Introduction
In late December 2019, a newly emerging virus caused an epidemic in Wuhan, China, which then became a pandemic that caused serious public health crises worldwide (1). The World Health Organization (WHO) announced the spread of the novel coronavirus on March 11, 2020 (2), and at the time of writing, the number of positive cases and deaths related to SARS-CoV-2 were reported to be 676 million and 6.77 million, respectively (3). Although fever, cough, sore throat, tiredness, loss of taste or smell, shortness of breath, and fatigue are the usual symptoms, some individuals have asymptomatic infections. The disease’s latency period ranges from 2 to 14 days, and it is transmitted easily by inhalation or close contact with infected people (4).
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a respiratory disease agent belonging to the Betacoronavirus genera (5). The genome of the virus translates into four major structural proteins, including spike (S), nucleocapsid (N), membrane (M), and envelop (E) proteins (6). During the pandemic, huge numbers of mutations in the genome have happened (7). These variances in nucleotide sequences altered the protein structures and then affected viral physiologic characteristics that significantly impacted on outbreak (8). Previous studies have demonstrated more than 10,000 mutations over the past two years (9-10). Thus, these variances, which were vastly distributed internationally, might have a massive influence on the diagnosis of COVID-19.
In order to prevent the viral pathogenesis and spread of the infection, breaking the chains of transmission is the best way through early diagnosis and isolation. Nowadays, there are many commercial kits and In-house tests for laboratory diagnosis of SARS-CoV-2. Among them, real-time reverse transcription-polymerase chain reaction (RT-PCR) is desired and extensively used for COVID-19 detection (11). Besides, real-time RT-PCR is accepted as the gold standard for diagnosing SARS-CoV-2, and both the WHO and the U.S. Food and Drug Administration (FDA) recommended it (12). However, this molecular test is expensive and needs complex equipment unavailable in all laboratories. Furthermore, a large number of samples should be collected to run the test, which is not cost-effective for a small number of cases.
In recent years, low-cost lateral flow immunoassays have been considered for detecting SARS-CoV-2 rapidly, and researchers have shown a growing interest in developing such tests. These rapid diagnostic tests (RDTs) are cost-effective, user-friendly, safe point-of-care testing, rapid, and results are easy to interpret (13). However, there is potential concern regarding the validation and performance of new commercial assays; investigating the specificity and sensitivity could be helpful for the accurate detection of infections and limiting the coronavirus pandemic.
We aimed to evaluate the commercial HARDSON COVID-19 Antigen Rapid Test Kit (ARBilim Biotechnology Inc., İstanbul, Turkey) compared with the commercial RT-PCR for diagnosing SARS-CoV-2 among the Iranian population.
Materials and Methods
Specimen Collections
We collected samples of 275 suspected patients from hospitals and healthcare centers to investigate the diagnostic accuracy of HARDSON COVID-19 Antigen Rapid Test Kit (ARBilim Biotechnology Inc., İstanbul, Turkey) in clinical samples. Two nasopharyngeal swabs were collected from each patient. One swab was tested with the rapid antigen test, and the results were recorded; the second swab was placed in 3 mL of a virus-transmitted inactivated media for RT-PCR testing. Following the manufacturer’s instructions, HARDSON COVID-19 Antigen Rapid Test Kit was performed immediately after sampling, and the results were obtained within fifteen minutes. Then, the results were evaluated in comparison with those of the commercial Pishtaz Teb Diagnostics COVID-19 RT-PCR Kit (Pishtaz Teb Diagnostics, Tehran, Iran), which was carried out within 24 hours.
Real-Time Polymerase Chain Reaction
According to the manufacturer’s protocol, viral nucleic acid extraction was performed on the samples by QIAamp Viral RNA Mini kit (QIAGEN Inc., Hilden, Germany). The whole procedure was completed in 24 hours to prevent RNA degradation. RT-PCR reactions were prepared in 25μl volume using a conventional real-time PCR instrument named Rotor-Gene Q (QIAGEN Inc., Hilden, Germany). In each reaction, positive and negative controls were used in parallel to assess the validity of the reactions.
All test procedures were applied according to standard manufacturer protocols. The cut-off cycle threshold (Ct) value for each specimen was recorded, and samples with a Ct value<40 were considered confirmed positive cases. The samples with discordant results were repeated.
Analysis of Results
Data analysis of HARDSON COVID-19 Antigen Rapid Test Kit was done using MedCalc Software (MedCalc Software Ltd., Osten, Belgium). The sensitivity, specificity, and accuracy with “exact” Clopper-Pearson confidence intervals (CIs) were calculated. Standard logit CIs were used to measure the negative and positive predictive values. HARDSON COVID-19 Antigen Rapid Test Kit outcomes were compared with the results of real time RT-PCR, considered a gold standard.
Results
A total of 275 suspected COVID-19 patients with a mean age of 49 years (range 23-75) were collected in this study. Of these, 162 positive and 113 negative samples were selected to evaluate HARDSON COVID-19 Antigen Rapid Test Kit and retested RT-PCR in duplicate to remove the false-negative or false-positive results. Among positive samples, 151 cases showed a low Ct value (<30) with high viral loads, and 11 samples had a high cut-off threshold (>30) with low viral loads. All the samples with no sigmoid shaped graph or Ct value higher than the positive range (>40) were interpreted as negative. Then, the samples were tested with HARDSON COVID-19 Antigen Rapid Test Kit to check the efficacy of the kit in Iran’s population. The average Ct value of 162 positive samples was recorded at 21.5 in the real-time RT-PCR test.
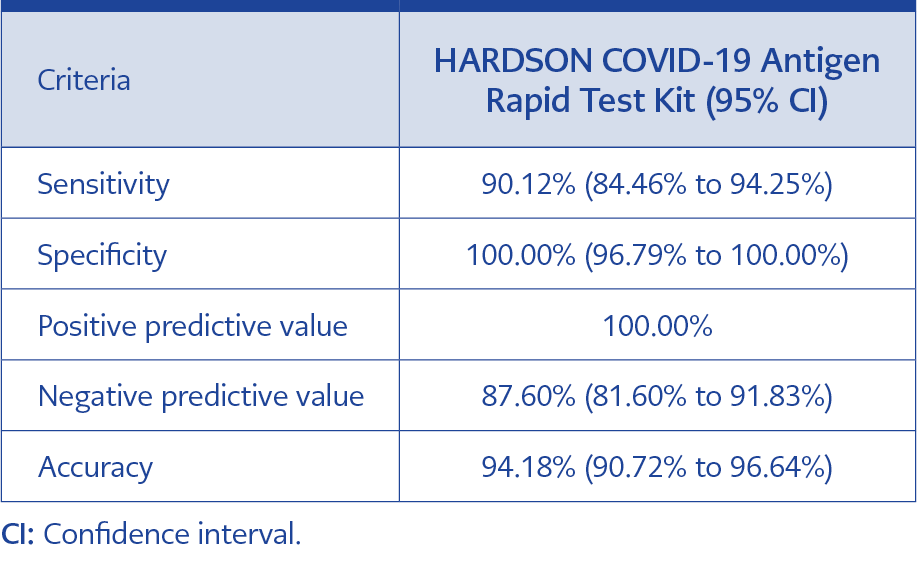
Table 1. Overall diagnostic efficacy of antigen-based rapid diagnostics test (RDT) used in this study.
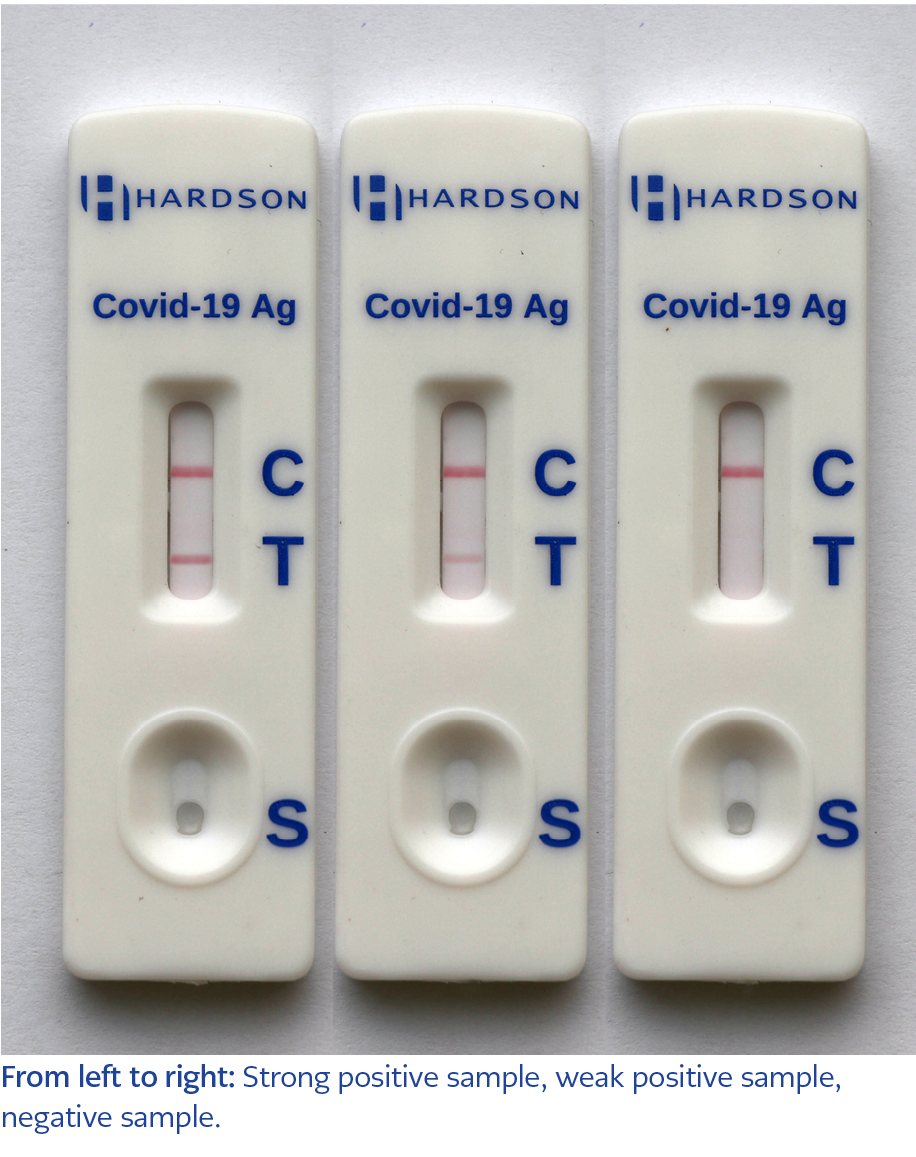
While 16 out of 162 positive samples (9.8%) were declared false-negative, the specificity was represented 100%, meaning there is no false-positive sample among 113 cases. Results indicated that HARDSON COVID-19 Antigen Rapid Test Kit performed well when positive samples with a low cut–off threshold were tested (Ct value <30). Among 11 weak positive samples with a cut threshold in the range of 31-39, none of the samples were detected by this kit. In this study, the sensitivity, specificity, and accuracy of HARDSON COVID-19 Antigen Rapid Test Kit were reported to be 90%, 100%, and 94%, respectively. The overall diagnostic efficacy of the kit is shown in Table 1, and strong positive, weak positive, and negative samples are illustrated in Figure 1.
Discussion
The SARS-CoV-2 pandemic has significantly challenged public health, economies, and society. The WHO has emphasized the importance of rapid and accurate diagnosis for transmission control through the community (14). Accurate and timely tests have been essential for controlling the pandemic. RT–PCR is a recommended test for the diagnosis of SARS-CoV-2 (15). However, a fully equipped molecular laboratory is required for this approach, such as a real-time PCR machine, biosafety cabinets, and RNA extraction instruments unavailable in all laboratories. In addition, trained technicians are needed to process the samples properly. On the other hand, antigen-based RDTs do not have these limitations and are regarded as self-tests that can be taken anywhere, even at home, to screen individuals and prevent symptomatic patients from referring to high-risk settings. In addition, antigen-based RDTs are more cost-effective than RT-PCR and only take 15 minutes to test and interpret the results with no additional reagents or consumables.
The WHO stated that acceptable COVID-19 RDTs should have a specificity of more than 97% and a sensitivity of more than 80% (16). So, this study was designed to compare and evaluate HARDSON COVID-19 Antigen Rapid Test Kit for detecting COVID-19 from clinical samples in the Iranian population. To our knowledge, this is the first report on the performance of commercial antigen-based RDTs in Iran. It is worth mentioning that the results revealed that HARDSON COVID-19 Antigen Rapid Test Kit could correctly identify 146 out of 162 strong positives (90%) and 113 negative samples (100%), but all 11 weak positive samples, along with five strong positive samples were reported false-negative. The present study found the specificity and sensitivity of HARDSON COVID-19 Antigen Rapid Test Kit 100% and 90%, respectively, higher than the WHO recommendation.
The reliability of COVID-19 antigen-based RDTs is different according to the manufacturers. The sensitivity rates have been reported in a range between 45%-97% among articles (17-19). In one study, COVID-19 antigen-based RDTs established high sensitivity (90%) based on the nasal swab, while in another study (13), 68% sensitivity was recorded among the Chinese community (20). Because of the global need for antigen-based RDTs, evaluating and reporting the accuracy of commercial assays for detecting COVID-19 is critical. The diagnostic kit analyzed in this study indicated excellent specificity and a relatively good overall sensitivity for the diagnosis of COVID-19 when compared to the RT-PCR detection kit. As 146 out of 151 samples (96%) with a good sigmoid graph at a low Ct value were detected, it could not be detected correctly by HARDSON COVID-19 Antigen Rapid Test Kit in Ct value higher than 24. Therefore, it is not recommended for low viral load samples before the onset of symptoms, and molecular methods like RT-PCR should be performed in such cases. However, it should be noted that the false positivity related to genome contamination (DNA/RNA) is more common in molecular procedures because of the large-scale extraction done in the laboratory (21).
In this study, the overall antigen-based RDTs sensitivity was directly dependent upon the RT-PCR Ct values, as expected. Based on our findings, we concluded that HARDSON COVID-19 Antigen Rapid Test Kit could be an excellent rapid test, especially in high viral loads (96%) for reporting. So, it would be recommended to be integrated into routine diagnostic laboratories and used as an at-home rapid antigen test.