Introduction
Coronavirus disease (COVID-19) has caused more than 660 million cases and more than six million deaths worldwide (1). COVID-19 can cause different clinical situations ranging from asymptomatic to life-threatening infections. Patients with severe COVID-19 may have severe pulmonary involvement leading to respiratory failure and death (2, 3).
In addition, lymphopenia is a common feature of SARS-CoV-2 infections; it may predict severe and fatal cases of COVID-19 (4-8). Studies have shown that both helper T (CD4+) and suppressor T (CD8+) cells appear below normal levels in COVID-19 patients. In severe cases, the number of CD4+ T lymphocytes in total T cells decreases; hence the CD4+/CD8+ ratio may decrease. Therefore, in cases of COVID-19 ranging from asymptomatic to life-threatening clinical presentations, there is a need to define lymphocyte subgroups (9-12).
This study aimed to describe the clinical relationship between lymphocyte percentage and helper T cell levels and prognosis in patients with various severity levels of COVID-19.
Materials and Methods
Study Design and Patients
This prospective study was conducted in a tertiary hospital with 1653 beds and 243 intensive care beds. Adult patients (>18 years) hospitalized with COVID-19 between April and October 2020 were included. Demographic characteristics and comorbidities of the patients were obtained from the hospital records.
All patients tested positive for SARS-CoV-2 PCR. The patients were divided into three study groups. The first group consisted of ‘asymptomatic’ patients without any signs or symptoms; the second group consisted of ‘mild/moderate’ patients with signs of upper respiratory tract infection or infiltration in the lung but with a peripheral arterial saturation (pulse oximetry) >94% in room air. The third group included ‘severe/critical’ patients who met at least two of the following criteria: peripheral arterial saturation <94% needing oxygen support, tachypnea (>30/min respiratory rate), and more than 50% progression in lung radiology within 24-48 hours (13, 14).
Furthermore, we performed another analysis to estimate the mortality of the patients, excluding asymptomatic patients. In our hospital, a complete blood count, peripheral lymphocyte and subgroups by flow cytometry method, and C-reactive protein (CRP) by the turbidometric method were studied within 2 hours of admission. In order to determine the prognostic tests, the same examinations were repeated on the seventh day of the follow-up. We compared the results of laboratory tests performed during hospitalization and on the seventh day between surviving and non-surviving patients.
All symptomatic patients received supportive treatment and favipiravir. Patients diagnosed with cancer, receiving chemotherapy, having an autoimmune disease, using corticosteroids for chronic disease, or pregnant were not included in the study. In addition, patients who died or were discharged before the 7-day hospital stay and who used corticosteroids for COVID-19 were excluded from the study.
Flow Cytometry
We performed a cellular analysis with 3 ml of peripheral blood samples collected at diagnosis and in the first week of treatment. The lymphocyte subtype analysis was performed by using specific anti-human antibodies for the T-cells subtype; CD3-FITC, CD4-APC, CD8-PE, CD45-PerCP and for B-cell CD19 -APC-Cy7 (BD Multitest™, BD Biosciences, San Jose, CA, USA) with the BD FACSCanto™ II flow cytometry system (BD Biosciences, San Jose, CA, USA).
Statistical Analysis
Statistical analyses were performed using SPSS 22.0 (IBM Corp., Armonk, NY, USA). Parametric variables were determined using one-way ANOVA and Tukey’s post-hoc test. Logistic regression analysis between-group significance was performed, and odds ratios (ORs) and 95% confidence intervals (CIs) were presented. The receiver operating characteristic (ROC) curve model was also used to predict death efficacy. The statistical significance was set as p<0.05.
Results
Demographic and Clinical Characteristics
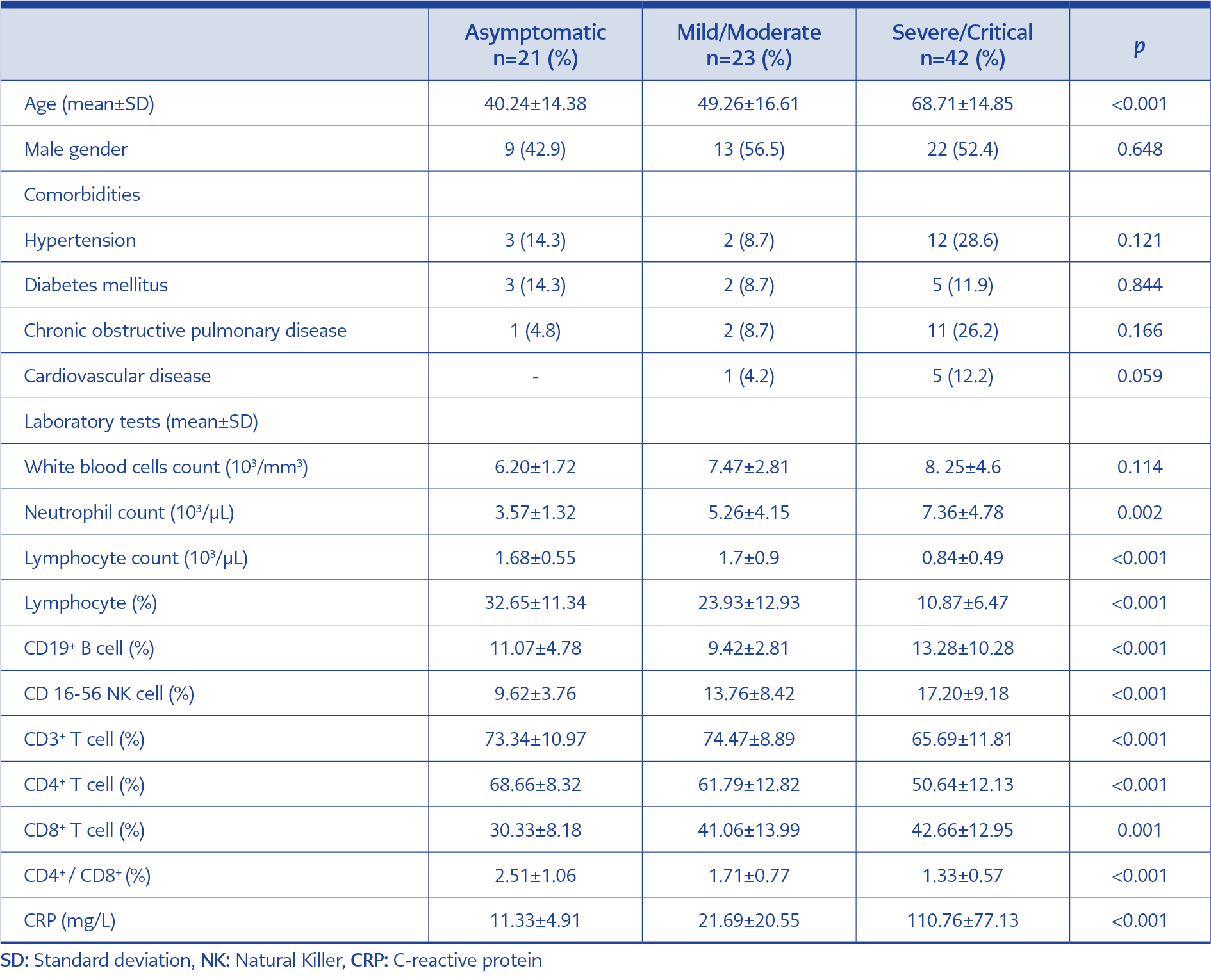
A total of 86 patients were included in the study. Twenty-one of the patients (24.4%) had asymptomatic, 23 (26.7%) mild/moderate, and 42 (48.8%) severe/critical COVID-19. The median age of patients (±SD) was 56.56 (±19.48), and 51.2% were male. The mean age in the severe/critical group was statistically higher than in the mild/moderate (p<0.001) and asymptomatic groups (p<0.001). Gender (p=0.648) and comorbidities were not significantly different between severity groups (p>0.005). The most common comorbidity was hypertension (19.8%). Demographic and laboratory data of the patients are presented in Table 1.
Severity of COVID-19
The age difference, lymphocyte subgroups, and CRP between the severity groups are shown in Figure 1 and Table 1. The white blood cell levels were similar in all groups (p=0.114). However, the absolute neutrophil count of the severe/critical group was higher than the mild/moderate (p=0.008) and asymptomatic COVID-19 groups (p=0.001).
The percentage of lymphocytes and subgroup analysis in 86 patients with various severity levels of COVID-19 were calculated after flow cytometry (Table 1). The mean lymphocyte count was lowest in the severe/critical group (p<0.001) (Table 1). Lymphocyte percentage was lowest in the severe/critical group and highest in the asymptomatic group (10.87±6.47 vs. 32.65±11.34) (p<0.001). The difference between the lymphocyte percentage value in the asymptomatic and the mild/moderate groups was also statistically significant (p=0.011).
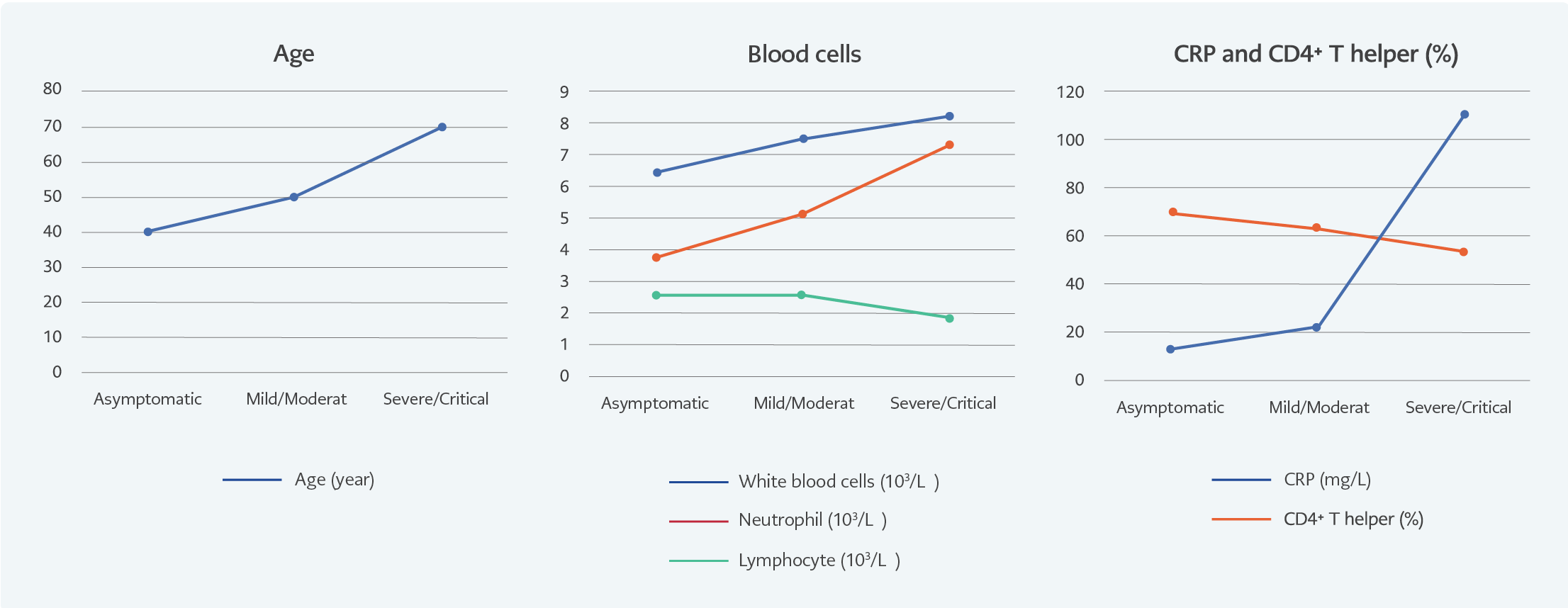
Figure 1. Age, blood cell, CRP, and CD4+ percentage according to various severity levels of COVID-19.
The lymphocyte subgroup analysis showed that the percentage of CD4+ T helper was the lowest in the severe/critical group (50.64±12.13) and highest in the asymptomatic group (68.66± 8.32) (p<0.001). The CD4+/CD8+ ratio was the lowest in the severe/critical patients. According to the post hoc test, it was significantly different from the mild/moderate group (p=0.003) and the severe/critical group (p<0.001). CRP levels also increased with disease severity (p<0.001) (Table 1 and Figure 1).
Mortality of COVID-19
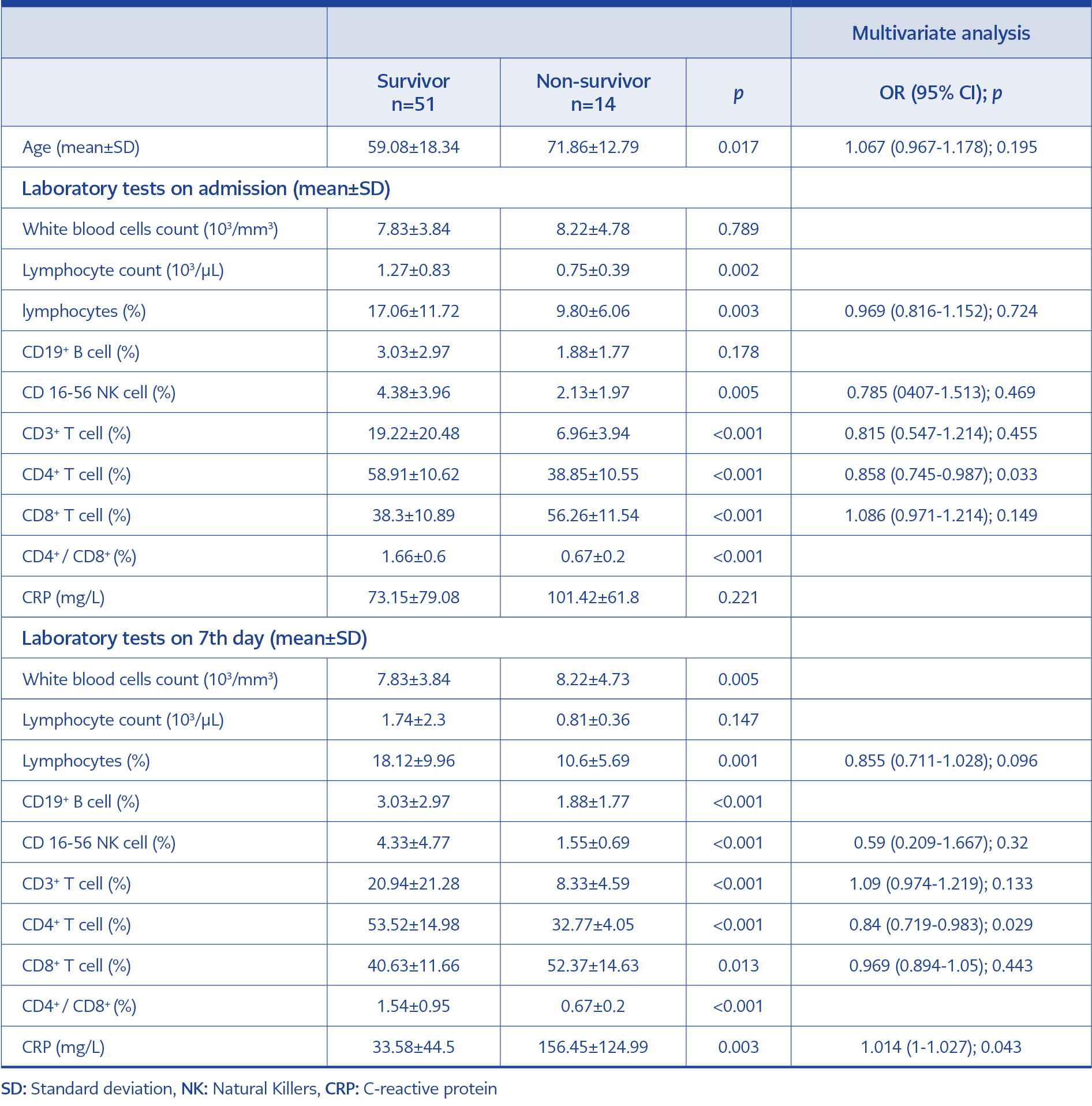
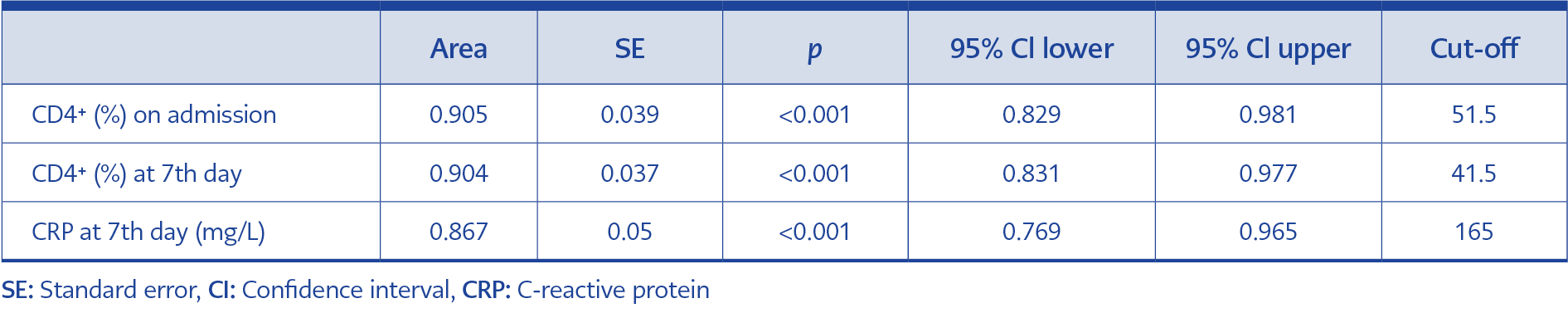
Table 2. Risk factors predicting mortality from laboratory values at admission and on the seventh day.
Sixty-five patients were included in the subgroup analysis. In the 14-day mortality assessment, 14 (21.5%) patients were non-survivors, and 51 (78.5%) were in the survivors’ group. Laboratory tests of survivor and non-survivor patients are presented in Table 2. The mean age (±SD) was 71.86±12.79 among non-survivors and 59.08±18.34 in survivors (interquartile range [IQR]=1.053; p=0.017). The CD4+ percentage on admission was lower in non-survivors than in survivors. According to multivariate regression analysis, a low CD4+ on admission and the seventh day and a high CRP on the seventh day were the factors predicting mortality (Table 2).
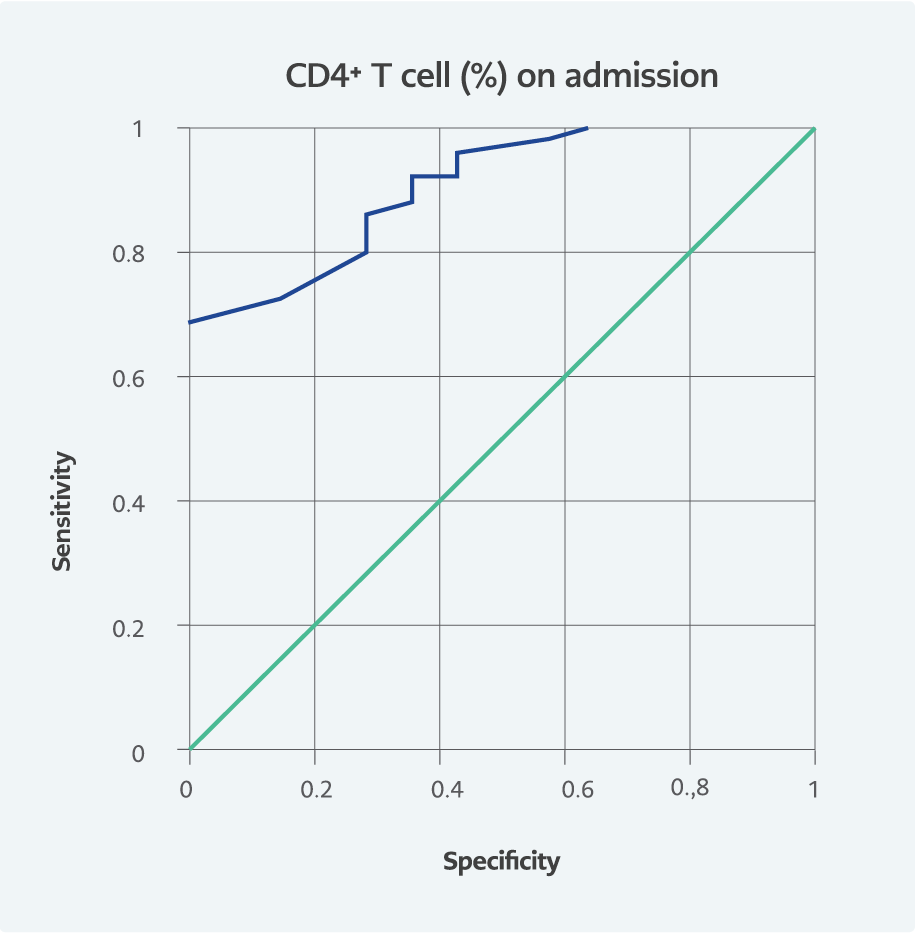
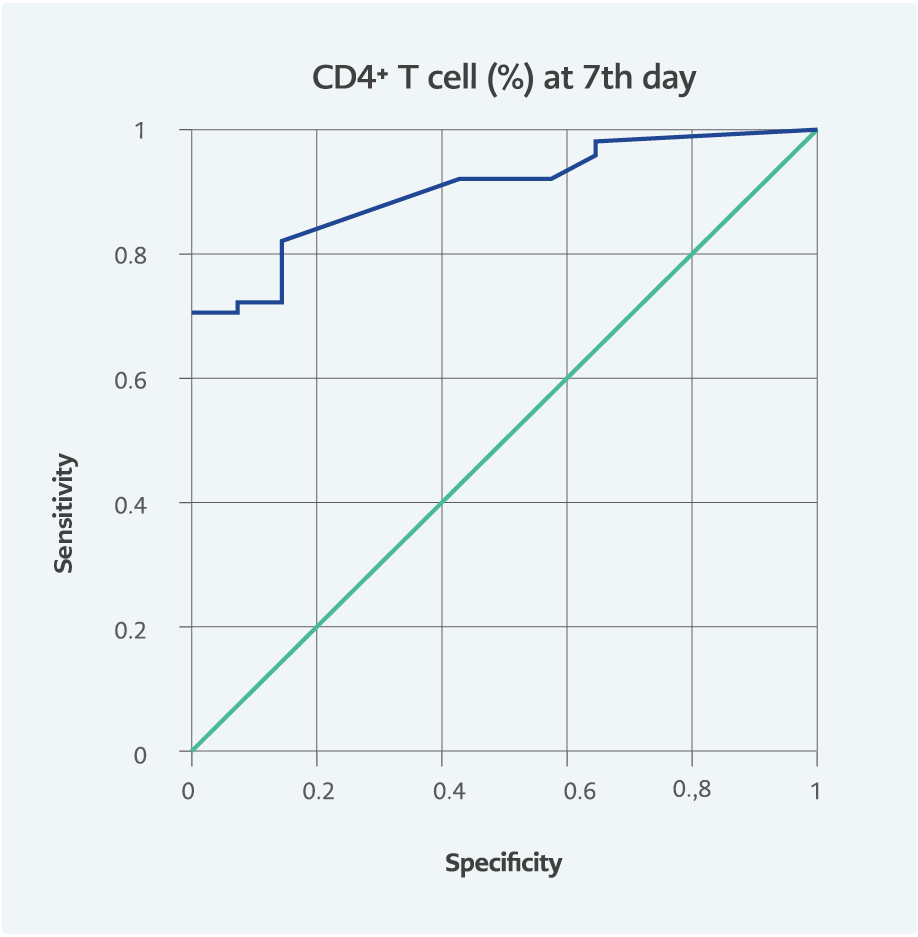
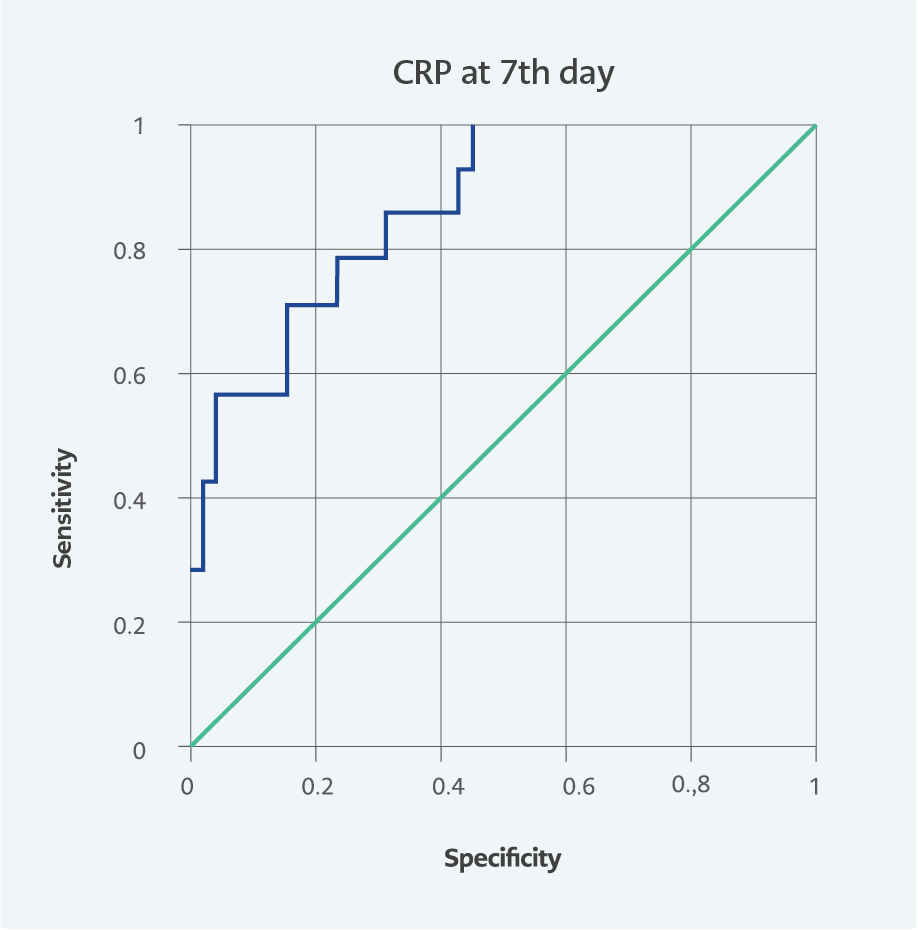
We also performed a ROC curve analysis to evaluate the role of these factors in predicting mortality efficiency. The area under the ROC curve (AUC) is presented in Table 3 and Figures 2, 3, and 4. On admission, the AUC for CD4+ T cells was 0.905 (95% CI=0.829-0.981).
Discussion
This study focused on identifying lymphocytic subgroups in various clinics and predicting mortality in COVID-19 patients. Our patients with severe/critical COVID-19 had older ages and lower lymphocyte counts. Lymphocyte percentages and absolute counts were statistically significantly lower in severe/critical COVID-19 patients compared to mild/moderate and asymptomatic patients. The older age and lower CD4+ helper T cells ratio were also the most predictive factor for mortality.
Respiratory virus infections can affect the number and distribution of peripheral lymphocytes, and most patients develop lymphocytopenia during the acute phase of severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS). In addition, the degree of decrease in T cells was associated with the severity of the disease (15). Similarly, severe lymphopenia appears to be associated with severe COVID-19 and increased mortality (16, 17). Pan et al. showed that the number of CD4+ T cells and CD8+ T cells was significantly lower in intensive care unit (ICU) patients than in the non-ICU group. However, the results of CD19+ B and natural killer (NK) cells differed from each other (18). Wen et al. found that both low CD4+ T and CD8+ T levels were associated with mortality, but low CD4+ T helper was an independent risk factor for mortality (12). In our study, the CD4+ T cell ratio was significantly reduced in critical COVID-19 patients. According to the seventh-day test results, low CD4+ was a risk factor for mortality. According to ROC analysis, the AUC value for CD4+ T helper ratio was more significant than 0.9 in both on-admission and the seventh-day tests. It had a high predictive value for mortality on admission and during follow-up.
There are different results regarding the low CD4+/CD8+ ratio in the literature. In addition to being a prognostic factor for mortality, there may be a relative effect because of a low CD4+ ratio (16-20). According to our study, the CD4+/CD8+ ratio decreased proportionally from asymptomatic to severe clinical conditions.
In the literature, there is no consensus on the results of NK cell distribution in various severity levels of COVID-19 disease. According to Jiang et al., NK cell counts were lower in COVID-19 patients than in healthy individuals (20). On the other hand, S. Cantenys-Molina et al. found that the percentages of NK cells were higher in deceased patients (21). Furthermore, a prospective study showed that the NK cell count was higher in severe patients during the first week of illness but lower in the following weeks (22). According to our results, it was higher on admission in mild/moderate patients (10% vs. 2%; p<0.001). In addition, tests performed on the seventh day were lower in deceased patients than in survivors.
Older age has been identified as the most significant risk factor for COVID-19 mortality. Previous studies reported that older age was associated with an impaired specific immune response to SARS-CoV-2 infection, resulting in increased inflammation and poor COVID-19 outcome. In addition, age-related defects in T-cell and B-cell function might lead to inadequate suppression of viral replication and prolonged pro-inflammatory responses (23-26). We found that the mean age was higher in the severe/critical COVID-19 group, and those who did not survive with confirmed COVID-19 were older.
CRP is a biomarker that predicts severity and mortality in COVID-19 patients (27, 28). In their meta-analyses, Kazemi et al. (27) and Danwang et al. (28) showed that CRP levels were correlated with the clinical severity of COVID-19. According to our study results, CRP was the biomarker with the most significant difference between mild/moderate and severe/critical patients. In addition, the most important biomarker predicting mortality from laboratory values measured on the seventh day was CRP.
The relatively small number of patients in our study brings some limitations. In addition, this study may not provide precise data on patients infected with SARS-CoV-2 mutant strains because our patients were infected with the original Wuhan strain.
In conclusion, the percentage of CD4+ T helper is a strong prognostic factor in COVID-19 patients. Furthermore, the difference in the CD4+ ratio determines clinical severity and predicts mortality. Therefore, it can be used in clinical follow-up of COVID-19 patients in combination with CRP.