Introduction
Hospital-acquired respiratory viral infections are associated with increased mortality and morbidity and prolonged hospital stays (1). Hospital-acquired influenza (HAI) accounts for 10-15% of these infections (2-4). Previous studies reported the proportion of HAI to influenza-diagnosed patients admitted to the hospital between 4%-25% (5-8). A systematic review showed that the average proportion of HAI among all hospitalized influenza cases was 11.3% (95% confidence interval [CI]=5.1-19.5) (1). These studies were done using varying diagnostic definitions for HAI and in the context of various vaccine uptake rates.
The level of influenza vaccine coverage in patients and healthcare workers is an important determinant of HAI (9,10). Vaccine uptake in Türkiye varies between 5.9%-27.3% (11). A single-center study from Türkiye involving 95 hospitalized influenza patients reported that none of the patients were vaccinated (12). A multicenter study conducted in Türkiye showed that influenza type A, older age, and 1-day delay in antiviral use predicted case fatality in hospitalized community-acquired influenza (CAI) patients. This study also reported that the vaccination rate for the study population was only 4% (13).
The full extent of the impact of HAI on patient outcomes, including morbidity and mortality, in an area of low vaccine uptake, especially when compared to CAI, is not well understood. Understanding these outcomes may point to opportunities for improvement in the effectiveness and efficiency of patient care.
This study aimed to compare 28-day mortality between patients who have HAI and those who have CAI during the 2017-2019 influenza seasons in a tertiary care center in İstanbul, Türkiye.
Materials and Methods
Study Design and Patients
This single-center retrospective cohort study was done in the adult wards of a 650-bed tertiary care university hospital in İstanbul between October 2017 and March 2020. The Clinical Research Ethical Committee of Marmara University approved the study with decision number 09.2021.1409 on December 3, 2021.
Nasopharyngeal specimens from patients were tested with the BioFire film array respiratory panel (RP) (BioFire Diagnostics, Inc., USA). This panel is a fully automated microarray-based multiplexed PCR technique that detects 14 viral agents: adenovirus (AdV), coronaviruses (CoV) (OC43, NL63, 229E, HKU1), influenza A (Flu A) (H1, H1-2009, H3), influenza B (Flu B), human metapneumovirus (hMPV), parainfluenza (PIV) 1-4, human rhinovirus/enterovirus (RV/EV), respiratory syncytial virus (RSV), and three bacterial agents: Mycoplasma pneumoniae, Chlamydophila pneumoniae, and Bordetella pertussis.
All hospitalized patients who had confirmed influenza virus infection over 17 years old were included in the study. Influenza confirmation was defined as a positive real-time reverse transcriptase-polymerase chain reaction (RT-PCR) assay for the influenza genome detected in a naso- or oropharyngeal swab specimen. The attending clinician made decisions about testing for influenza and admitting to the hospital.
A negative influenza test or no influenza-like illness signs on admission but a positive test result at any point after 72 hours of admission was defined as HAI (Figure 1). A positive influenza test within 72 hours of admission or earlier was defined as CAI.
Measurements
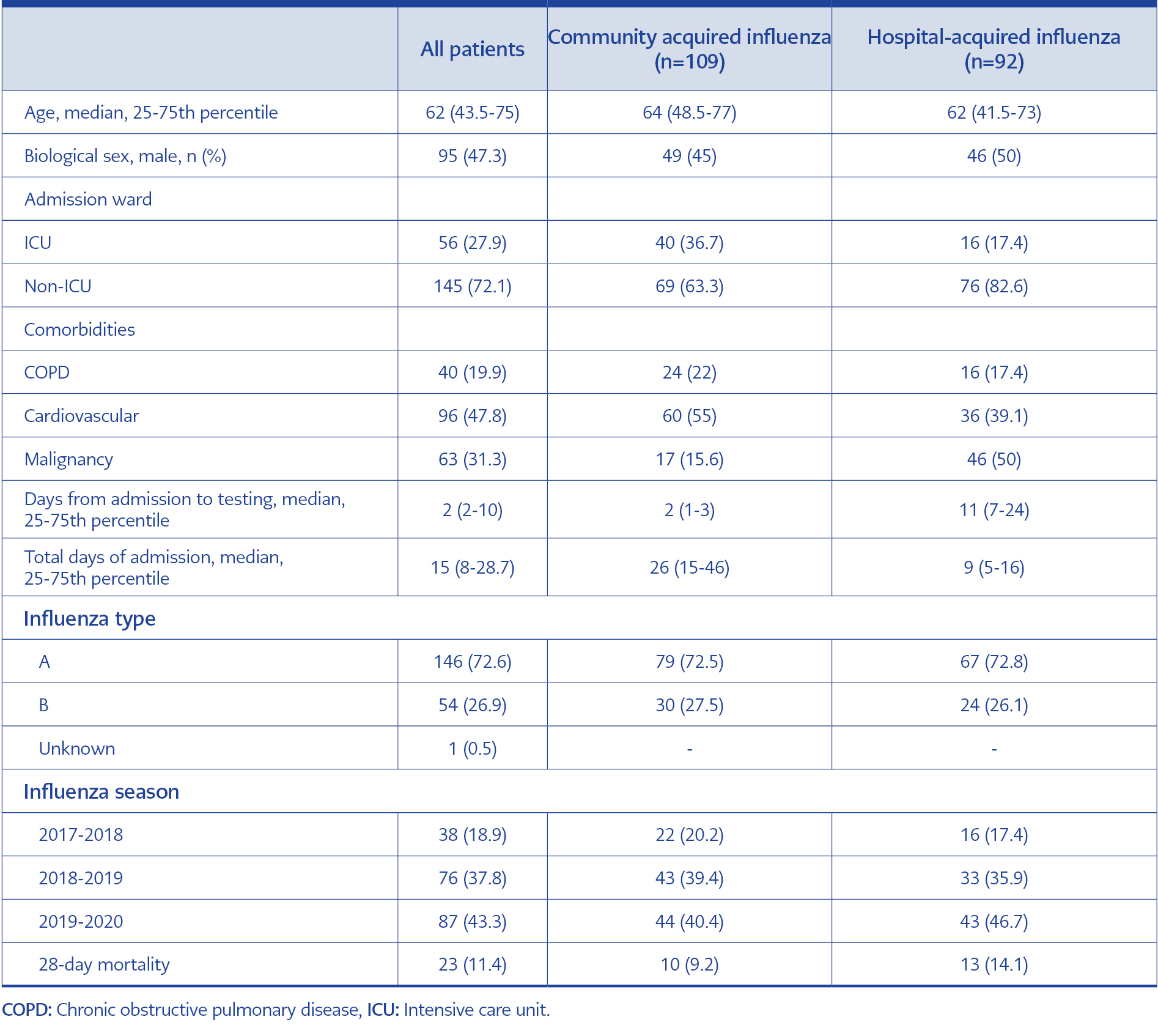
Age, gender, influenza subtype (A or B), influenza season (2017-18, 2018-19, and 2019-20), admission to intensive care unit (ICU), presence of chronic obstructive pulmonary disease (COPD), cardiovascular disease (CVD) and malignancy, were compared between the patient groups.
The primary outcome was 28-day mortality. Survival status was checked from the patient’s electronic records and the centralized electronic registry kept by the Turkish government.
All data were collected retrospectively on forms standardized for influenza inpatients. Information on forms was transferred to an electronic database. Once this transfer was complete, two physicians (HB, TB) went through the data to cross-check the accuracy and completed any missing information using archival and electronic records.
Statistical Analysis
Numerical variables were compared using the Mann-Whitney U test, and the categorical variables were compared using the Chi-square test between survivors and non-survivors.
For the multivariate analysis, age, biological sex, admission to the ICU, presence of COPD, cardiovascular and immunosuppressive comorbidities, influenza subtype, and other variables identified with univariate analyses (p<0.25) were entered into a logistic regression analysis to determine independent associations with 28-day mortality. Admission to the ICU at any point in the hospital stay was used as a surrogate for the severity of illness in this analysis.
Variance inflation factor (VIF) values were calculated for each variable to control for potential collinearity among the confounders. We included all selected predictors based on a priori criteria that none had VIF>2. The statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) 22.0 (IBM Corp., Armonk, NY, USA). A p-value of <0.05 was considered statistically significant.
The minimum sample size was calculated as 193 patients (58 HAI and 135 CAI) based on an estimated 15% mortality difference between groups (14,15), a two-sided alpha 0.05, and 80% power.
Results
During the study period, 201 hospitalized patients were identified as influenza positive. Of these, 92 (46%) were categorized as HAI, and the remainder (109) were categorized as CAI. The demographic characteristics of the two groups were similar, except the CAI patients had a higher ICU admission rate and a lower 28-day mortality rate (Table 1).
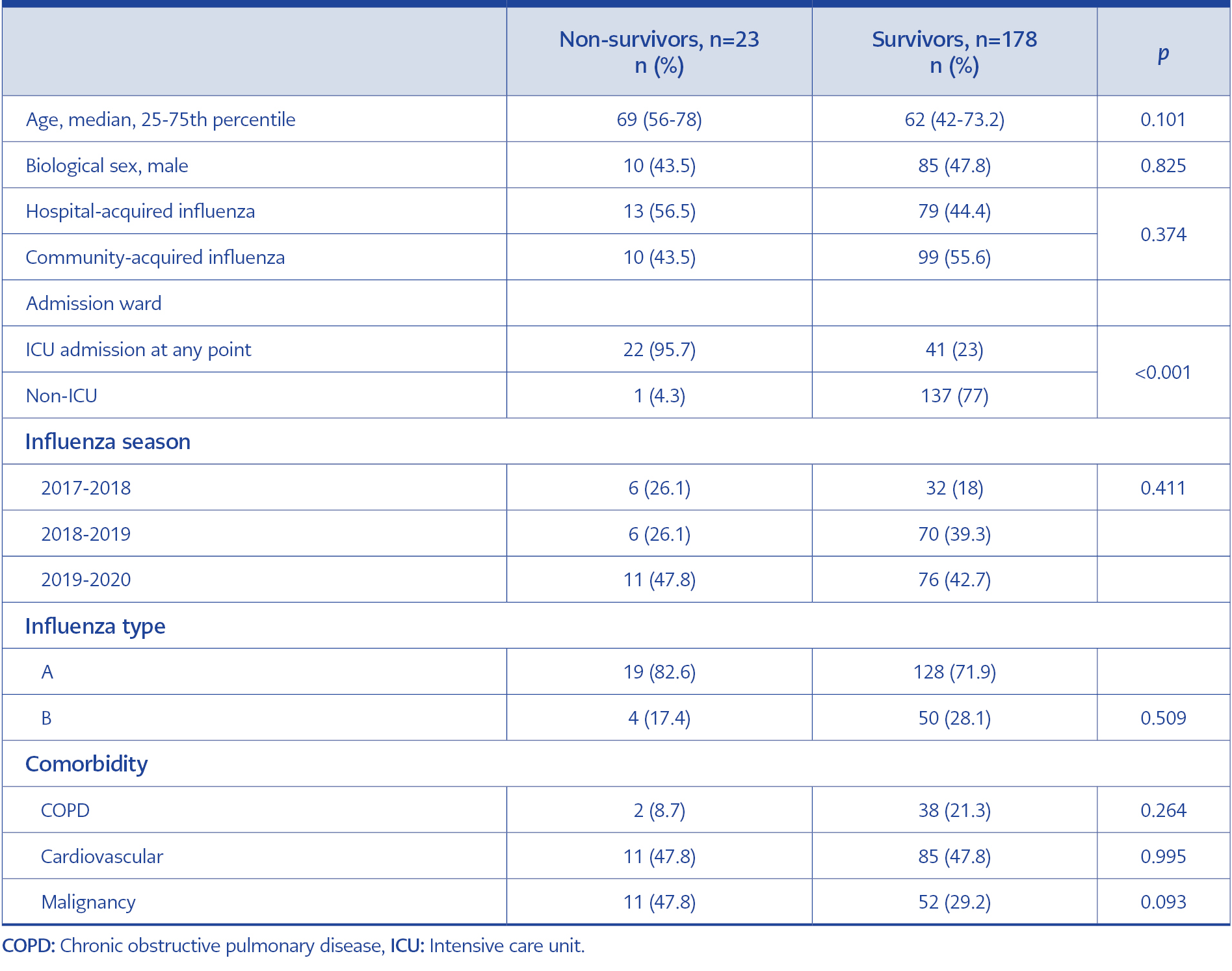
Univariate analysis showed no differences between survivors and non-survivors regarding patient characteristics, except non-survivors were more likely to have an ICU admission (Table 2).
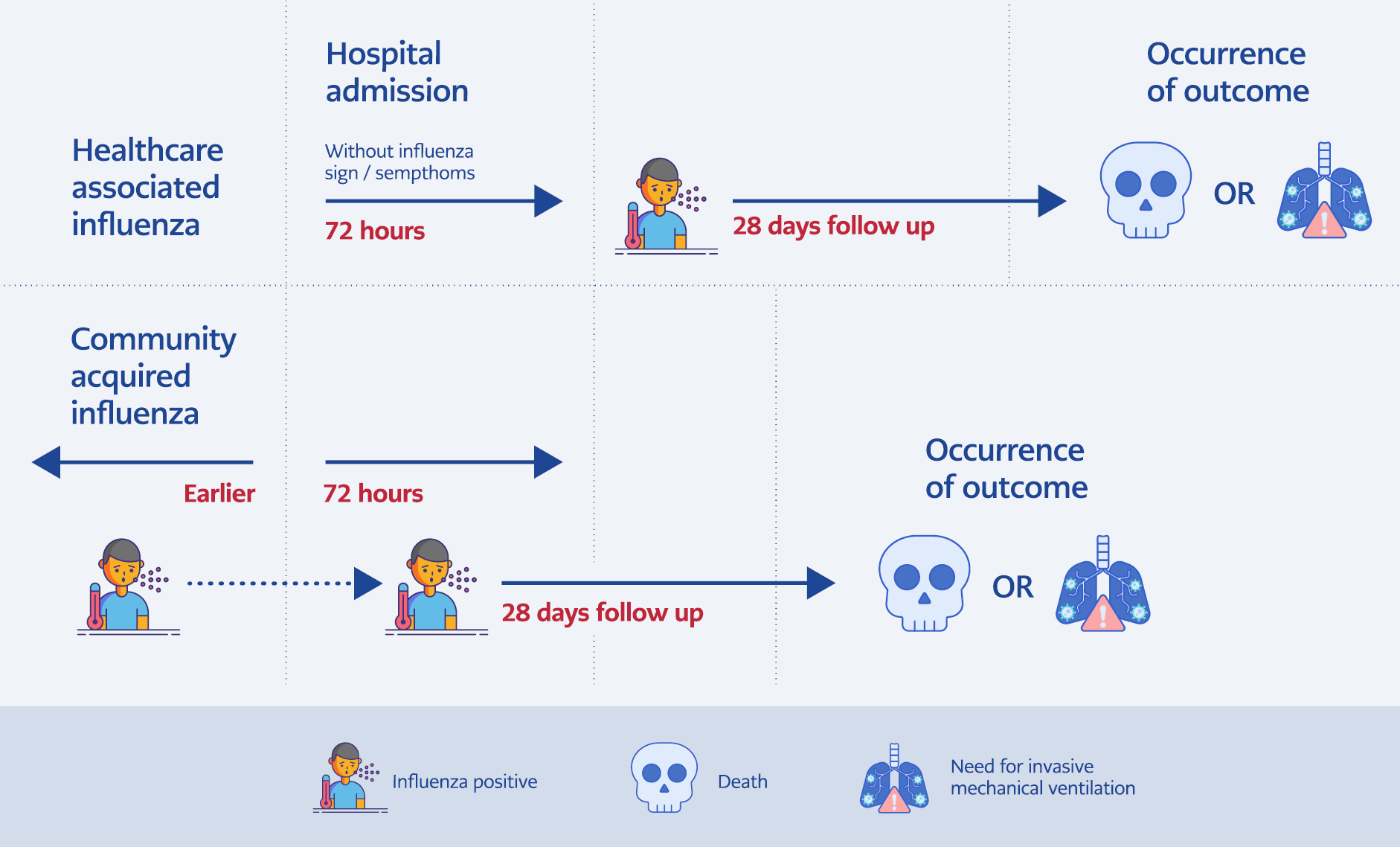
Figure 1. Explanation of the categorization and follow-up of patients. Hospital-acquired influenza was defined as patients who had no symptoms suggestive of influenza during the first 72 hours and tested positive any time after the first 72 hours of admission.
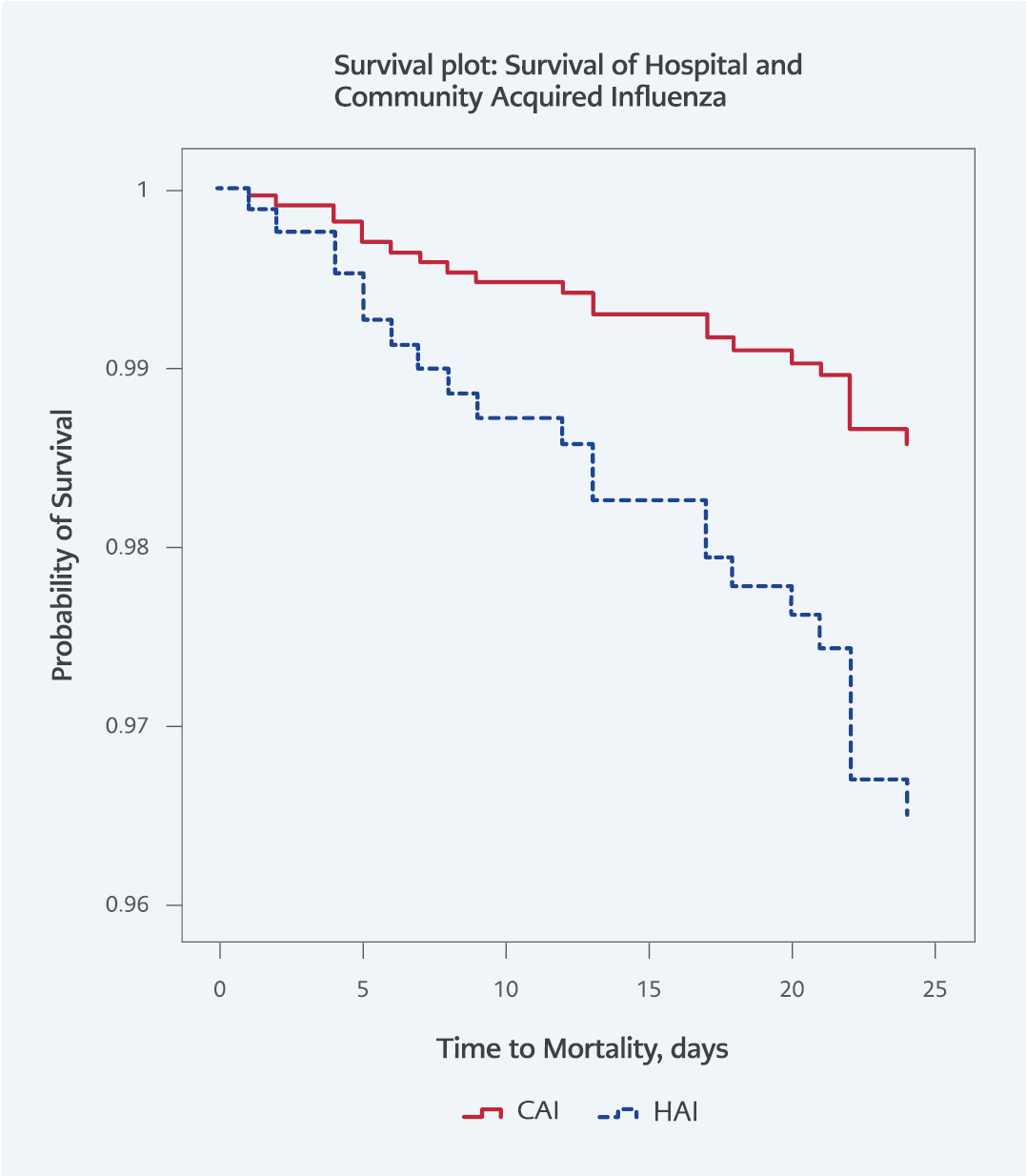
The results of the multivariable logistic regression analysis showed that HAI was associated with 5.6-fold increased odds of mortality (95% CI=1.6-19.3; p=0.006) after adjustment for age, gender, comorbidity, and ICU admission (Figure 2).
Discussion
This study showed that in a low vaccine coverage setting such as Türkiye, HAI was associated with a greater 28-day mortality than CAI after adjustment for important confounders. This finding highlights the importance of early recognition of this diagnosis so that early treatment with antivirals and implementation of isolation measures can be started (1).
We found that HAI was 46% of the total number of cases of influenza, which was higher than reported from other settings (7,16-18). For example, multicenter studies from Spain, Australia, and Canada reported HAI rates between 4.4 and 17.3% (7, 16, 18). A single-center retrospective study from Germany reported HAI rates between 20-24% (17). Another study from Germany reported the HAI rate during the 2014-2015 influenza season as 35.5% (19). The difference in HAI case rates could be due to varying definitions, influenza subtypes, and vaccination uptake rates. Potential factors explaining the high HAI proportion in our cohort include low vaccination rates, lack of understanding of the importance of infection prevention and control measures, and surveillance for respiratory viruses in the hospital. High rates of HAI in the study also indicate probable hospital transmission, either from the patient, the patient’s relatives, or healthcare workers to patients. This situation raises concerns regarding the respiratory infection control precautions in the healthcare facility. It is worth mentioning that throughout the study period, apart from the regular infection control measures, no additional initiatives for respiratory infection control were implemented until the beginning of the COVID-19 pandemic, which coincided with the end of the study period. In light of these findings, it is crucial to emphasize the paramount significance of robust infection control measures in healthcare settings to reduce hospital-acquired influenza infections.
The present study found 5.6-fold increased odds of 28-day mortality when comparing HAI to CAI. This finding is consistent with other studies, such as the study by Godoy et al., which found a 3.26 times higher risk of in-hospital death in HAI cases (95% CI=1.5-6.9) (16). Taylor et al. reported an almost two-fold increase in 30-day mortality. However, the authors also found that 30-day influenza-related mortality did not differ between HAI and CAI cases in the age-adjusted multivariable analysis, which could be explained by the high vaccine coverage of the patients: 54.6% in HAI patients and 36.3% in CAI patients (18). In addition, a multicenter study in Spain reported higher mortality rates in HAI (20). These authors also indicated increased mortality risk in the context of lower vaccine coverage. Other studies support our findings; however, we did not adjust for vaccination rates and antiviral treatment status.
Despite the high mortality rate in HAI cases, CAI patients showed higher ICU admission rates in univariate analysis. Similar results were shown by Naudion et al.; nosocomial influenza cases significantly had lower respiratory failure rates and lower ICU admission rates (21). This finding can be explained by the fact that patients with CAI have to be hospitalized when they have a complication, such as pneumonia, respiratory failure, or ICU admission need. Also, since they are in the hospital already, the HAI patients can be intervened faster than CAI patients.
There are studies reporting similar mortality rates between HAI and CAI cases (7, 22). However, these studies did not adjust for possible confounders when comparing HAI and CAI cases.
Based on the literature with varying vaccine uptake and regional or seasonal differences, our research findings demonstrate that HAI carries a significant mortality risk, highlighting its severity and poor prognosis compared to CAI (7, 16, 18). Therefore, early treatment, infection prevention, and control measures are essential to improve patient outcomes (23). This study conducted in Türkiye with low vaccine uptake rates also underlines the importance of enhancing vaccine coverage in this population to mitigate the risk of nosocomial transmission.
Our study has some limitations. First, the retrospective nature of data collection introduces the possibility of information bias, including factors such as the time elapsed between admission and testing, as well as the decision to conduct an Influenza test being determined by the treating physician’s discretion. Second, the lack of vaccination status data limited our interpretation of the results; for example, a low vaccination rate could have explained the effect size of HAI on mortality, and comparing our results with studies that include vaccination status as a confounding variable is difficult. However, several studies from Türkiye covering similar periods showed that the vaccination rate was between 0-27%, which was lower than reported in other studies (12, 13). Although several studies did not show an effect of vaccination rates on HAI and CAI mortality rates, it limits our results to generalize with the higher vaccine settings (7, 16). The lack of data on discharged patients who could have developed HAI after being exposed to influenza in the hospital may be the third limitation.
This study also has some strengths. It was conducted in a tertiary care center in İstanbul, TÜrkiye, and its findings may be generalizable to other similar hospital settings in Türkiye. The data quality check and data collection consistency also increased the internal validity of the research. We used the Influenza virus RT-PCR, a test with a good performance characteristic for diagnosis. Furthermore, we adjusted our analysis of 28-day mortality for important confounders.
This study revealed that patients with HAI are more likely to die within 28 days compared to those with CAI after controlling for key confounders that could affect the results. Further research and implementation of targeted interventions against hospital-acquired influenza should be conducted to mitigate the burden of influenza-related complications in vulnerable patient populations, ultimately improving overall patient outcomes.