Introduction
Urinary tract infections (UTIs) are seen in all age groups and are among the most frequent bacterial infections. Enterobacterales are the most common bacterial species that cause UTIs (1). Escherichia coli is the most common cause of both complicated UTIs (65%) and uncomplicated UTIs (75%). E. coli is followed by other Enterobacterales such as Klebsiella spp., Proteus spp., and Serratia spp. (2, 3). Resistance of urinary tract pathogens to antibacterial agents (amoxicillin-clavulanic acid, trimethoprim-sulfamethoxazole, and ciprofloxacin), widely used for empirical treatment, is gradually increasing worldwide. In European countries, the resistance of E. coli to amoxicillin-clavulanic acid, trimethoprim/sulfamethoxazole, and ciprofloxacin has been reported to range from 5.3% to 37.6%, 10.5% to 39.8%, 14.6% to 21.4%, respectively. (4, 5). In addition, some studies have reported that extended-spectrum β-lactamases (ESBL) are increasing among Enterobacterales (6). Current guidelines do not recommend the usage of the antibiotic for empirical therapy if resistance to the antibiotic locally does not exceed 20% (7). Currently, fosfomycin has become more critical in treating UTIs because of increasing resistance to other agents and its limited adverse effects. A single dose of 3 g is recommended for the treatment of uncomplicated UTIs (7, 8).
Fosfomycin is an inhibitor of the MurA enzyme, UDP-N-acetylglucosamine enolpyruvyl transferase; thus, it inhibits the synthesis of peptidoglycan, which is necessary for bacterial cell wall synthesis (9). It has a broad spectrum of activity against most of both Gram-positive and Gram-negative bacteria (10). Because of its unique structure and action mechanism, cross-resistance is uncommon. However, mutations in MurA cause inherent resistance to fosfomycin in some bacteria, such as Chlamydia spp., Mycobacterium tuberculosis, and Vibrio fischeri. Fosfomycin-susceptible bacteria like E. coli usually develop resistance with mutations in the uptake systems. The susceptibility of ESBL-producing E. coli isolates was reported as 81%-100% and 15% -100% in ESBL-producing K. pneumoniae isolates (12). A study conducted in Germany, Belgium, and Spain showed that only <1.5% of E. coli were resistant to fosfomycin (13). In another study conducted in Switzerland, fosfomycin resistance among ESBL-producing E. coli was reported as 1.4% (14).
This study aims to evaluate the distribution of pathogens isolated from urine samples in our center to find the frequency of resistance to fosfomycin in both inpatients and outpatients and to find alterations in the frequency of fosfomycin resistance over time.
Materials and Methods
In this retrospective study, all urinary samples from both outpatients and inpatients sent to the microbiology laboratory of Ondokuz Mayıs University Medical Center from January 2012 to December 2020 were enrolled. In addition, demographic data, such as patient age and gender, and laboratory data were extracted from the hospital records. Only one urinary culture from each patient was included in the study.
Urine samples were routinely inoculated onto 5% sheep blood agar (bioMérieux, Marcy l’Etoile, France) and eosin methylene blue agar (bioMérieux, Marcy l’Etoile, France) and the plates were then incubated for 20-22 hours at 35 oC. The VITEK® MS automated system (bioMérieux, Marcy l’Etoile, France) was used for identification. The antimicrobial susceptibility and ESBL production of the isolates was determined using the VITEK® 2 Compact (bioMérieux, Marcy l’Etoile, France) and evaluated according to the European Committee on Antimicrobial Susceptibility Testing (EUCAST) Guideline specific to that year.
The study was approved by Ondokuz Mayıs University Medical Faculty Ethics Committee (B.30.2.ODM.0.20.08/498).
Statistical Analysis
The association between years and fosfomycin resistance rates was analyzed with the ANOVA test, and the association between outpatients/inpatients and fosfomycin resistance rates was analyzed with Pearson’s χ2 test. The statistical significance was set as p<0.05.
Results
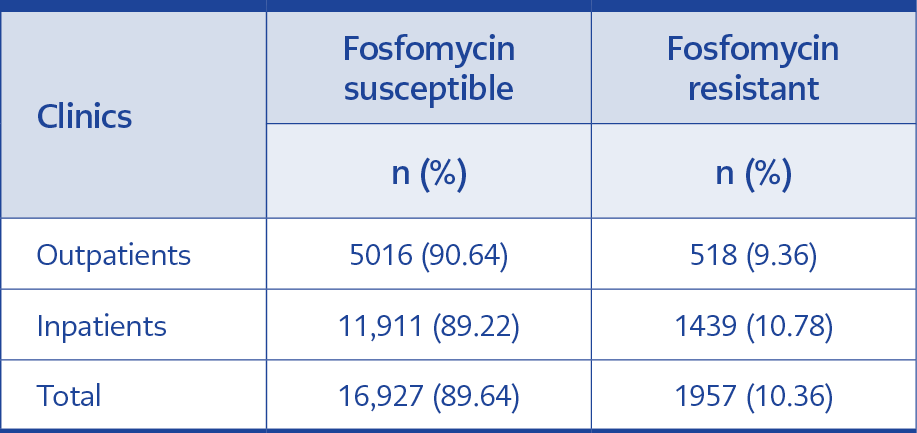
Table 3. Fosfomycin susceptibility and resistance in
isolates from inpatients and outpatients’ clinics.
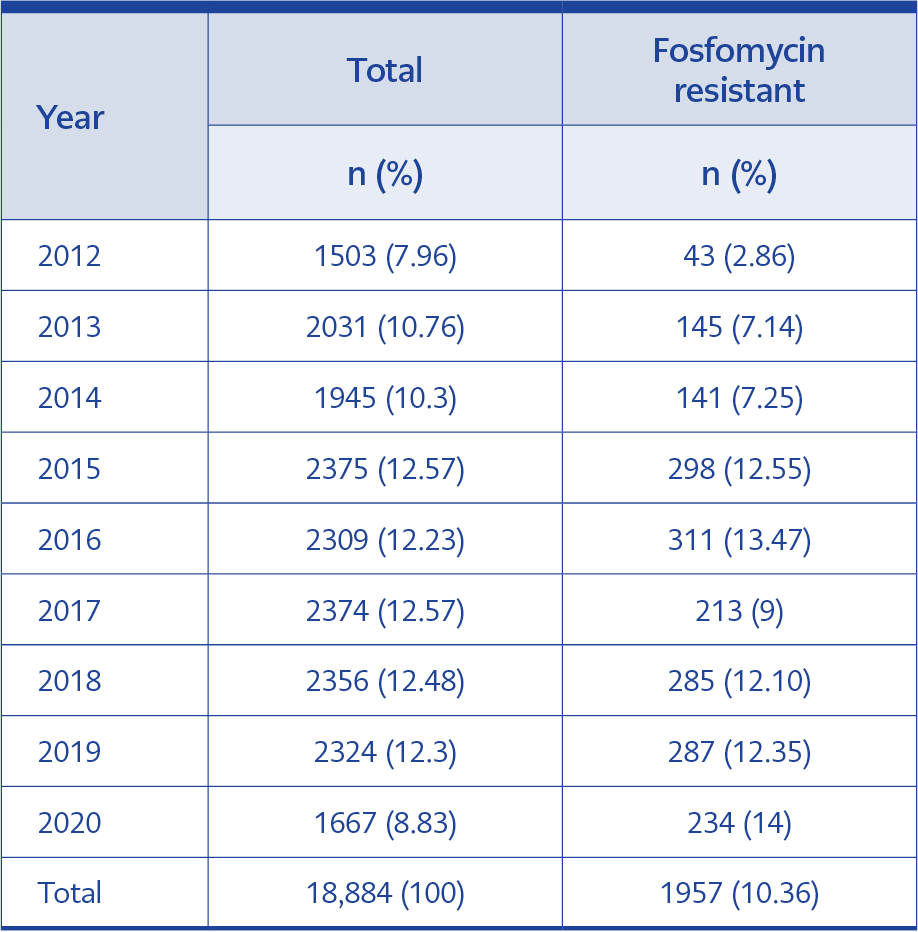
A total of 18,884 urine cultures were tested between 2012 and 2020; 12,063 (63.88%) cultures were from female patients. The mean age was 48.74 years (2-99). The total rate of fosfomycin-resistant isolates was 2.86% in 2012. The rate increased between 2012-2016; it was 13.47% in 2016. However, it was 9% in 2017 and 14% in 2020; the trend was statistically significant (95% confidence interval [CI], p<0.05). The alterations in fosfomycin resistance over the years are shown in Table 1.
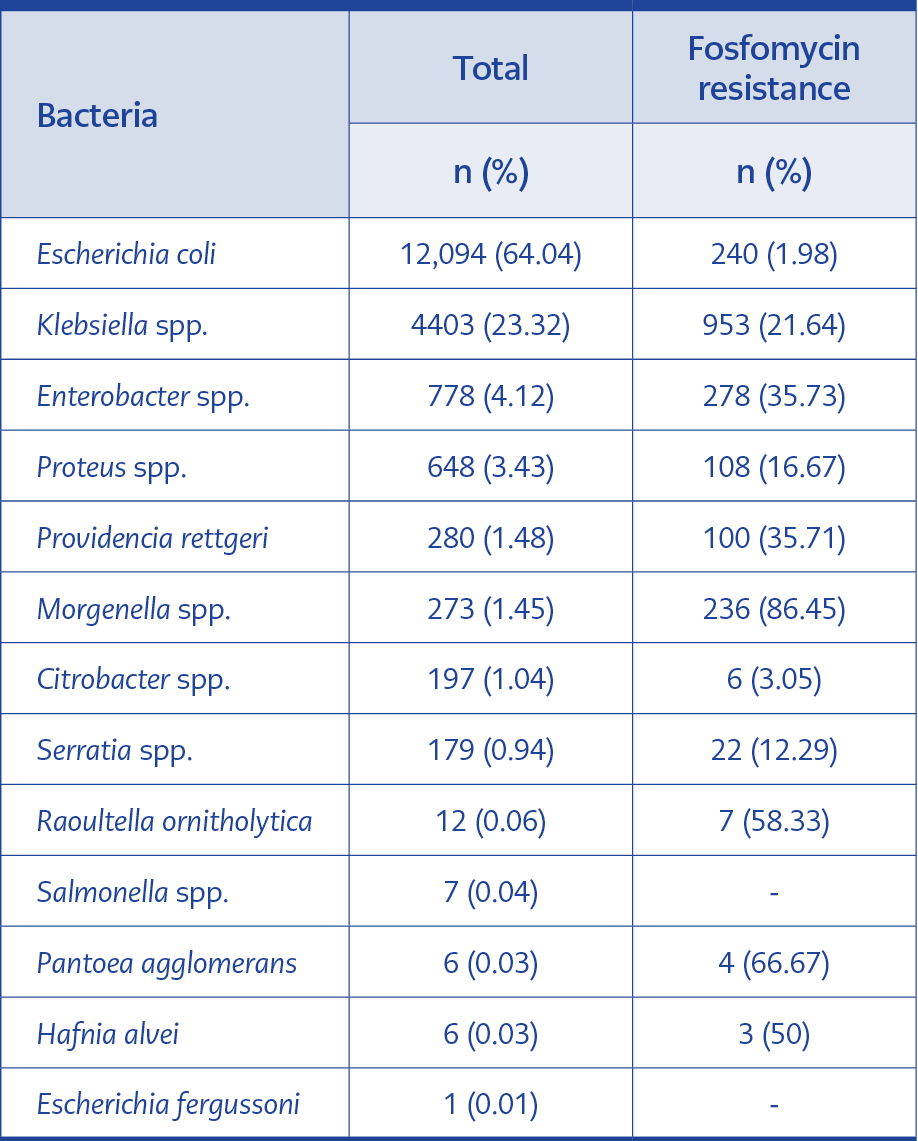
E. coli was isolated as the most common urinary pathogen (64.04%). Klebsiella spp. was the second most common causative agent over the years (23.32%). In total, the fosfomycin resistance rate in E. coli was 1.98%, 21.64% in K. pneumoniae, 35.73% in Enterobacter spp., and 16.67% in Proteus spp. The resistance ratio was 0% in Salmonella spp. (7 isolates over 9 years). The frequency and fosfomycin resistance rates of bacterial species are shown in Table 2.
29.31% of total isolates (n=5,534) were from outpatient clinics, and 70.69% (n=13,350) were from inpatient clinics. Fosfomycin resistance was 9.36% from isolates of outpatient clinics (n=518) and 10.78% of isolates from inpatient clinics (n=439). The results were statistically significant (95% CI, p<0.05). Fosfomycin resistance rates according to clinics are shown in Table 3.
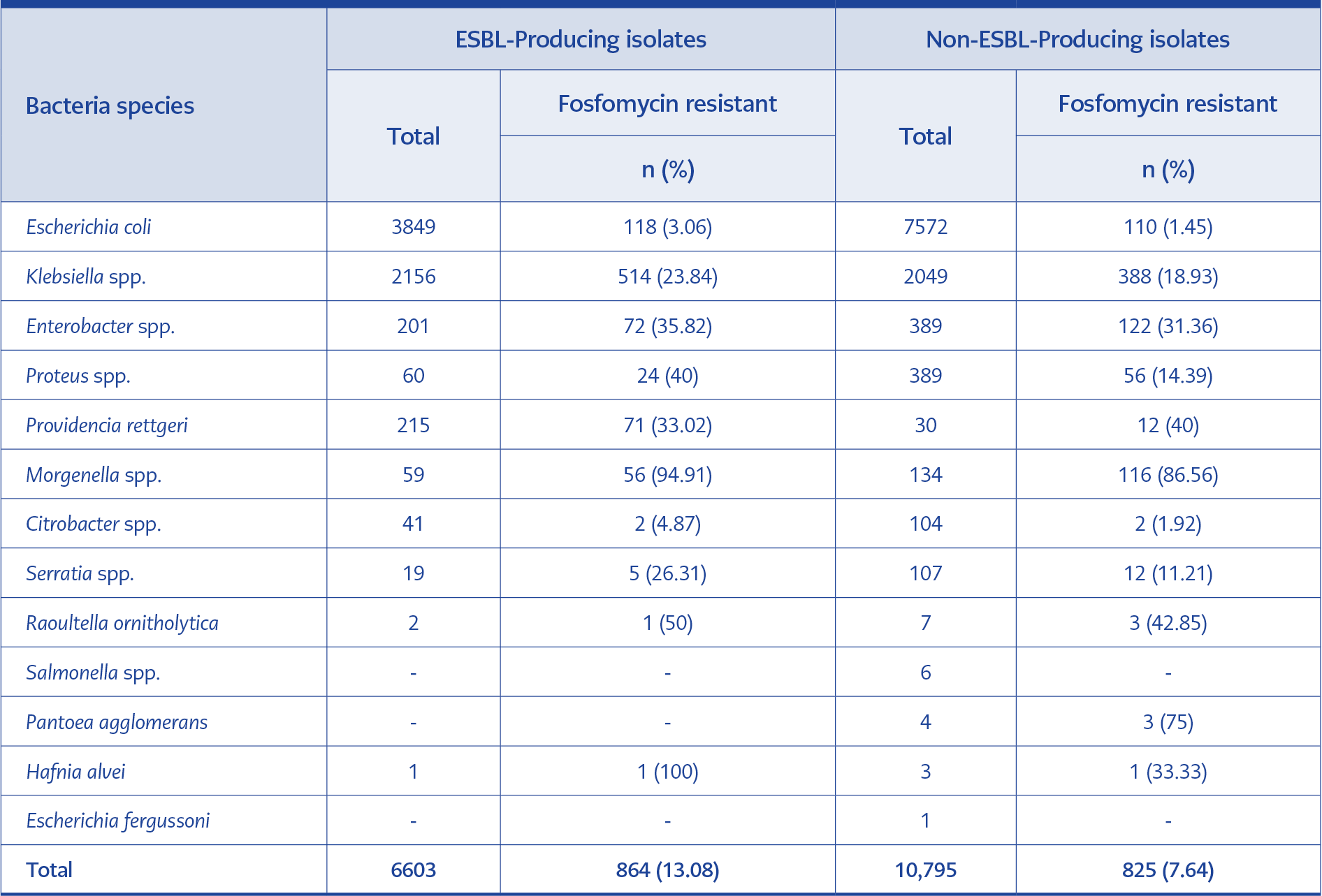
Total of 18,884 isolates, 17,398 (92.13%) were tested for ESBL with VITEK® 2 Compact (bioMérieux, Marcy l’Etoile, France). Moreover, 34.97% of the total isolates (n=6,604) were ESBL-producing. The fosfomycin resistance rate of ESBL-producing isolates was 13.08%, and fosfomycin-resistant non-ESBL-producing isolates were 7.64%. The fosfomycin resistance was 3.06% among ESBL-producing E. coli and 1.45% among non-ESBL-producing E. coli. The fosfomycin resistance among ESBL-producing Klebsiella spp. and non-ESBL-producing Klebsiella spp. was 23.84% and 18.93%, respectively. The relationship between ESBL production and the fosfomycin resistance of all bacterial species is shown in Table 4.
Discussion
Recent guidelines recommend fosfomycin for empirical treatment of uncomplicated UTIs (7, 8). However, this situation may lead to an increase in fosfomycin resistance. In addition, fosfomycin may be a choice for multidrug-resistant bacteria such as vancomycin-resistant enterococci (VRE), methicillin-resistant S. aureus (MRSA), and ESBL–producing Gram-negative bacilli (7). Therefore, this study investigated the frequency of bacterial species, fosfomycin resistance rates, and the relationship between ESBL-producing and fosfomycin resistance in uropathogenic Enterobacterales isolates.
Our study found the most common urinary pathogen as E. coli (64.04%), followed by Klebsiella spp. (23.32%) inconsistent with previous studies (2, 3). The majority of the cultures were from females (63.88%), similar to previous studies (15). Our results showed statistically significant increases in fosfomycin antibiotic resistance rates between 2012 and 2020 (from 2.86% to 14%, p<0.05). This significant trend may be related to the use of fosfomycin effectively for more than 30 years (16). Our results are also consistent with a study that found an increase in fosfomycin resistance in ESBL-producing E. coli from 4.4% in 2005 to 11.4% in 2009 (17).
We found the fosfomycin resistance of E. coli as 1.98%, similar to a previous study from Turkey, which found 2% (18). In our study, fosfomycin resistance of Morganella spp. was found to be 86.45%. This result was lower than previous studies from Turkey and Iran, which both found 100% (18, 19); the cause may be the relatively low sample number of previous studies. The high sample size in our study allowed a more optimal assessment of fosfomycin resistance to rare bacteria. Fosfomycin resistance of Klebsiella spp. was found at 21.64%. This result is markedly higher than a study from Turkey, which found 6.9%, but in contrast, lower than a study from Switzerland, which found 40.6% in 2009 and 23.2% in 2016 (18, 20). Our study’s high rates of resistance may result from the fact that most patients were inpatients (70.69%).
In our study, a statistically significant difference in fosfomycin resistance between inpatients and outpatients was observed, which were 10.78% and 9.36%, respectively (p<0.05). The fact that fosfomycin resistance is significantly higher in inpatients is an expected result since these patients have more resistant bacteria and nosocomial infections than outpatients. Furthermore, these findings are consistent with a study that compares inpatient and outpatient fosfomycin resistance of E. coli, K. pneumoniae, and Proteus mirabilis (20).
In Greece, India, and Turkey, fosfomycin resistance among ESBL-producing Enterobacterales was reported at 1.9%, 8%, and 8.8%, respectively (18, 21, 22). In our study, fosfomycin resistance among ESBL-producing Enterobacterales isolates was higher (13.08%) than the results of the studies above. Also, a statistically significant difference in fosfomycin resistance between ESBL-producing and non-ESBL-producing isolates (p<0.05) was determined, especially in ESBL-producing Klebsiella spp. (23.84%) and ESBL-producing Enterobacter spp. (35.82%) compared to ESBL-producing E. coli (3.06%). Similar to our results, a study from Switzerland showed that 17 of 1225 (1.38%) ESBL-producing E. coli were resistant to fosfomycin (14). This may result from the fact that non-E. coli Enterobacterales, especially Klebsiella spp., tend to be higher resistant to fosfomycin (21). On the other hand, a study from Turkey showed that ESBL-producing Klebsiella spp. (73.3%) had a lower fosfomycin susceptibility rate than ESBL-producing E. coli (94.1%) (18).
Our study has some limitations. First, since it is single-centered, it is insufficient to represent the whole country. Furthermore, it is retrospective, and some data, such as the clinical status of the patients, prior antibiotic use, risk factors for infection with resistant bacteria, and whether the patients had asymptomatic bacteriuria, uncomplicated UTIs, or complicated UTIs, were not available and not included in the study.
In conclusion, fosfomycin seems a good option for effectively treating UTIs caused by E. coli. On the other hand, we found that fosfomycin resistance tends to increase over the years. Therefore, these findings should be considered, and further studies should be conducted to evaluate fosfomycin resistance.