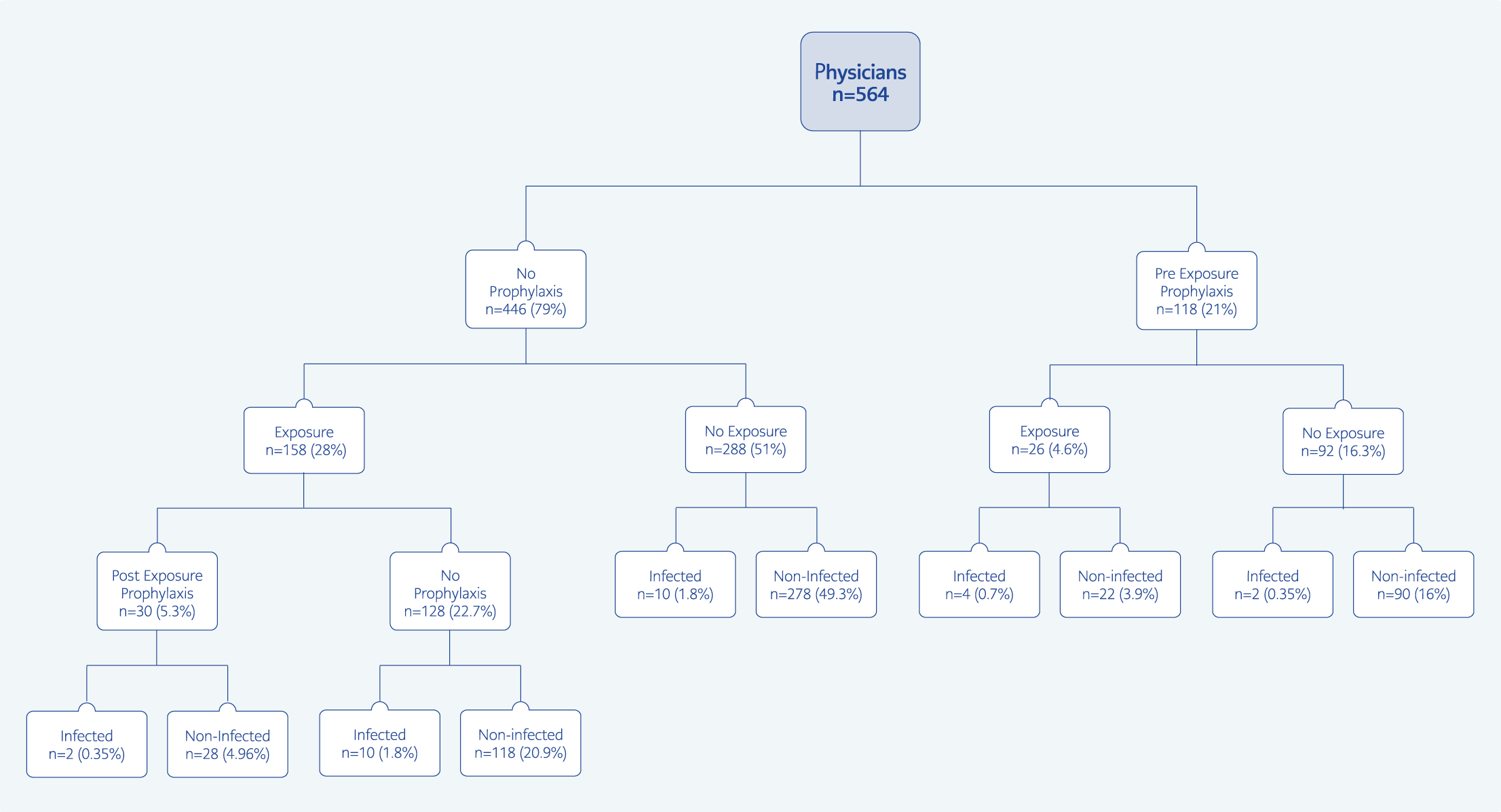
At the end of the data collection period, we received 718 responses. Sixty-eight responses were incomplete, 36 were from doctors outside of selected pandemic hospitals, and 50 were from non-physician healthcare workers, so they were excluded. After eliminating the participants who were out of our target population from seven pandemic hospitals, 564 physicians remained for analysis comprising 93% of the minimal sample size, which accounted for in the power analysis. The mean age was 36 years (SD=8.9), and 295 (52%) were women (Table 1). Our results indicate that 28 out of 564 (5%) physicians reported that they were infected with COVID-19, and pneumonia developed in 15 of 28 (54%) infected physicians. One hundred forty-eight (26%) physicians out of 564 used hydroxychloroquine for a while or continuously for preventive purposes. Among the physicians who reported using HQN, eight of 148 (5.4%) got infected, compared to 20 of the 416 (4.8%) physicians who did not report using HQN (p=0.773) (Figure 1).
There were 310 (55%) physicians in the front-line departments. Out of these 310 physicians, 21 (6.7%) reported that they were infected, while only seven physicians (2.7%) reported from other departments (p=0.029). Also, physicians providing care for COVID-19 patients more than 12 hours a week were infected more than physicians working fewer hours (23/352; p=0.027) (Table 1).
In the study, 184 physicians stated that they had contacted at least one COVID-19 patient without having appropriate PPE (unprotected exposure). Among the physicians with suspected contact, 16 (8.7%) were infected (p=0.005). Fifty-six (30.4%) physicians out of 184 used HQN for prophylaxis. While 26 physicians used HQN before exposure to a COVID-19 patient, 30 began HQN after exposure. In addition, HQN did not differ statistically in terms of being infected among the exposed physicians (p=0.52).
Diarrhea (7.4%) was the most common side effect among physicians using HQN, and only one physician having diarrhea was infected. Arrhythmias (3.4%), nausea (3.4%), weakness (3%), abdominal pain (2.7%), rash (1.3%), and dizziness (1.3%) were the other most frequent side effects.
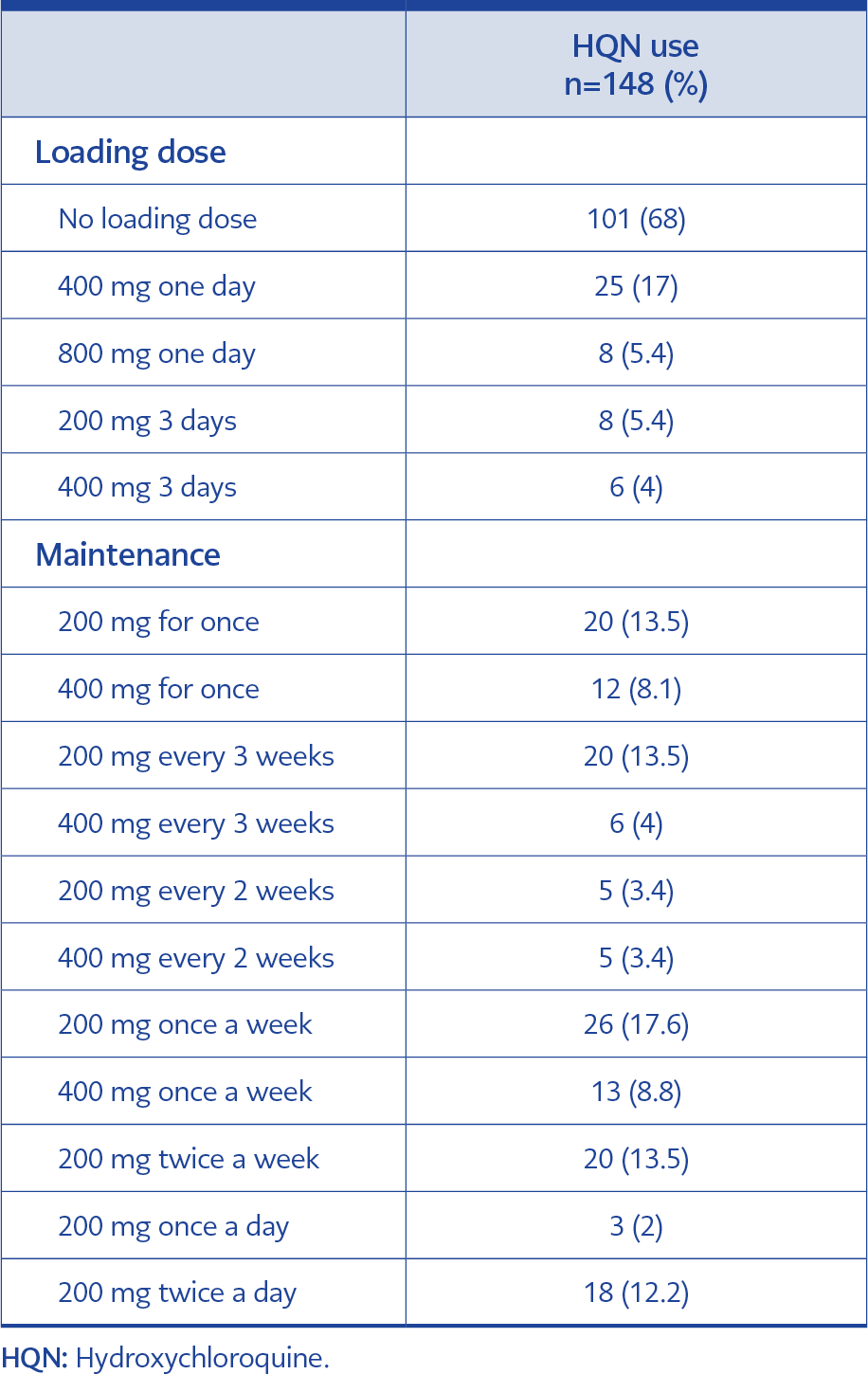
While 68% of the physicians never used a loading dose of HQN, those who used the loading dose mostly received 400 mg for a day (17%). Eleven different maintenance dosages were documented in the study, and 200 mg once a week (17.6%) was the most preferred dose for maintenance (Table 2).
Of the 148 physicians using HQN, 44 were still on prophylaxis when the study was terminated. Five of the 144 (3.4%) physicians who stopped the drug stated that after stopping prophylaxis, they developed a complaint or finding related to COVID.
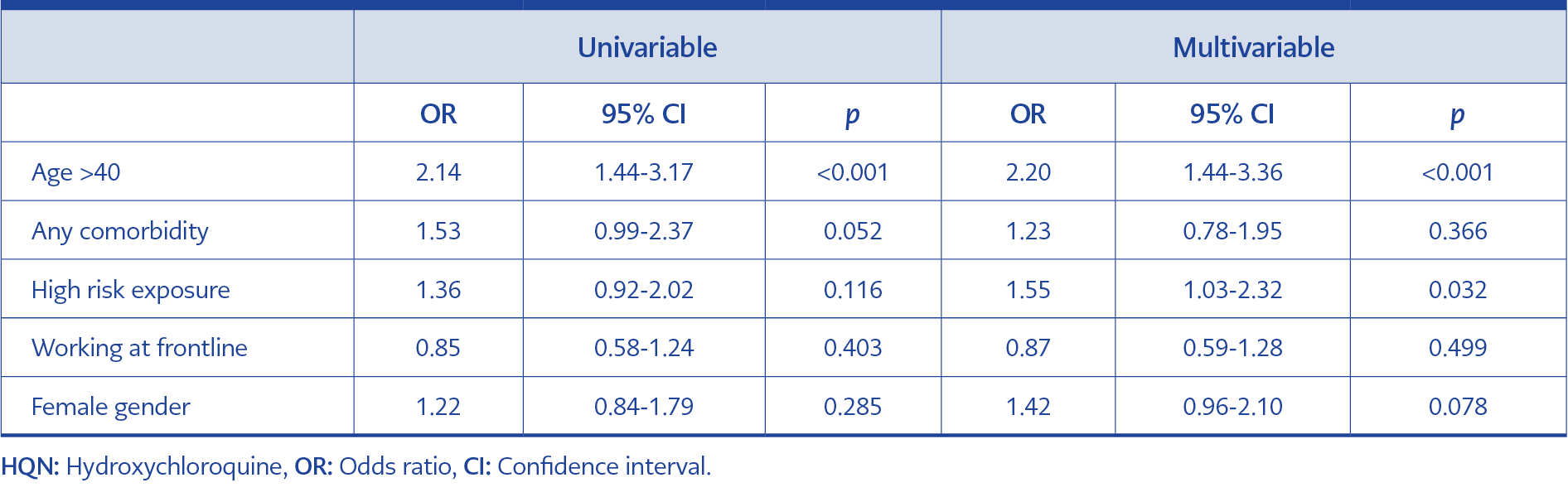
In the multivariable analysis conducted to determine who preferred HQN prophylaxis, we found that physicians over 40 years of age (p<0.001) and who had unprotected exposure to a COVID-19 patient (p=0.032) preferred prophylaxis (Table 3).
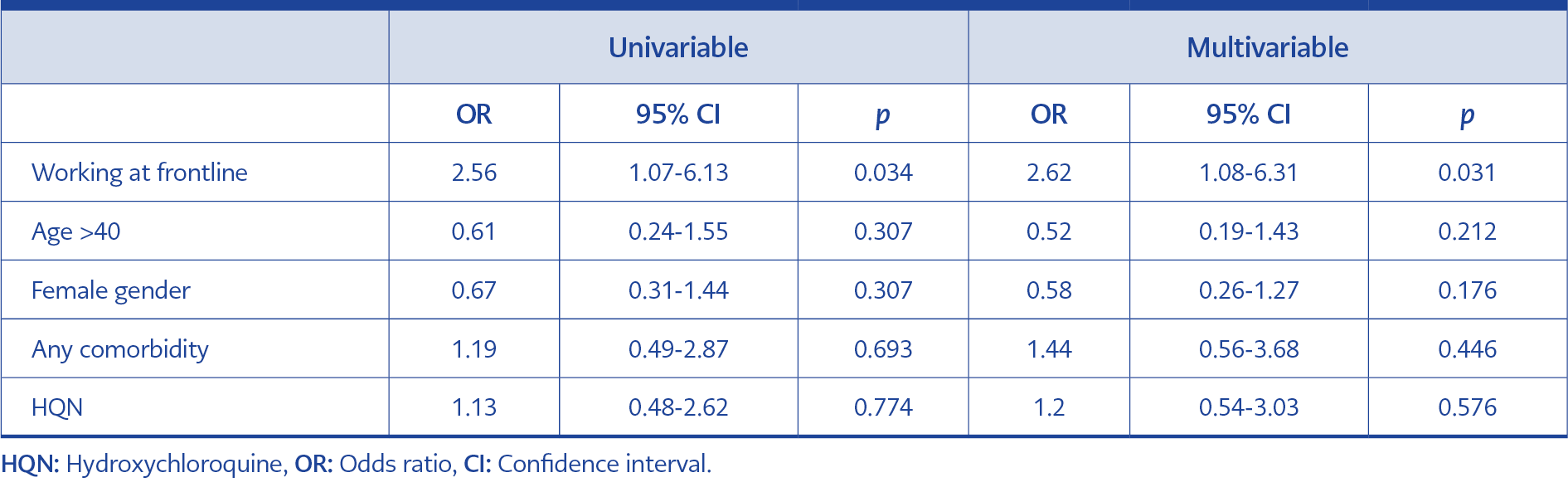
We also examined the predictors of COVID-19 infection, and in both univariate and multivariable analysis, working on the front line was the single significant variable for developing an infection while using HQN was not found to be protective (Table 4).
Discussion
Prophylaxis with HQN was suggested in some countries, although its effectiveness was not proven by clinical studies (8). The physicians working in pandemic hospitals tried to treat patients and protect themselves simultaneously. Since the HQN in prophylaxis was highly controversial, physicians themselves had to decide whether to use this drug for protection.