Introduction
Rational use of medicines (RUM) has been a concern worldwide for half a century, and is of great importance, given that almost half of all prescribed drugs are irrational (1). The irrational use of drugs can be disastrous and may cause adverse events, creating a considerable burden. The emergence of drug-resistant microorganisms is still a global concern, mainly due to the irrational use of antimicrobials. Therefore, the evaluation of antibacterial utilization in hospitals is of great importance. Preventing the irrational use of different antibacterials decreases the cost for both hospitals and patients (2). Moreover, an increase in multi-drug resistant microorganisms can be avoided, the rise of adverse drug reactions can be decreased, and the lack of antibacterials for emerging cases can be solved by applying the principles of RUM. Drug utilization studies (DUS) can be used to ensure and encourage prudent use of antibacterials, especially in tertiary hospital settings. They are convenient for studying the rational use of antibacterial agents, and can provide the actual status of antibacterial utilization for a particular hospital (3). The objectives of DUS cover the identification, analyses, importance, and consequences of the problems, with various approaches according to the needs of the users, including the health authorities, drug manufacturers, academic and clinical health professionals, social scientists, economists, the media, and the consumers (4). DUS may be quantitative or qualitative. The objective of a quantitative DUS is to quantify the present state, the developmental trends, and the time course of drug usage at national, regional, local, or institutional levels of the healthcare system.
Drug utilization numbers are variable statistics because pandemics, wars, famines, etc. have a considerable effect on overall drug use. For example, the British Society for Antimicrobial Chemotherapy Council was concerned about the increasing antibiotic consumption rates among patients without bacterial infections during the 2009 swine flu pandemic (5). The emergence of coronavirus disease 2019 (COVID-19) has raised similar concerns and questions about drug utilization in both hospital-settings and national level. COVID-19 was characterized as a pandemic by World Health Organization (WHO) on March 11, 2020 (6). Republic of Turkey Ministry of Health reported their first case on the same day (7).
This study was conducted at the Koç University Hospital (KUH), a tertiary hospital in Istanbul. We aimed to evaluate and identify the antibacterial utilization patterns among adult inpatients before and after the initiation of the COVID-19 pandemic, using the recommended parameter, DDD/100 admissions. Our secondary objectives were to identify and compare the five most frequently prescribed (top five) antibacterial classes, and to understand the antibacterial utilization patterns among the departments of KUH, both before and after the initiation of the COVID-19 pandemic. The information from this tertiary hospital-based retrospective study may influence other hospital-based and nationwide pharmacoepidemiology studies on drug utilization and rational use of medicines during the pandemic.
Materials and Methods
General Study Design
ANTIBACDUS-PAN is a cross-sectional, antibacterial utilization study conducted in a single tertiary hospital-based setting. The study protocol was designed and conducted according to the European Network of Centers for Pharmacoepidemiology and Pharmacovigilance (ENCePP) methodology and guidelines (8). The overall methodological approach is descriptive with a one-year overall study period, specifically looking into two 6-month periods: before and during the COVID-19 pandemic. The DU90% method was used for the selection of systemic antibacterials, and the two periods were compared with the recommended parameter DDD/100 admission.
Index Date
The announce date, March 11, 2020, of the first COVID-19 case in Turkey by the Republic of Turkey Ministry of Health (7) was set as the cut-off date to determine pre-COVID-19 and COVID-19 6-month periods: the pre-COVID-19 period was defined as the period from September 10, 2019, to March 10, 2020, and the COVID-19 period was defined as the period from March 11, 2020, to September 11, 2020. These 6-month periods were considered appropriate for evaluating the antibacterial utilization patterns as the COVID-19 pandemic continues.
Eligibility Criteria
We included adult (aged ≥ 18 years) inpatients of both genders, and those who were prescribed at least one systemic antibacterial (ATC code J01) within the one-year study period (September 10, 2019, to September 11, 2020).
Exclusion Criteria
We exluded pediatric inpatients (aged < 18 years) and systemic antibacterial prescriptions with missing information.
Data Source
The KUH Inpatient Electronic Order System (CP) was used as the data source to retrospectively identify and extract all systemic antibacterials prescribed for all adult inpatients admitted to KUH during the study period.
Data Extraction
The systemic antibacterials (ATC code J01) available in Turkey were identified in the global medicines list that is publicly available on the website of the Turkish Medicines and Medical Devices Agency (9). The WHO ATC/DDD system was used to classify individual antibacterials and to identify their DDDs (10). For all subjects meeting the eligibility criteria, data related to socio-demographic data: age, gender, and body weight, prescription data (name of the medical department; date of prescription; generic name, dosage, administration route, date of start, and duration of use of the prescribed antibacterial agent(s); main diagnoses codes) were extracted.
Data Protection, Data Anonymization, and Coding
Both acquisition and processing of the data was done by the data processing specialist at KUH and in accordance with the Law on the Protection of Personal Data that entered into force in 2016 (11). All prescriptions meeting the eligibility criteria for any systemic antibacterial agent were given a code number. The file evaluations were made over the code numbers. A database specific to the study was created by extracting and storing the data in a data-specific Google Drive folder and a Microsoft Excel file. The researchers only used the data that were within the scope of this research, no data that could reveal the identity of the patients were used.
Statistical Analysis
Statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) 22.0 (IBM Corp., Armonk, NY, USA) and Microsoft Excel (Microsoft Corp., USA). All categorical and continuous data are described using descriptive statistics. The descriptive analysis of categorical and ordinal variables provided the frequency and percentage of each modality and missing data. The descriptive analysis of quantitative variables provided the mean, standard deviation, and median (minimum-maximum). Statistical significance was set at p <0.05. Categorical variables were compared using the chi-square test (or Fisher’s exact test/Yates’s continuity correction where available). Normality was evaluated using the Shapiro-Wilk test. The Mann-Whitney U test was used to compare two independent non-normally distributed variables. Student’s t-test was used to compare two independent normally distributed variables.
DDD Calculations
DDD/100 admissions was calculated to compare the drug utilization. The following formulations were used:
Calculation of DDD/100 admission-day:
DDD/100 admissions = (No. of units administered in a given period)/(DDD × number of admissions) × 100
DDD number = (Prescribed dose [g]) / (DDD according to the ATC code [g])
For the selection of antibacterials, the Drug Utilization 90% (DU90%) method was used, which is defined as the number of drugs accounting for 90% of the drug volume to increase DDD. This method serves as a powerful tool for determining the quality of drug prescriptions (12). Antimicrobial utilization was also evaluated according to different major clinical departments.
Results
Data Management and Inclusion of Inpatient Prescriptions
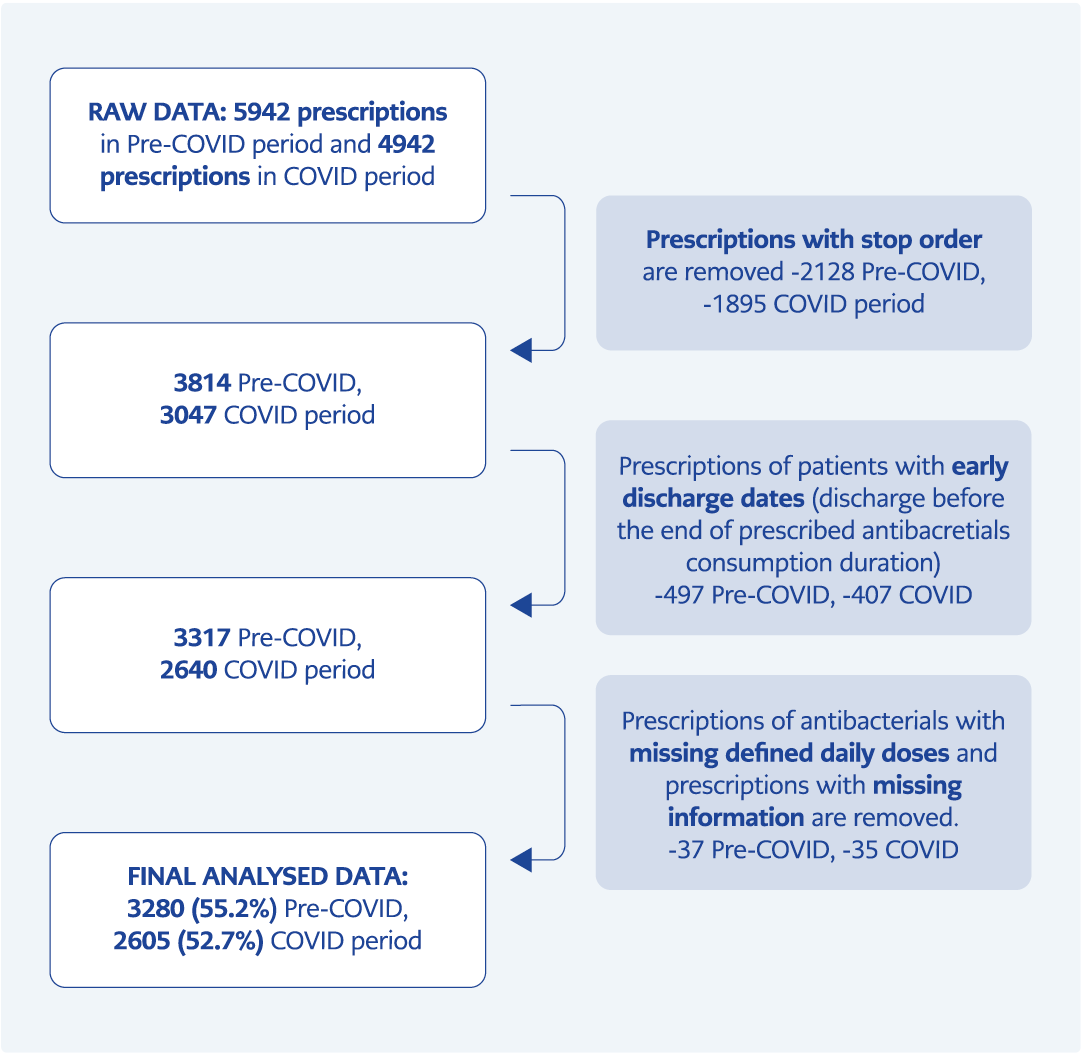
Within the one-year study period, 5942 prescriptions for the pre-COVID-19 period, and 4942 prescriptions for the COVID-19 period were extracted from the KUH CP. A total of 2662 (55.2%) prescriptions from the pre-COVID-19 period, and 2337 (52.7%) from the COVID-19 period were excluded. A relatively high percentage of prescription exclusion was mainly due to the different operational functions of the CP inpatient order system. Finally, 5885 (3280 pre-COVID-19 period, 2605 COVID-19 period) systemic antibacterial prescriptions from the KUH CP were analyzed and included in the study (Figure 1).
Demographics
During the pre-COVID-19 period, the number of females (1155 [52.9%]) with antibacterial prescriptions were higher than the number of males (1028 [47.1%]), while during the COVID-19 period, males (778 [51.5%]) were higher. Compared with the pre-COVID-19 period, there was a significant difference between males and females in the two time periods (p = 0.009). For the total study period, the mean age was 58.08 years (±17.9 SD), with actual being 57.89 (±17.9 SD) and 58.35 years (±17.9 SD) for pre-COVID-19 and COVID-19 periods, respectively. During the COVID-19 period, the mean age slightly increased by 0.79%, which was not significant according to the Mann-Whitney test (p = 0.388). The body weight variable could not be used because there was a high percentage of missing data.
Analyses by Prescription Numbers
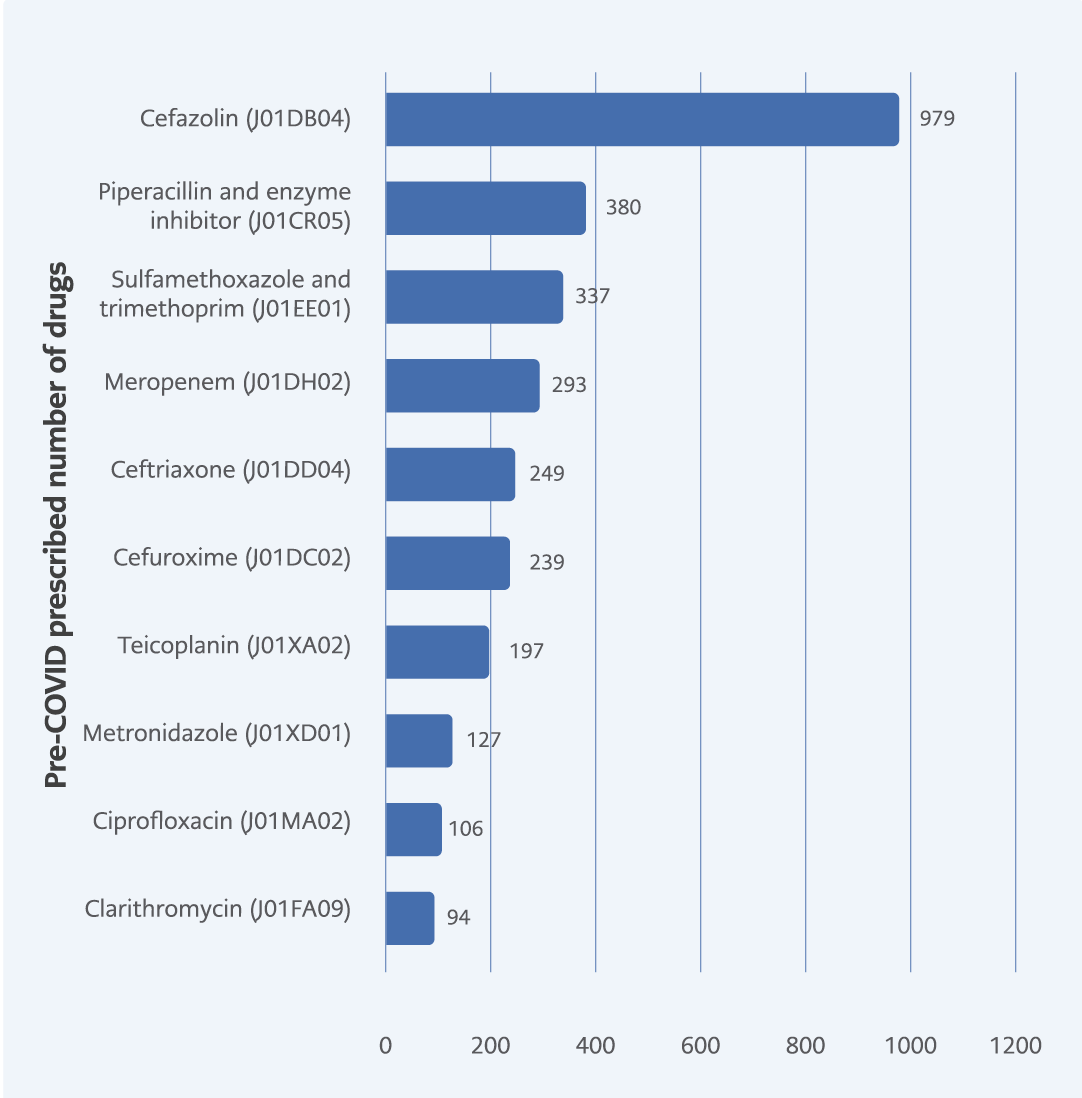
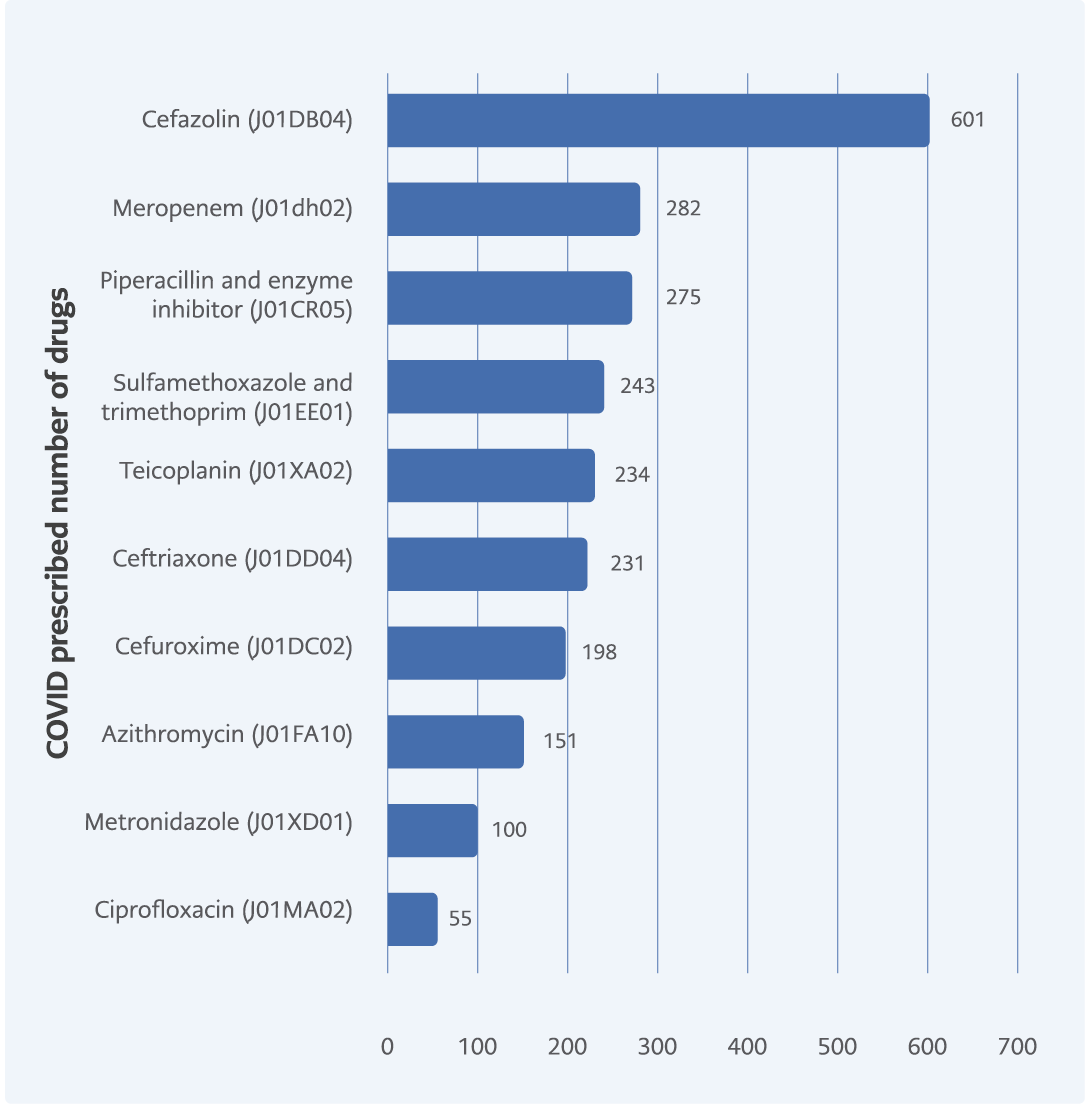
According to the prescription numbers in the pre-COVID-19 period, cefazolin was the most prescribed systemic antibacterial agent, followed by piperacillin-tazobactam, trimetoprim-sulfamethoxazole, and meropenem (Figure 2). In the COVID-19 period, cefazolin was first in the top 10 antibacterial prescriptions list, and meropenem moved to second place. However, according to prescription numbers meropenem was prescribed fewer times, when compared to the pre-COVID-19 period. Piperacillin-tazobactam, trimetoprim-sulfamethoxazole still had high utilization patterns during the COVID-19 period. Moreover, azithromycin emerged in the top 10 list, replacing clarithromycin (Figure 3).
Analyses by DDD/100 Admissions
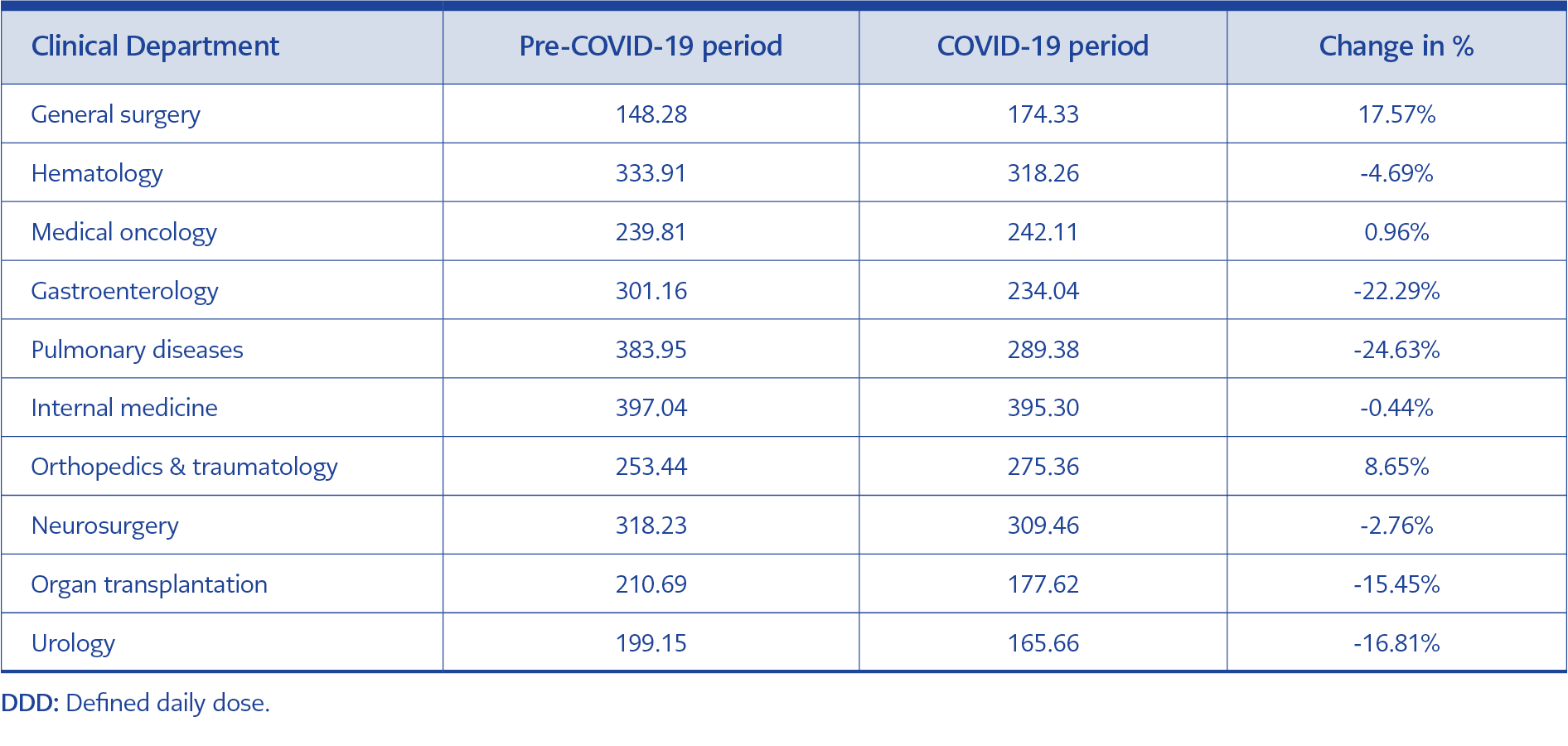
Table 1. Number of DDD/100 admissions and percentage changes by clinical departments during pre-COVID-19 and COVID-19 periods.
DDD/100 admissions increased by 3.76% in the COVID-19 period (from 193.96 to 201.26) showing that systemic antibacterial utilization has increased in the COVID-19 period compared to that in the pre-COVID-19 period. Systemic antibacterial utilization increased by 17.57% in the general surgery department and increased by 8.65% in the orthopedics and traumatology department during the COVID-19 period. In almost all other departments, the systemic antibacterial utilization decreased (Table 1).
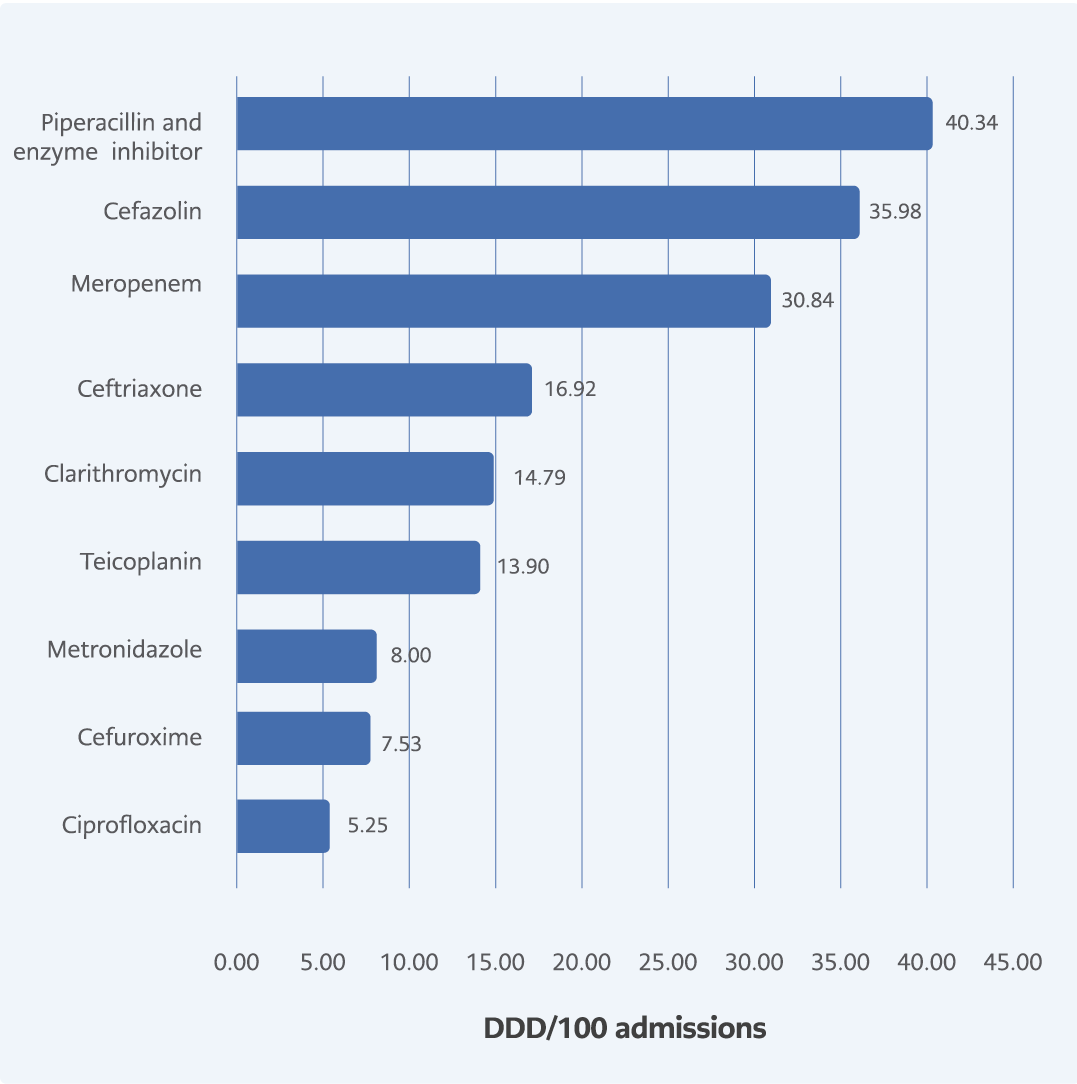
Figure 4. Most utilized systemic antibacterials accounting for DU90% with DDD/100 admissions in pre-COVID-19 period.
Analyses by DU90% Methodology
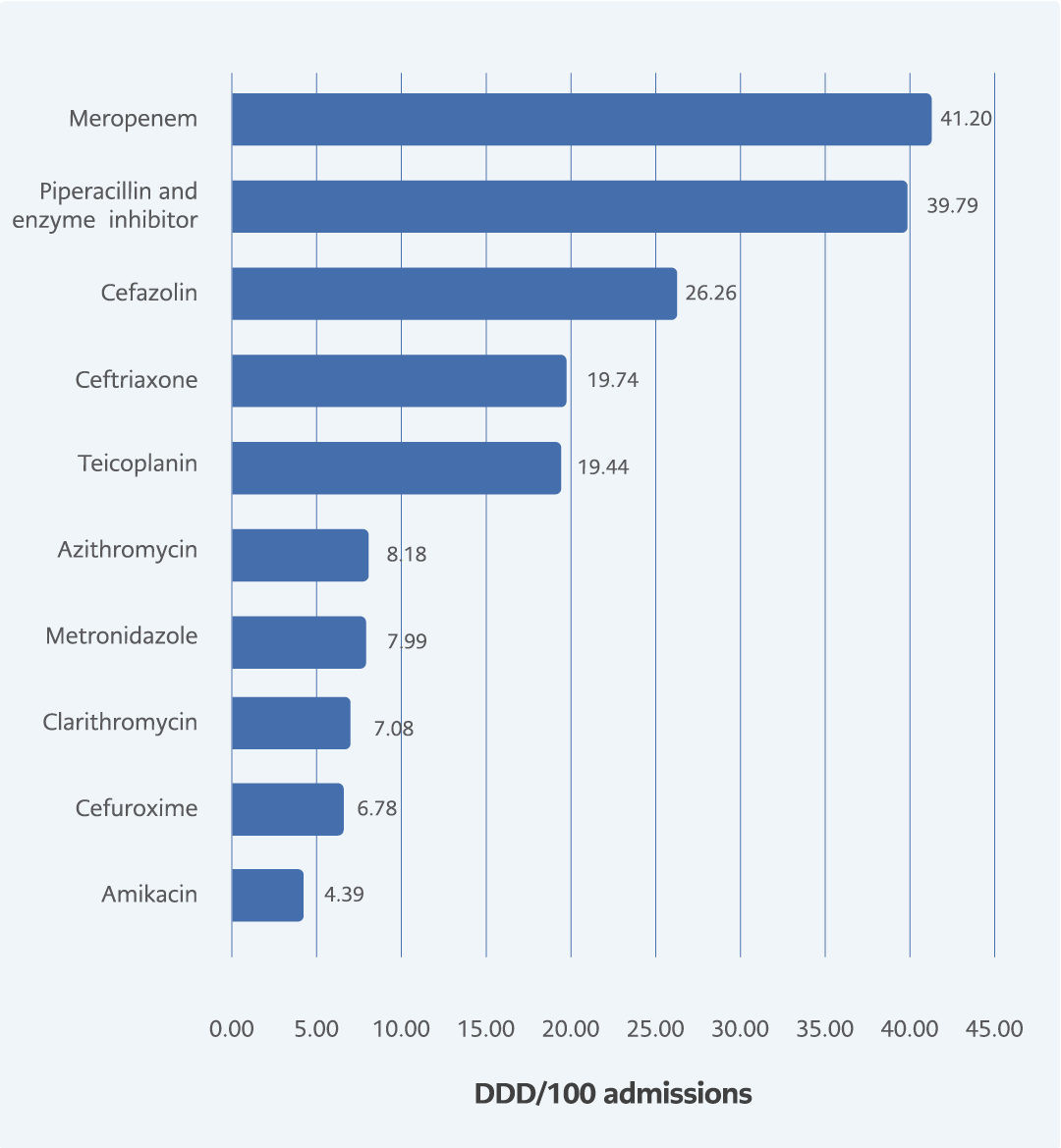
Figure 5. Most utilized systemic antibacterials accounting for DU90% with DDD/100 admissions in COVID-19 period.
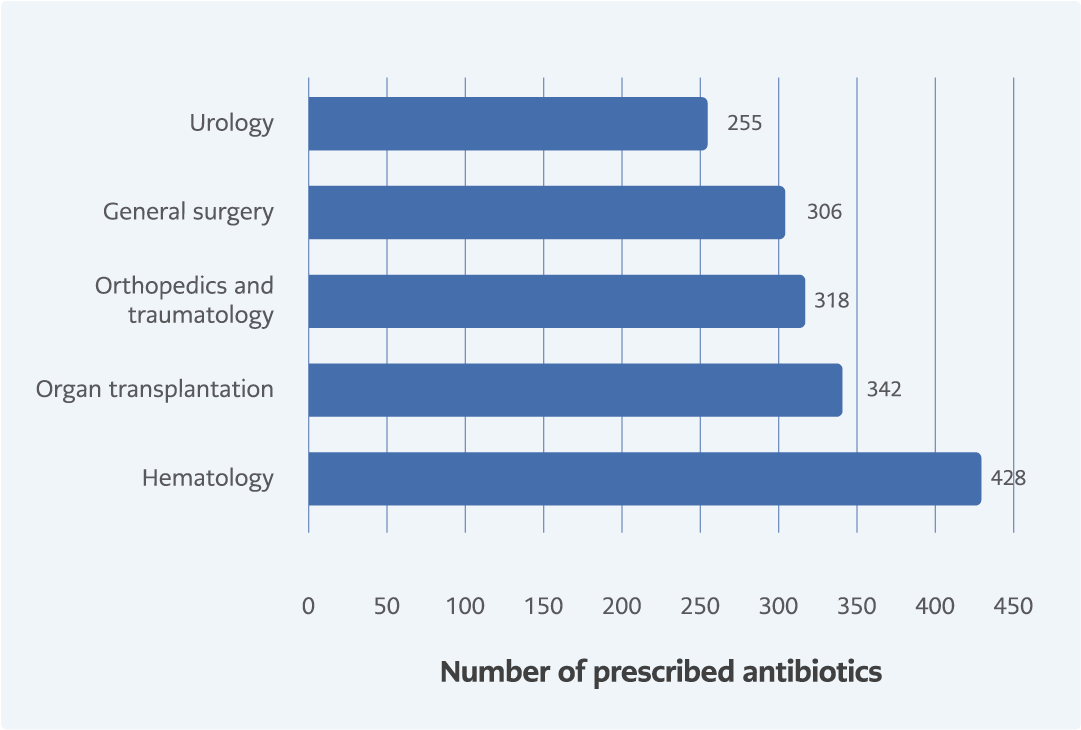
Figure 8. Top 5 departments according to their number of systemic antibacterial prescriptions in pre-COVID-19 period.
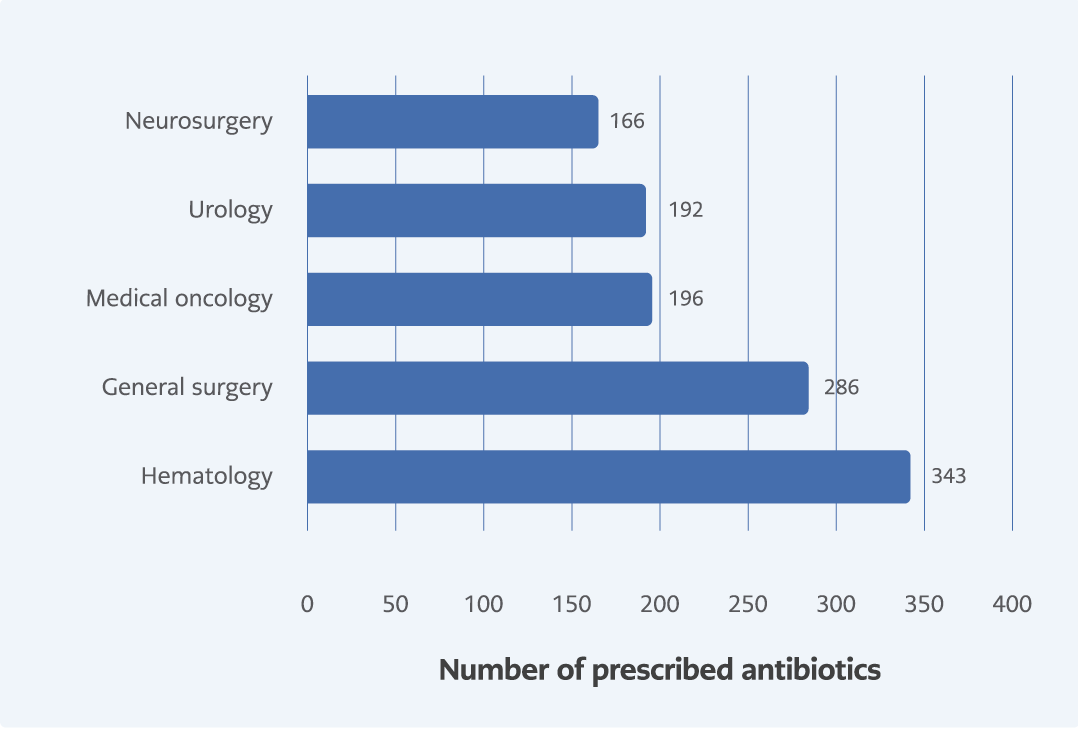
Figure 9. Top 5 departments according to their number of systemic antibacterial prescriptions in COVID-19 period.
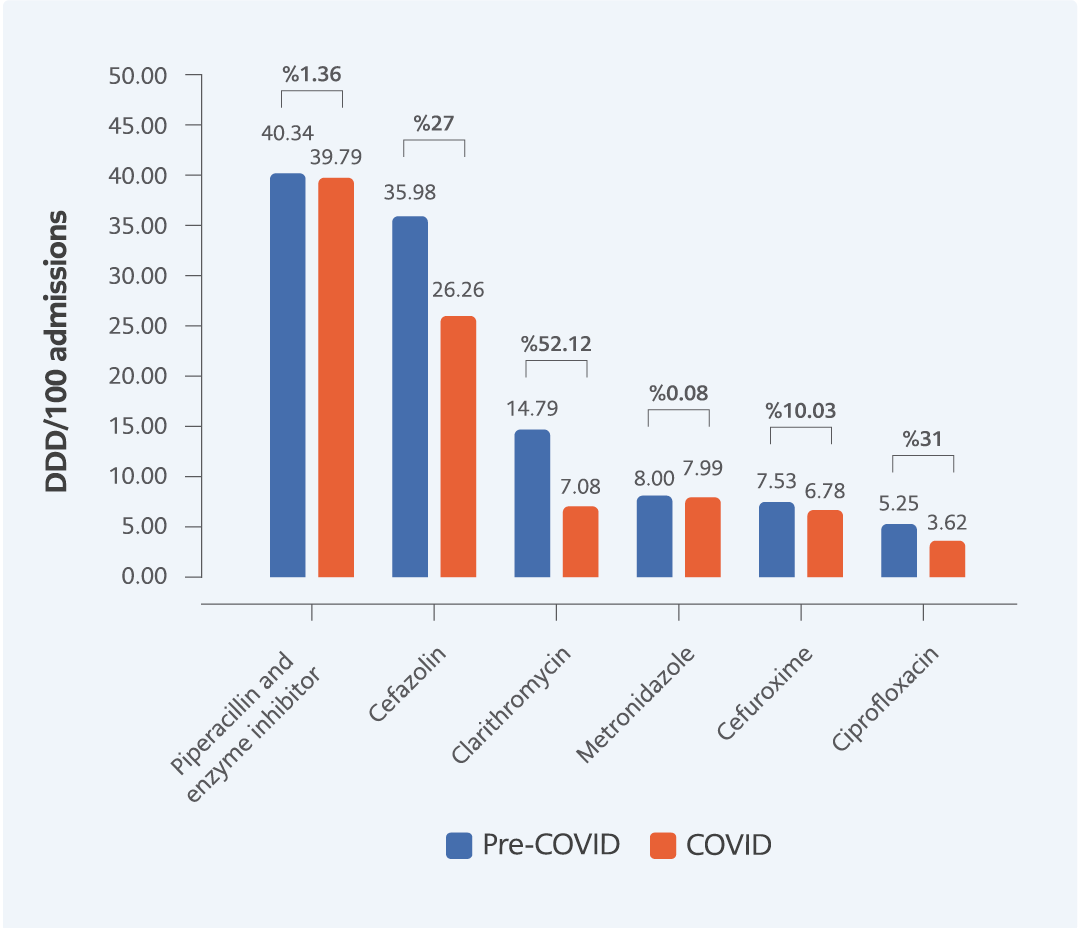
Using the DU90% methodology, systemic antibacterials that constituted the top 90% of the DDD were sorted. Piperacillin and enzyme inhibitors were the most commonly used systemic antibacterial agents in the pre-COVID-19 period, followed by cefazolin and meropenem according to DDD/100 admissions (Figure 4). In the COVID-19 period, meropenem was the most utilized antibacterial. Azithromycin and amikacin emerged in the top ten list of most utilized antibacterials (Figure 5). Ciprofloxacin lost its place in the top 10 utilized antibacterial agents during the COVID-19 period.
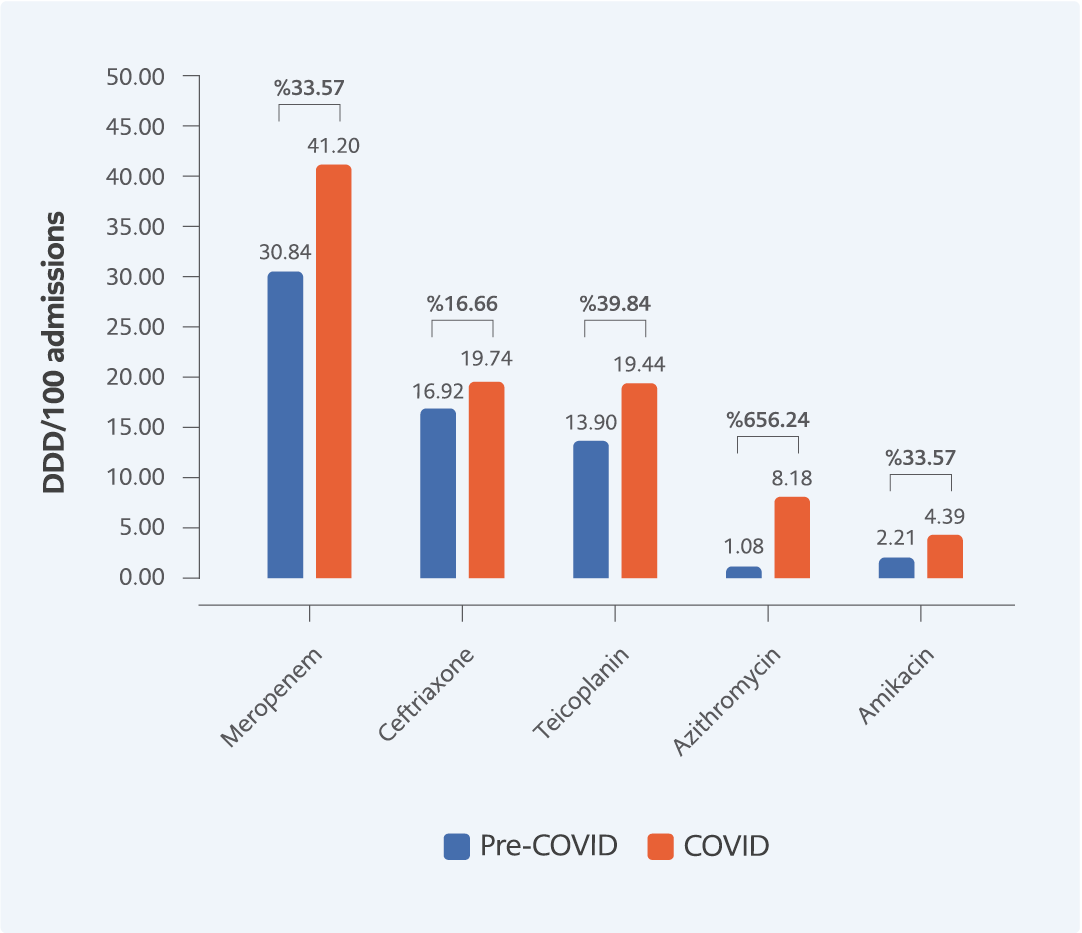
The use of antibacterial agents such as meropenem, ceftriaxone, teicoplanin, azithromycin, and amikacin increased during the COVID-19 period (Figure 6). The maximum amount of meropenem utilization increased from 30.84 to 41.20 DDD/100 admissions, whereas azithromycin utilization increased by 656.24%. Cefazolin, clarithromycin, cefuroxime, and ciprofloxacin decreased during the COVID-19 period (Figure 7). The highest percentage decrease was 52.12%, observed for clarithromycin in the COVID-19 period. Piperacillin-tazobactam, and metronidazole did not change significantly during the COVID-19 period according to DDD/100 admissions.
Analyses by Departments by Number of Antibacterial Prescriptions
Prescriptions were analyzed according to the number of antibacterial prescriptions at different departments of KUH during the pre-COVID-19 and COVID-19 periods. During the pre-COVID-19 period, the hematology department had the highest number of prescriptions compared to the other departments, followed by organ transplantation, orthopedics and traumatology, general surgery, and urology (Figure 8). During the COVID-19 period, the hematology department was still in first place according to the number of prescriptions. Urology and general surgery remained in the top five. However, medical oncology and neurosurgery replaced organ transplantation, and orthopedics and traumatology after the emergence of the COVID-19 pandemic (Figure 9).
Discussion
This study showed that overall systemic antibacterial utilization at the Koç University Hospital increased after the emergence of SARS-CoV-2 in Turkey. This is an expected finding, given the guidelines enforced by the Republic of Turkey Ministry of Health during the pandemic. Guidelines for the management of COVID-19 advised the utilization of β-lactams, macrolides, and respiratory fluoroquinolones to prevent concomitant bacterial infection for moderate and severe pneumonia. Azithromycin is an example of this situation, with nearly 650% increment. Meropenem, which is a broad-spectrum carbapenem, was the systemic antibacterial with the highest DDD/100 admission during COVID-19 period, with an increased utilization by 33.57%, which was similar to that of azithromycin. The utilization increment among antibiotics such as meropenem, azithromycin, and teicoplanin could be indicators of changes in the antibacterial prescription pattern during the COVID-19 period. During the conduct of the study, there were not any guidelines regarding antibacterial prescription in KUH. Therefore, in case of COVID-19 period, physicians took into considered the recommendations of Republic of Turkey Ministry of Health during their routine practice. According to these recommendations, ceftriaxone, amikacin, and meropenem are under the list of antibacterials recommended for avoiding drug-drug interactions during antiviral and glucocorticoid therapy (13). One can observe that these drugs were also utilized after the emergence of COVID-19 pandemic. The increment in the utilization of these antibacterials can be prevented if KUH had a local guideline for antibacterial prescription.
The WHO ATC/DDD methodology was preferred for evaluating antibacterial utilization patterns because it minimizes misconceptions and bias (10). DDD is defined as “the assumed average maintenance dose per day for a drug used for its main indication in adults.” DDD is not prone to error from regional variables unlike drug consumption expressed in terms of cost, number of units, number of prescriptions, or the physical quantity of drugs. Therefore, these parameters were not considered in this study. By applying DDD, it is possible to examine changes in drug utilization over time, evaluate the effect of the pandemic on drug use, document the relative therapy intensity with various groups of drugs, follow the changes in the use of a class of drugs, and evaluate regulatory effects on prescribing patterns. Drug utilization patterns of a particular period should be normalized using the recommended DDDs of WHO and the number of admissions. Thus, DDD/100 admission, which evaluates utilization as a number of DDDs, is a variable that can be used for comparative studies because of its reliability and precision.
The main strength of this study is the utilization of the DU90% methodology, which is a simple method for assessing drug utilization in healthcare (12). DU90% is the number of drugs accounting for 90% of the drug volume in order of increasing DDD. DU90% is an improvement over the traditional ‘Top-10’ listing since it provides both qualitative and quantitative information. The remaining 10% contained drugs for complex comorbidities, adverse reactions, drug intolerance, and rare conditions (14). Hence, using DDD and DU90% represented the antibacterial utilization patterns more accurately in our study.
Distribution of DDD/100 admissions according to departments is important because it provides a different perspective of patterns in antibacterial utilization in the KUH. During the study period, there were not any wards allocated to COVID-19 patients. Therefore, COVID-19 patients were hospitalized at any department designated by the chief medical office of KUH and transferred to any department when necessary. Although most of the departments showed decrement according to their cumulative DDD/100 admission, overall utilization increased after the emergence of the pandemic. This is due to an increase in utilization in specific departments, such as general surgery (17.57% increase) and adult intensive care unit (18.25% increase). Moreover, the Infectious Diseases Department utilized less antibacterials (30.41% decrease) after the emergence of COVID-19, which could be a result of the intense preventive measures taken by the Republic of Turkey Ministry of Health, such as social distancing, use of face masks, and informing the public on infectious diseases (data not shown in Table 1). It is also compatible with the results from different countries with decreasing numbers of respiratory infections (15).
A prospective observational study examining antimicrobial utilization in the intensive care unit of a tertiary care hospital concluded that the prescription of drugs was rational (16). A more recent study examining the antimicrobial patterns at a tertiary care hospital using the ATCC/DDD system and the DU90% method revealed that DUS using these metrics is helpful in monitoring trends of drug use over time and promoting the rational use of antibiotics (17). DDD metrics were also used in a cross-sectional descriptive study measuring antibacterial drug utilization patterns at a regional hospital, providing documentary evidence about antibacterial utilization patterns and antibacterial drugs included in the DU90% segment, which contributes to decision making for purchasing medicines or preparing medicine budgets (18).
Due to the difficulties in obtaining the number of bed-days from the KUH prescription database for the study period, DDD/100 bed-day analyses could not be performed. However, this may not be considered a limitation because DDD/100 admission is also a trusted method for evaluating drug utilization patterns.
This study was conducted under the scope of hospital pharmacoepidemiology, and it is possible that the results cannot be generalized for the entire Turkish population. The utilization indicators were not predictive of the clinical perspective. It is unknown whether the prescriptions were for treatment or prophylaxis purposes; therefore, the results of the study cannot be used to analyze the quality of diagnosis and treatment. This study aimed to evaluate only the utilization patterns of systemic antibacterials of KUH pharmacy-approved inpatient orders, which was extracted from the KUH CP. System-based errors can still exist beyond the capability of data management techniques.
The one-year study period was divided into two 6-month periods of pre-COVID-19 and COVID-19, which were set according to the formal date of initiation of the SARS-CoV-2 pandemic in Turkey, the first COVID-19 case declared by the Republic of Turkey Ministry of Health, because of this timeline, seasonal admission variations could have affected the results of our study. However, this limitation is minimal because of the nature of the pandemic. Moreover, there were preventive programs that enhanced the pace of communication between health professionals and the public, raising awareness about the pandemic. The one-year study period was deliberately chosen to prevent the Turkish vaccination program from interfering with the results. This is another major strength of the study for evaluating the effects of the COVID-19 pandemic on systemic antibacterial utilization.
An important outcome of this study, although not expected, was the relatively high percentage of excluded inpatient prescriptions due to the problems with the coding system of the CP algorithm used at KUH. Prescriptions with missing or non-understandable data or coding errors were excluded. This may be considered as a signal for choosing a better inpatient electronic order system.
The results of this study could potentially lead to the identification of irrational use of systemic antibacterial drugs and the impact of pandemic on antibacterial utilization, or even drug utilization in an extended state. This may further lead to regulatory intervention or to changes in the understanding of the rational use of antibacterial drugs, especially under specific circumstances, such as the current pandemic.
Conclusion
Systemic antibacterial utilization increased at KUH during COVID-19 pandemic with a trend of increased utilization of reserved antibacterials. The effect of the pandemic on antibacterial utilization is a concern for antibacterial resistance. More vigilance is required for the rational use of systemic antibacterials, especially during pandemics.