Introduction
SARS-CoV-2 is transmitted via the inhalation of infected droplets or through direct contact with contaminated surfaces. Aerosol-generating procedures can also lead to airborne transmission (1). Healthcare workers (HCWs) are at higher risks for infections than the general population because of their close contact with patients or infectious materials (2). According to the World Health Organization (WHO), approximately 14% of COVID-19 cases worldwide were reported among healthcare workers, as high as 35% in some countries (3). The rapid increase in the number of COVID-19 cases may result in more healthcare professionals becoming infected. Furthermore, the more healthcare professionals get infected, the more the health system is disrupted, and the effects of the pandemic become more severe (4). Previous studies on the COVID-19 exposure status of HCWs in Turkey are limited. Therefore, we aimed to evaluate the clinical characteristics of admissions in HCWs for COVID-19 and the exposure status of those admissions.
Materials and Methods
This retrospective study enrolled HCWs admitted to the COVID-19 outpatient clinic due to COVID-19 symptoms or exposed to COVID-19 patients between March 13 and August 31, 2020, at a university hospital in Ankara, Turkey. The occupational, clinical, laboratory and radiologic data were retrospectively obtained from their medical records. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist was used for the study design.
The WHO’s definitions for probable and confirmed cases were used in the diagnosis of COVID-19. A probable case is a suspected case with radiological findings suggestive of COVID-19, whereas a confirmed case is a person with laboratory confirmation of COVID-19, irrespective of clinical signs and symptoms [(5)]. Typical COVID-19 symptoms were recorded according to the Centers for Disease Control and Prevention criteria (6).
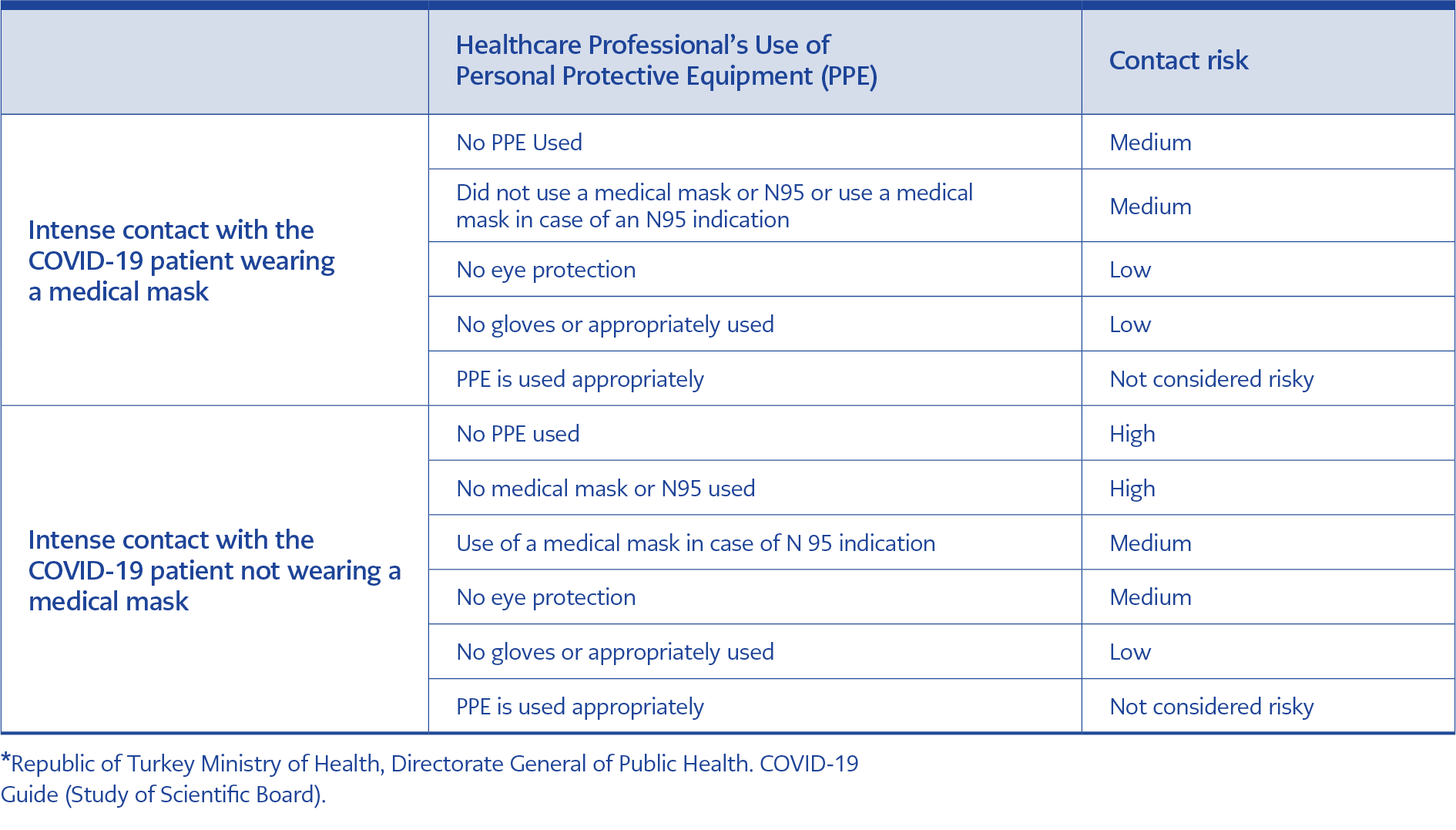
Combined nasopharyngeal and oropharyngeal swabs were obtained from symptomatic HCWs at the time of admission and from asymptomatic HCWs five or seven days after their exposure to a COVID-19 patient if their contact status were high/or medium. Exposure risk assessment was made according to the Republic of Turkey Ministry of Health COVID-19 Guide (7). Assessment of healthcare professionals’ contact status with COVID-19 patients is shown in Table 1. Laboratory confirmation was made based on a positive polymerase chain reaction (PCR) test.
SARS-CoV-2 RNA was extracted using the viral nucleic acid extraction buffer (vNAT®, Bioeksen R&D Technologies Inc., Turkey), and reverse transcription-PCR was performed using the Bio-Speedy® Direct RT-qPCR SARS-CoV-2 nucleic acid detection kit (Bioeksen R&D Technologies Inc., Turkey).
All data were analyzed using the IBM SPSS Statistics for Windows, Version 25.0. (Armonk, NY: IBM Corp.). The chi-square test was used to analyze categorical variables. Continuous numerical variables were not normally distributed and thus are presented as median values and were compared using the Mann-Whitney U test. Multivariate analysis was performed to assess the risk of infection. Logistic regression was performed by including all the variables; a p-value <0.05 was considered statistically significant.
Results
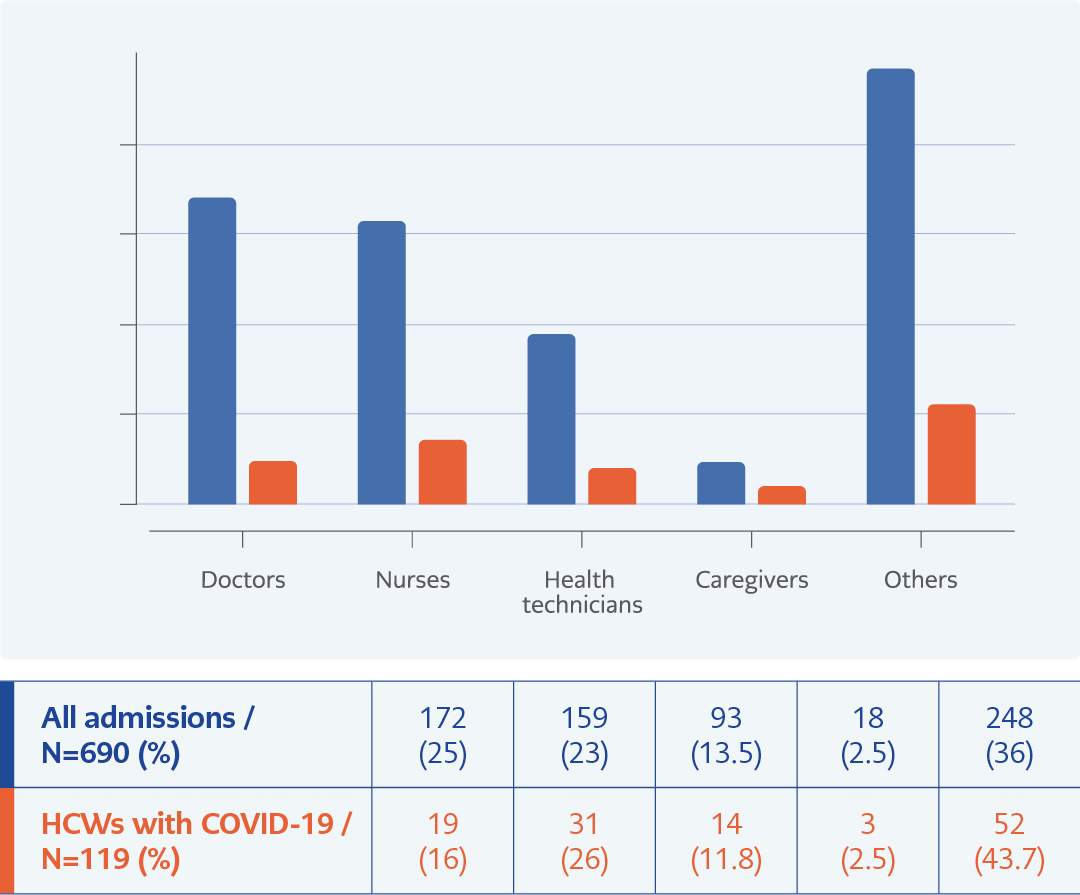
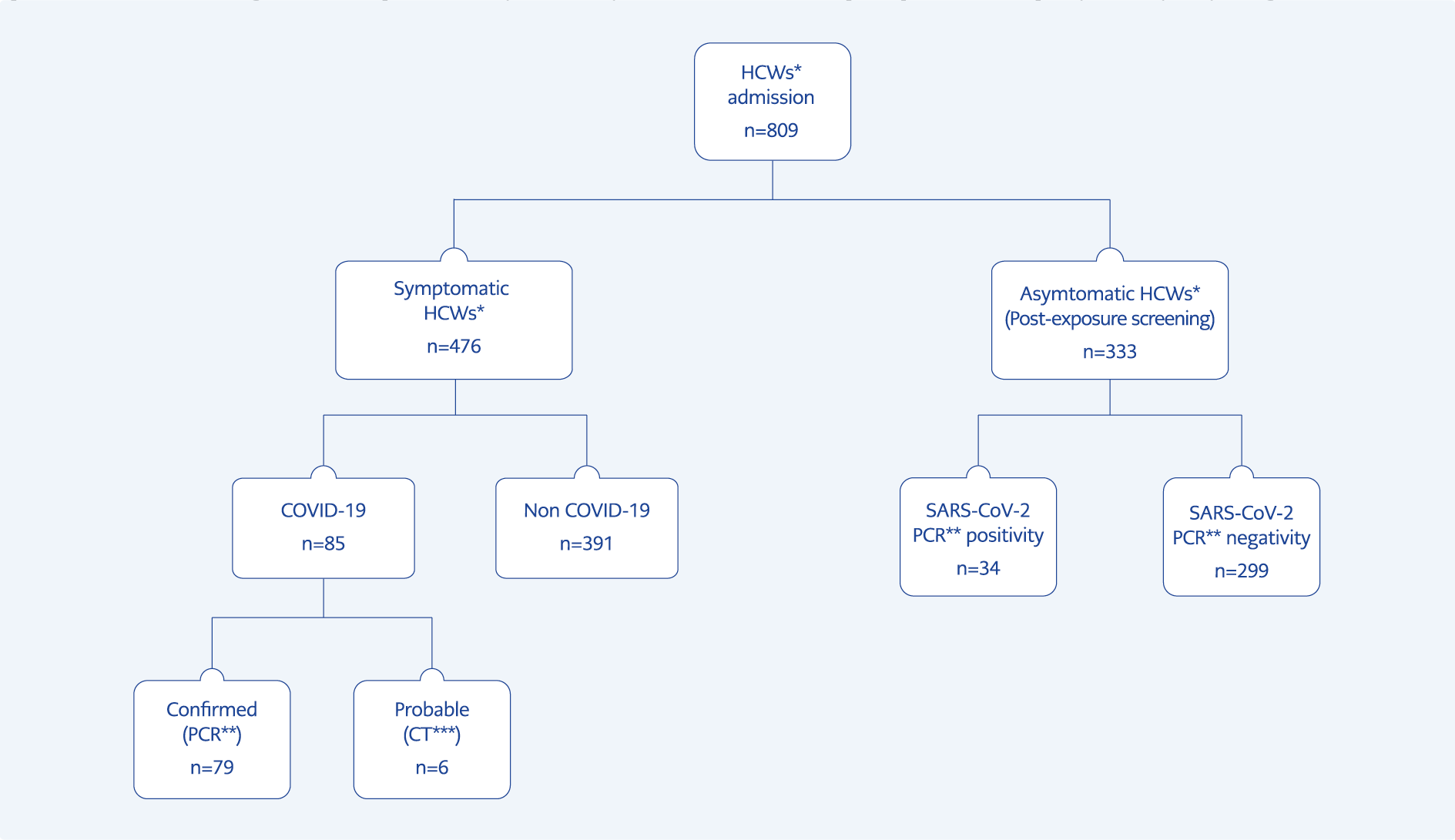
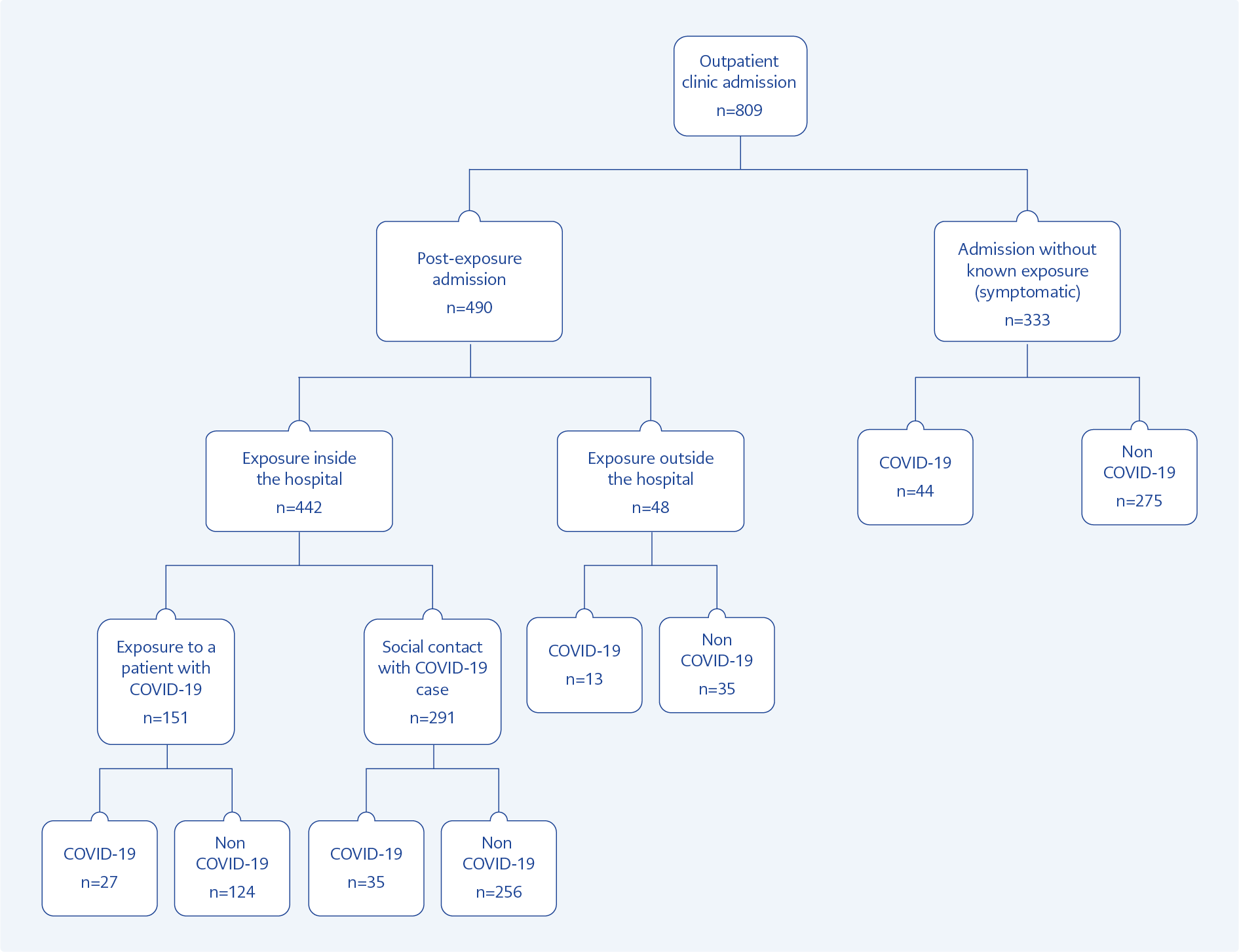
Overall, 809 admissions of 690 HCWs were analyzed in this study. The female to male ratio was 1.7:1. The median age was 33 years (interquartile range [IQR] 26-42 years). Among the HCWs, 25% were doctors, and 23% were nurses. The distribution of occupations is shown in Figure 1. In total, 476 HCWs (59%) had typical COVID-19 symptoms during admission, of whom 119 (25%) were diagnosed with COVID-19. There were 113 confirmed cases and six probable cases (Figure 2). Among infected HCWs, 24% (n=28) worked in departments where COVID 19 patients are primarily followed as emergency room, intensive care unit, inpatient and outpatient COVID-19 departments. A control PCR test was performed in 103 patients, and the median time for negative PCR test results was ten days (IQR 7-14 days). Characteristic radiological features on chest computed tomography were found in 33% of the COVID-19 cases and 29% confirmed cases. Among HCWs diagnosed with COVID-19, 13% required hospitalization. There was no intensive care unit (ICU) admissions and no deaths.
During the study period, 64% (n=443) of all HCWs at our hospital were exposed to a COVID-19 patient at least once. Although exposure history was significantly more common among doctors (78%; P=0.001) than other HCWs, SARS-CoV-2 positivity was detected only in nine doctors (7%) after a suspected exposure. There were no statistically significant differences among nurses, health technicians, caregivers, or other HCWs (p=0.718, 0.946, 0.212 and 0.286, respectively).
There were 490 admissions owing to a suspected exposure either inside (the high- and medium-risk groups) or outside (close contact) the hospital. In 32% of admissions, typical COVID-19 symptoms were observed, and 15% developed COVID-19 after exposure. The exposure rates were 90% and 10% inside and outside the hospital, respectively. The risk of COVID-19 development after suspected exposure outside the hospital (family member or social contact) was 2.3 times higher (27%) than that after exposure inside the hospital (14%), and the difference was statistically significant (P=0.020 95% confidence interval [CI] 1.141-4.543). Inside the hospital, social contact with colleagues was (66%) more common than exposure to a patient (34%).
For exposures inside the hospital, the risk of COVID-19 development was 1.6 times higher after exposure to a patient than the risk after exposure through social contact with a colleague; however, the difference was not statistically significant (P= 0.095; 95% CI 0.923-2.749; (Table 2).
Furthermore, 15 of 27 cases were associated with exposure to two patients with severe COVID-19. One of these patients was in the emergency room and had received cardiopulmonary resuscitation; within 11 days, three doctors, three nurses, and two caregivers had positive PCR test results for SARS-CoV-2 despite the proper use of personal protective equipment (PPE). The second patient received care for four days in two different departments before COVID-19 was diagnosed; within 14 days, three doctors, four nurses, one caregiver, and one housekeeper became infected from this patient and they have been used PPE properly. The exposure status of the HCWs on admission is shown in Figure 3.
The distribution of occupations for suspected exposure rates of admissions was 31% doctors, 25% nurses, 12% health technicians, 2% caregivers, and 30% other HCWs, who did not have direct contact with patients. Doctors and nurses were more likely than other HCWs to be exposed to COVID-19 patients inside the hospital (P=0.001 and P=0.001, respectively), while others were exposed via a family member or through social contact via other relatives or friends (P=0.001). For doctors, infection development after exposure was statistically lower (P=0.001; 95% CI 0.128-0.547), whereas, for nurses, it was 1.5 times higher.
Inexperienced HCWs (<10 years of experience) had 1.3 times more exposure than experienced HCWs. Remarkably, the former was exposed to COVID-19 patients inside hospital 1.7 times more often than experienced HCWs (P=0.001; 95% CI 1.142-2.565). However, there were no differences in exposure rates between inexperienced and experienced HCWs through social contact. However, the rate of COVID-19 development was 1.5 times higher in experienced HCWs than in inexperienced HCWs (P= 0.20; 95% CI 0.938-2.517).
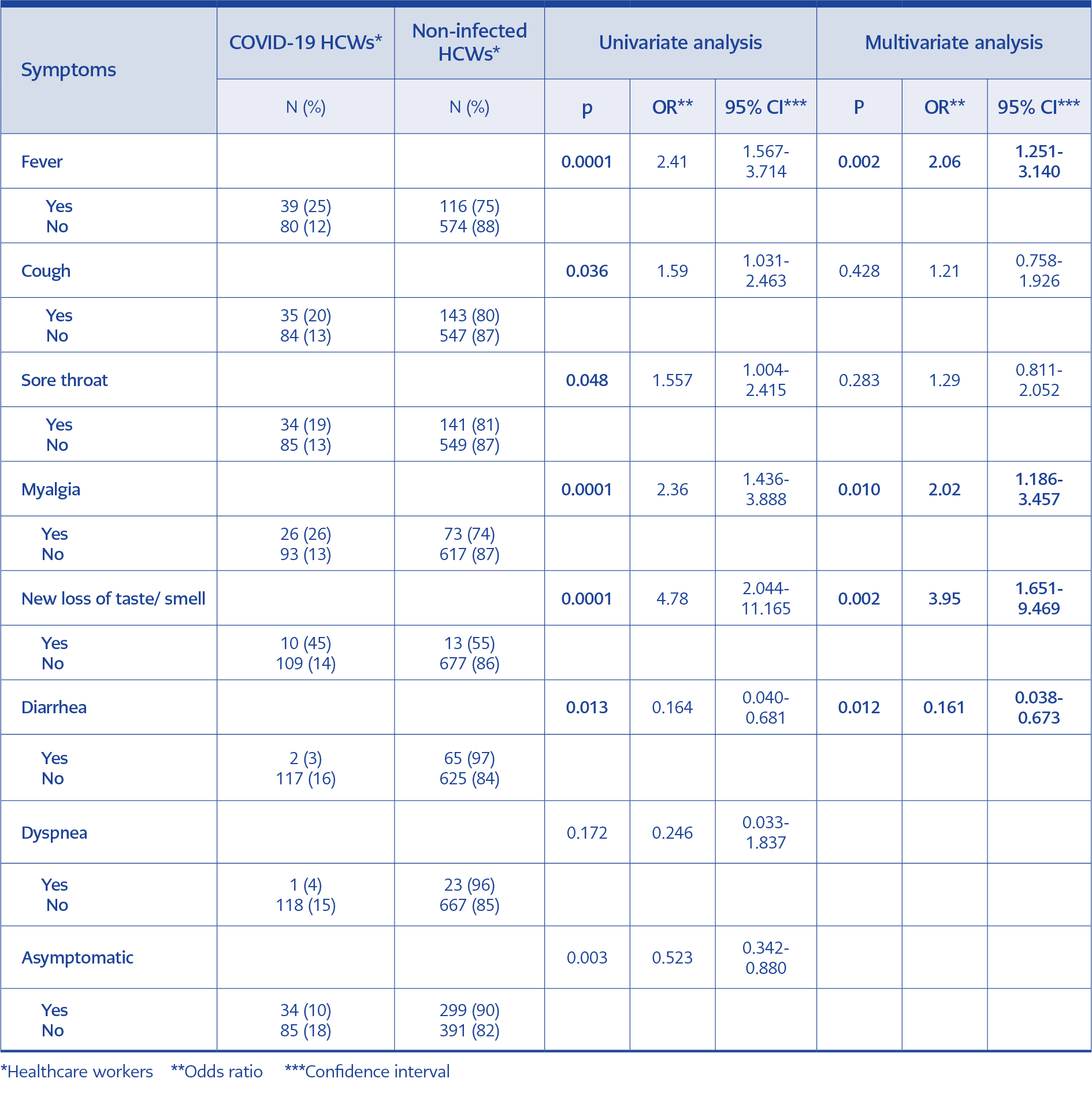
Among 809 admissions to the outpatient clinic, 476 patients (59%) had typical COVID-19 symptoms. The most common symptoms were cough (37%), sore throat (37%), and fever (33%). The rate of COVID-19 diagnosis was significantly higher in patients with loss of taste or smell (44%), myalgia (26%), fever (25%), cough (20%), and sore throat (19%) at admission (P<0.05). In multivariate analysis, loss of taste or smell, myalgia, and fever were detected as the predictors of COVID-19. However, diarrhea seemed to be an exclusion criterion for COVID-19. (Table 3). The PCR test for SARS-CoV-2 was positive in 10% of asymptomatic HCWs. The most common symptoms in HCWs diagnosed with COVID-19 were fever (33%), cough (30%), and sore throat (29%). Diarrhea (2%) and shortness of breath (1%) were less common symptoms.
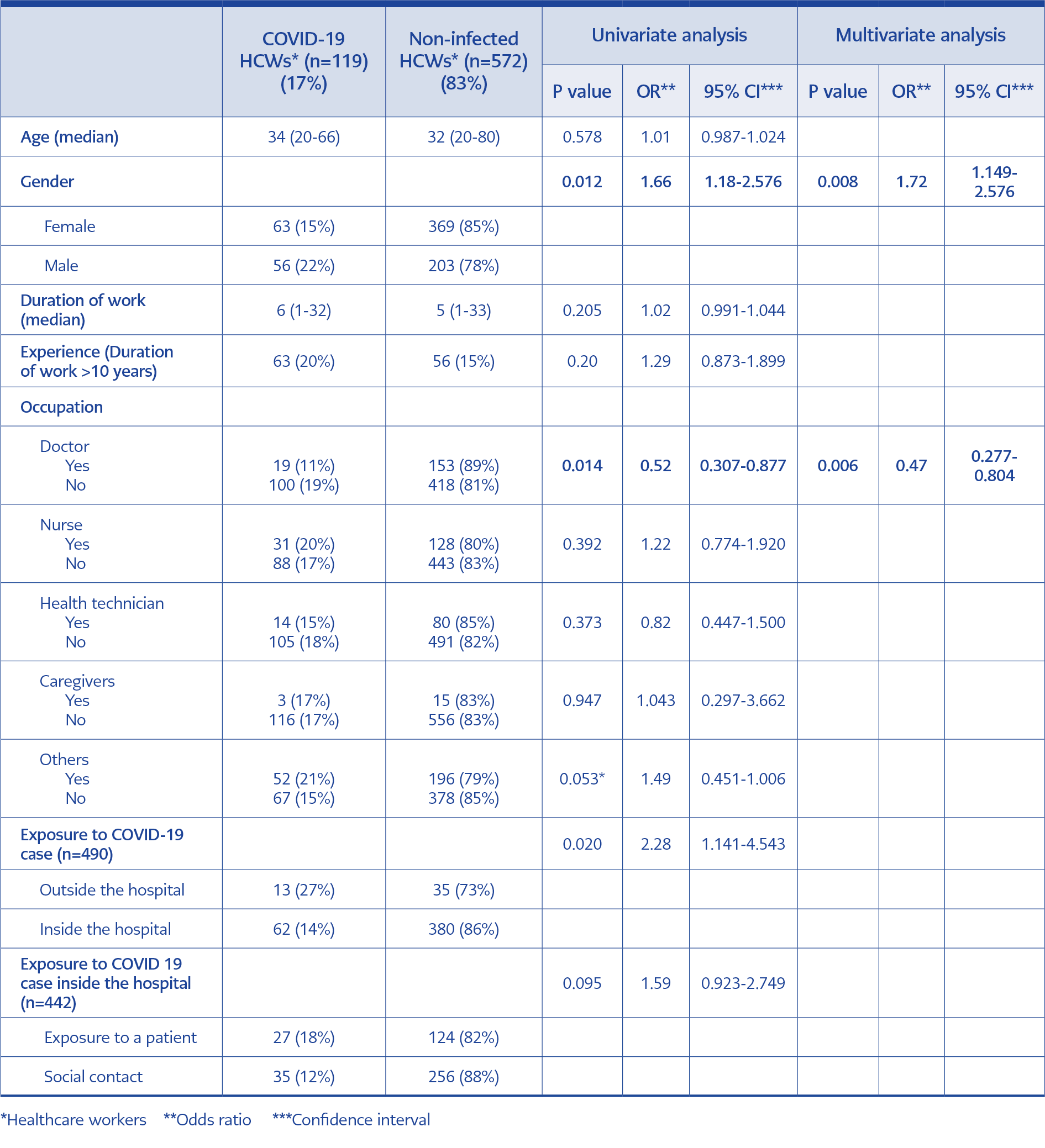
Factors such as male gender, being a doctor, and exposure to COVID-19 cases were significantly different between the group diagnosed with COVID-19 and the group not diagnosed with COVID-19 (P=0.012, P=0.014 and P=0,020, respectively). However, there were no differences with regard to years of experience (P>0.05). In addition, multivariate analysis revealed that male gender and occupation categories besides doctors were independent risk factors for the development of COVID-19 in HCWs (P<0.01; OR 1.72; 95% CI 1.149-2.576 and P<0.01; OR 0.47; 95% CI 0.277-0.804).
Discussion
We described the COVID-19, and its predictors among the HCWs admitted to the outpatient clinic with the suspect of infection. Seventeen percent of HCWs diagnosed with COVID-19 were admitted to the COVID-19 outpatient clinic, and they accounted for 16% of the total COVID-19 cases in our center. Although there are no published reports from Turkey, the Republic of Turkey Ministry of Health announced that >120,000 HCW PCR test results were positive for SARS-CoV-2 at the beginning of December 2020 [(8)]. According to the WHO’s report in September 2020, nearly 14% of COVID-19 cases globally were among HCWs, and this proportion may be as high as 35% in some countries (3, 9).
Admissions of female HCWs were more common in our study; however, unlike previous studies, COVID-19 diagnosis was higher in males than in female individuals (10-13). Nurses were the most infected group among different occupation categories, similar to previous study results (10, 13). Although admissions after suspected exposure were higher in doctors, infection rates were lower. Higher infection rates in nurses may result from longer duration and higher exposure rates to the patients. Furthermore, doctors may be assumed to have better education about COVID-19, resulting in appropriate attitudes and behaviors for protection both inside and outside the hospital (14).
Like recent studies, the most common symptoms among admitted patients were respiratory symptoms (13). However, loss of taste or smell, myalgia, and fever were predictors of COVID-19, and nearly half of the patients with these symptoms were diagnosed. Sudden loss of taste or smell is an important symptom for diagnosing COVID-19 in the early stage, especially in mild-to-moderate cases (15, 16). Therefore, it was recommended that during the COVID-19 outbreak, HCWs with a sudden loss of smell should isolate at home, practice social distancing, and undergo a SARS-CoV-2 diagnostic test as soon as possible (17). HCWs with diarrhea were mostly found to be SARS-CoV-2 negative. Although detailed analysis for causative agents was not performed, we postulate that other viral and bacterial diarrhea were expected to be common during our study period because of the seasonal properties of diseases (18, 19). Although most cases were mild, the hospitalization rate reported in this study was higher than that reported in other studies, likely because of the governmental approach for hospitalization indications (isolation to limit dissemination) at the beginning of the pandemic (10, 13). However, unlike other studies, there were no ICU admissions and deaths (10, 20).
Asymptomatic infection is another important source of SARS-CoV-2 transmission in healthcare facilities. We found positive SARS-CoV-2 PCR test result in 10% of asymptomatic HCWs. Numerous studies highlight the importance of screening and early detection in limiting the spread of the disease (21, 22).
Our main study finding was that the most important risk factor for SARS-CoV-2 infection in HCWs in the early phase of the outbreak was suspected exposure to COVID-19 cases outside the hospital, similar to other studies (23, 24). Among HCWs admitted to the COVID-19 outpatient clinic, 61% had suspected exposure inside or outside the hospital, of whom 16% got infected with COVID-19, which is a higher percentage than those reported in other studies (25). The highest test positivity rate was detected after social or familial contact outside the hospital. This could be related to HCWs ignoring social distancing guidelines and personal protection protocols in social or household settings outside of patient care areas (25). A study from Singapore reported that in the initial SARS-CoV-2 outbreak phase, community transmission, rather than healthcare-associated transmission, predominated (26).
While most studies reported that the risk of transmission inside the hospital is higher in breakrooms, nursing stations, and other social areas, our study showed that the risk of COVID-19 development was higher after exposure to a patient rather than after social contact. Most HCWs infected after patient exposure were associated with two index cases. These two clusters constituted 55% of the total cases of patient exposure. Adam et al. reported that 19% of SARS-CoV-2 infections in Hong Kong were responsible for 80% of all transmissions, highlighting the significance of superspreading events in the COVID-19 pandemic (27). Yuki et al. analyzed 61 clusters among various communities in Japan and found that 30% were related to healthcare facilities (28). Many studies have reported SARS and the Middle East respiratory syndrome coronavirus outbreak clusters (26, 29).
In our study, only a quarter of the infected HCWs were working in departments where COVID-19 patients are primarily. These data emphasized that all HCWs, not only working in departments where COVID-19 patients are primarily followed, are at risk of SARS-CoV-2 exposure. Therefore, training on complying with infection control measures and the appropriate use of PPE for all HCWs is crucial. Many studies have reported that the transmission rate of SARS-CoV-2 is higher in areas not considered high risk for COVID-19 (13, 14, 25). Work experience is another factor in the exposure rate. In our study, exposure to a COVID-19 case inside the hospital was higher among inexperienced HCWs (<10 years of work experience); however, the development of infection was 1.7 times higher in the more experienced group. Zhang et al. reported that frequent participation in training on COVID-19 and PPE use negatively correlates with years of experience and frontline status (14).
Our study was limited in that we did not question the participants about their underlying medical conditions. We also could not compare our data with local data because there have not been many studies from Turkey.
In conclusion, HCWs are at higher risks of COVID-19 than other members of the community because of the exposure rate. Ideally, after suspected exposure to a COVID-19 case, HCWs should be screened for SARS-CoV-2 and monitored closely for symptom development. These risks can be minimized by continuous training, strict infection control protocols, and appropriate PPE use both inside the hospital and in social or household settings.