Introduction
Lyme disease, also known as Lyme borreliosis, is the most common vector-borne disease in Europe and North America, caused by the spirochete Borrelia species, which can be transmitted through the bite of an infected black-legged tick (1). The Centers for Disease Control and Prevention (CDC) data on Lyme disease in the United States showed a fluctuation in annual cases, with the highest reported in 2017 at 42,743, the lowest in 2020 at 18,000, and a mean annual rate of approximately 29,393 cases during the five years from 2017 to 2021 (2). The number of estimated cases is much higher, and the annual number of patients diagnosed and treated for Lyme disease is approximately 476,000 (3, 4). Conversely, Belgium, Finland, the Netherlands, and Switzerland exhibited the highest Lyme borreliosis incidences (>100 cases per 100,000), while the Czech Republic, Germany, Poland, and Scotland reported rates ranging from 20 to 40/100,000, with lower rates (<20/100,000) observed in multiple other European countries. Additionally, subnational areas displayed markedly elevated incidences, reaching a peak of 464/100,000 in specific locales across Europe (5). In Turkey, the studies reported on Lyme disease are mainly case reports, including a limited number of patients (6). On the other hand, there are difficulties in the diagnostic process and surveillance level. Lyme disease diagnosis is based on a combination of factors, including symptoms, medical history, findings upon physical examination, and laboratory tests.
Dark field microscopy is a diagnostic technique that can be used for the detection of various microorganisms; however, imaging techniques, such as immunofluorescence staining or cell sorting of cell wall-deficient or cystic forms of Borrelia burgdorferi, are not currently recommended for the diagnosis of Lyme disease because of certain limitations (7). This study aimed to conduct a literature review on the utility of dark field microscopy as a diagnostic tool for Lyme disease to provide physicians with valuable insights into recommended diagnostic approaches aligned with national and international guidelines.
Clinical and Research Consequences
An elaborate search was designed and conducted to present an evidence-based approach for the laboratory diagnosis of Lyme disease using direct microscopic visualization techniques like dark field microscopy. A structured literature search was conducted based on the question “What is the exact role of dark field microscopy in diagnosing Lyme disease?” We searched Pubmed, Scopus, and Web of Science with the medical subject headings (MeSH) search terms, including Lyme borreliosis, Lyme disease, B. burgdorferi, diagnosis, and microscopy. In addition, we analyzed selected national and international guidelines to assess the recommended diagnostic approach for Lyme disease. We did not filter the date field and included all types of articles. Studies that did not have an eligible full text, such as those that did not provide information on the use of dark field microscopy for diagnostic purposes in the context of Lyme disease, were excluded.
Dark Field Microscopy in the Diagnosis of Lyme Disease
Several publications were investigated in detail for information about the clinical manifestations and recommended approaches for diagnosing Lyme disease. The consensus statement of Spanish scientific societies points out that there are stains to demonstrate the presence of spirochetes in tissues. However, only immunohistochemistry is mentioned to be specific and could lead to the direct diagnosis via molecular biology techniques (8). According to the European Society of Clinical Microbiology and Infectious Disease (ESCMID) Study Group on Lyme borreliosis (ESGBOR), molecular methods can be used for the detection of Borrelia as supplementary diagnostic methods for particular indications, and the visual contrast sensitivity test cannot be recommended for diagnosis due to low specificity (9). Dark-field or phase-contrast microscopy is not recommended for Lyme borreliosis because of a lack of sensitivity and specificity in guidelines from the French scientific societies and CDC (7, 10, 11).
A previous systematic review of ‘direct microscopy of human tissues’ emphasized that the modified dark-field microscopy technique should not be used for diagnosis and Borrelia detection by microscopy can only be used for research purposes (12). Laane et al. performed a modified dark field microscopy technique with a 66% (21/32) positivity for Borrelia in blood samples of patients with non-specific symptoms (13). On the other hand, Aase et al. revealed an 85% false positivity for Borrelia and/or Babesia among 41 healthy controls and a 66% positivity in the patient group that had previously supposedly tested positive for Borrelia or Babesia by the microscopy method (14). In addition, the structures interpreted as Borrelia and Babesia by this method could not be confirmed by the polymerase chain reaction (PCR) method. Therefore, the modified dark field microscopy method was determined to be invalid and unfit for clinical use (14).
Summarized Recommendations in the Diagnostic Process for Lyme Disease by Several National and International Guidelines
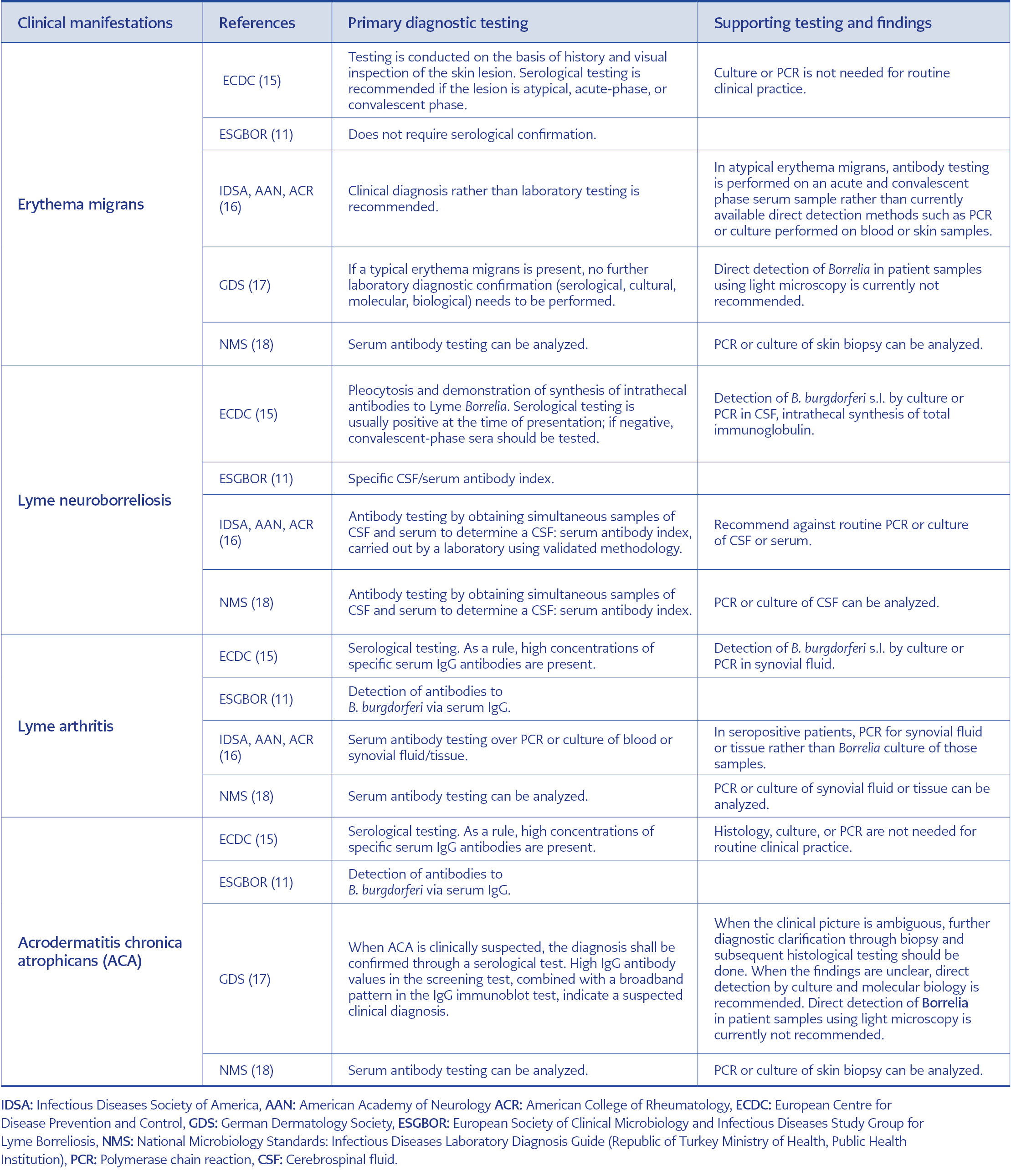
The evidence-based guidelines and the CDC currently recommend the two-tier serology, which is first based on an immunoenzymatic technique (ELISA) and then, if positive or equivocal, on a confirmatory immunoblot test (western blot, WB) for the laboratory diagnosis (15-18), so-called standard two-tier testing. In addition, the U.S. Food and Drug Administration (FDA) cleared several serologic assays, allowing for an enzyme immunoassay (EIA) rather than WB as the second test in the testing algorithm (19), referred to as modified two-tier testing. Selected national and international guidelines are presented in Table 1 regarding the recommended diagnostic approach for Lyme disease.
The diagnosis of Lyme borreliosis should be based on the patient’s epidemiological history (residence or recent travel to endemic areas, engagement in outdoor activities in high-risk environments, exposure to potential tick habitats, a history of tick bites, consideration of seasonal factors, and involvement in outdoor occupations), clinical symptoms and signs, and microbiological findings. Although seropositivity rates ranging from 2% to 44% have been reported for B. burgdorferi, the actual prevalence of Lyme disease is not fully understood in Turkey (20).
Patients in Turkey often receive non-recommended tests (such as dark field microscopy and lymphocyte transformation tests) during the diagnostic process because recommended diagnostic methods are not available in many centers. As a consequence, we came across a group of patients who were misdiagnosed with Lyme disease through dark field microscopy on a blood sample in which live Borrelia spirochaetes were supposedly observed, similar to the experiences of our colleagues in Norway (21). Therefore, we believe that this review will be helpful for our colleagues in correctly diagnosing Lyme disease, guiding treatment and management, and improving patient care of patients suspected of Lyme disease.
Branda et al. reviewed the laboratory diagnosis of Lyme disease and emphasized that direct visualization of Borreliae in blood or other infected tissues easily leads to misinterpretation. Direct visual detection is less sensitive or practical than a first-line diagnostic or adjunctive test because of the low in vivo organism burden of primary tissue samples (22). Lohr et al. also reviewed the diagnostic utility of direct microscopy for Lyme disease and mentioned the limited clinical utility because of the sparseness of organisms in samples (23).
There are also several publications about spirochete detection by electron microscopy, silver staining with light microscopy, or focus-floating microscopy in various samples. However, these methods also had poor sensitivity and high rates of false positivities (12, 24-26).
The guidelines have recommended serologic tests as the primary diagnostic (erythema migrans, which is more a clinical diagnosis, excluded) approach using standard 2-tier testing or modified 2-tier testing. Emerging technologies using biomarkers may be helpful in early Lyme disease, but more data is needed to recommend these newer methods (27).
The literature clearly shows that the sensitivity of dark field microscopy is low, and it may produce false negative and positive results. In addition, factors such as the technician’s expertise and the specimen’s quality should also be considered. Therefore, direct detection of Borrelia from patient material using dark field or focus floating microscopy is not recommended for diagnostic purposes (28).
In terms of the limitations of our study, it was conceived as a comprehensive review rather than a systematic one, and not every guideline pertaining to Lyme disease could be encompassed. The selected guidelines were succinctly summarized, highlighting recommended and unrecommended tests based on the clinical presentation in the diagnostic process of Lyme disease.
Conclusion
Clinicians should be aware of the diagnostic tests’ sensitivities and specificities and combine them with the patients’ epidemiologic factors and clinical signs and symptoms to accurately diagnose Lyme disease. Based on our findings, dark field microscopy is not recommended as a diagnostic method for Lyme disease due to low sensitivity and high false positivity rates.