Introduction
Respiratory illness from an acute infection remains the most common etiology for inpatient pediatric hospitalization (1). Imminent respiratory arrest from prolonged hypoxemia may lead to bradycardia and subsequent cardiac collapse (2). As such, apnea (or respiratory distress) followed then by bradycardia is the classic modus operandi seen at the bedside, especially in neonates whose airways are still developing. Positional bradycardia, especially as an initial warning sign, in an otherwise previously healthy neonate, has never been reported (3).
A retropharyngeal abscess (RPA) is a deep neck space infection and should be considered in the differential diagnosis of a child with fever, decreased oral intake, and respiratory distress. It can become potentially life-threatening from upper airway compression or seed into the mediastinum causing severe sepsis (3,4). The general appearance of a child with RPA ranges from an irritable child with minimal neck tenderness to a more toxic appearance with a palpable neck mass and stertor or stridor. Physical examination may include a posterior bulging of the oropharyngeal wall. RPAs are not uncommon in the pediatric population and can be found in children typically two to four years old. However, they are remarkably rare in infants under six months (4). Our patient’s extremely unusual presentation of bradycardia triggered solely by head-turning provided a great challenge to the clinical team necessitating an “outside-the-box” mentality to ultimately confirm the diagnosis.
Case Presentation
The patient is a 40-day-old male born at 37 weeks gestation via cesarean section for breech presentation to a 37-year-old G2P1 mother, with APGAR scores of 9 and 9 at one and five minutes, respectively. Mother had an uncomplicated pregnancy with the exception of gestational diabetes, which was well controlled with insulin. There were no clinical findings associated with chorioamnionitis, including no maternal fever during delivery. His birth weight was 3695 grams (62% percentile), and he had an uncomplicated delivery requiring typical newborn resuscitation. He was uneventfully discharged on day-of-life (DOL) 2 with normal physical exam findings and was breastfeeding/latching appropriately. He was seen by his pediatrician on DOL 4 and was noted to be breastfeeding or formula-feeding one to two ounces every two to three hours with an appropriate number of wet diapers and bowel movements.
On DOL 11, parents sought medical care due to decreased intake by 50% and were advised to monitor for signs of dehydration. He was seen for another well visit on DOL 24, where his weight was 4014 grams. Parents sought care again on DOL 35 due to worsening feeding (only taking ~25% of total feeds). He was noted to have nasal congestion but was otherwise well appearing. The parents went to urgent care on DOL 36 for ongoing congestion, poor feeding, and new onset emesis and were promptly sent to the emergency department (ED). There was no fever or cyanosis. Respiratory syncytial virus (RSV), influenza, and COVID-19 tests were negative. Otolaryngology (ENT) was consulted, and bedside flexible laryngoscopy revealed no significant airway findings. The patient maintained oxygen saturation≥ 95% and was discharged home with reflux precautions, humidifier use recommendation, and strict return precautions.
On DOL 40, the patient continued to have persistent vomiting prompting another return to the pediatrician. Chest x-ray was normal, and oxygen saturation remained above 95%, but he was referred back to the ED for admission due to leukocytosis (WBC=22K), hyponatremia (Na=126), and concern for severe dehydration with respiratory fatigue. In the ED, several episodes of bradycardia (heart rate as low as 70) and apnea with associated hypoxemia (as low as 70%) were noted with mild recovery that eventually required non-invasive positive pressure ventilation. He was admitted to the Pediatric Intensive Care Unit (PICU) after initiation of sepsis workup, including blood culture, urine culture, and lumbar puncture. He was started on empiric IV ceftriaxone and vancomycin. Initial inflammatory markers were noted to be low-normal with CRP 2.0 (reference range <5.1 mg/L) and procalcitonin 0.21 ng/mL (normal <0.08 ng/mL). Electroencephalogram (EEG) monitoring began due to concern for seizures in the context of apnea. EEG showed no electrographic seizures despite multiple subsequent witnessed bradycardic and apneic episodes. Echocardiogram did not reveal structural heart abnormalities. Blood culture from admission remained negative to date, and lumbar puncture results were reassuring, with no concerns for underlying meningitis or viral encephalitis. An immunologic workup was initiated simultaneously to assess for underlying primary immunodeficiency, and the results were negative.
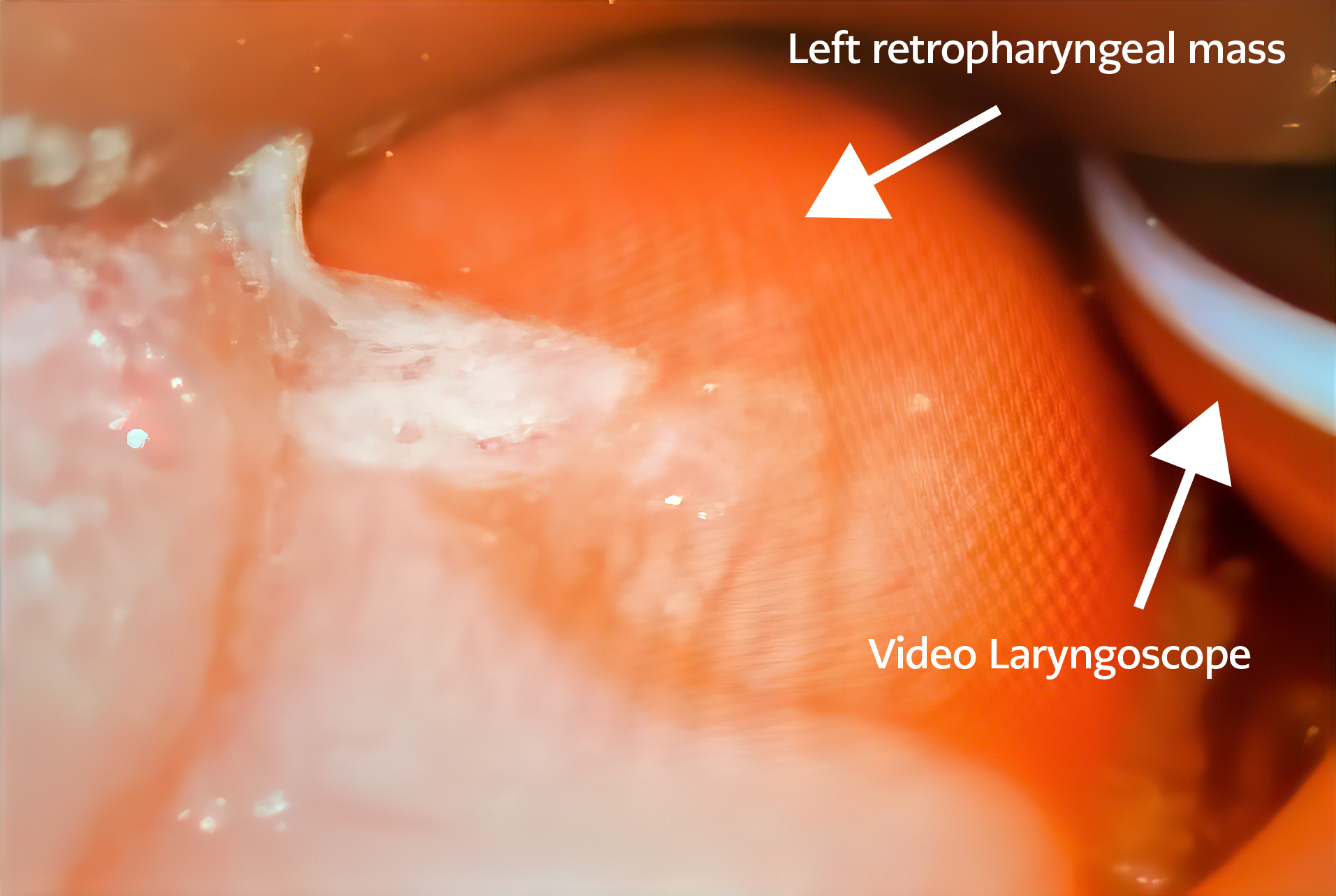
Figure 1. The operating room findings of the direct laryngoscopy of the left retropharyngeal mass (white arrow) causing near-complete collapse of the upper airway.
On closer inspection, it was noted that the bradycardic episodes were triggered only when the head was turned to the left and followed soon thereafter by apnea and hypoxemia. Apnea and desaturations never preceded bradycardia. No findings occurred with head turns toward the right. Ultrasound of the bilateral neck was also unremarkable. Repeat complete blood count (CBC) revealed increased leukocytosis (WBC=33K). Further neurologic and vascular imaging was recommended after a multidisciplinary discussion between PICU, ENT, neurology, and infectious diseases. The patient was semi-electively intubated on hospital day (HD) one in the late evening for protection of the airway and planned diagnostic imaging requiring sedation. Despite intubation, the patient continued to experience increasingly frequent bradycardic episodes down to the 60s with desaturations to the 50s. The patient was taken emergently to the operating room (OR) by pediatric ENT in the early morning of HD two for laryngoscopy and bronchoscopy and found to have a large, left-sided retropharyngeal mass (Figure 1).
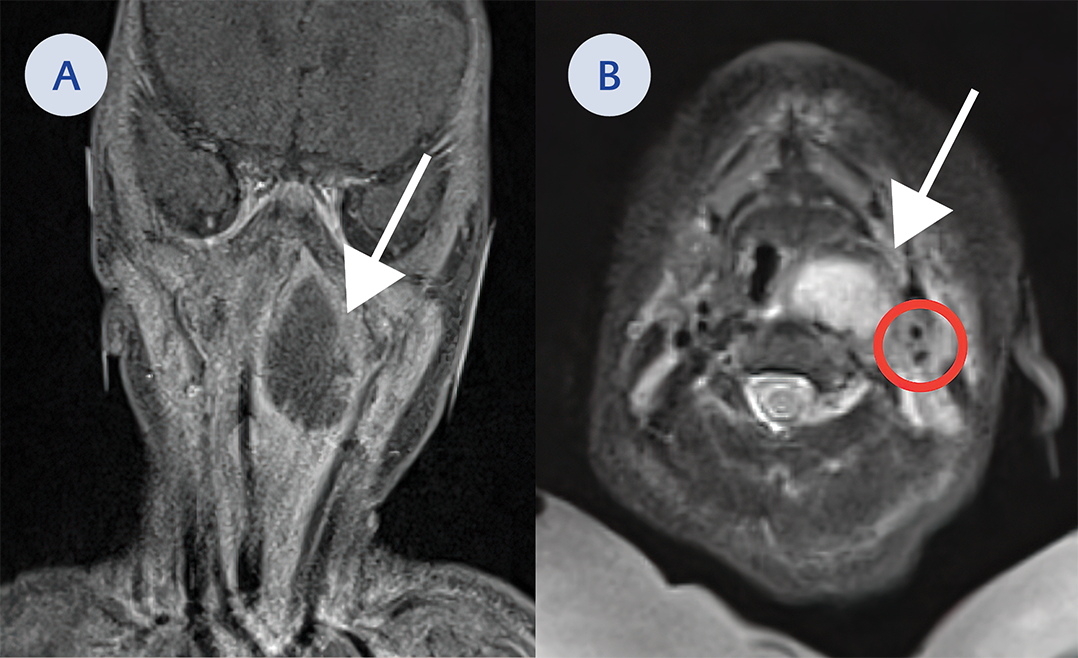
Figure 2. A. Coronal cut on MRI of a large mass in the left retropharyngeal space (white arrow). This extends from the nasopharyngeal region down to the level of the hypopharynx and just to the margin the larynx. It measures approximately 2.6 cm side to side by 1.8 cm anterior posteriorly and 3.1 cm craniocaudally. B. Axial cut on MRI of large mass in the left retropharyngeal space (white arrow). Red circle indicate location of the left carotid in relation to abscess.
Subsequent magnetic resonance imaging (MRI) of the neck delineated a large 2.6 cm cystic lesion in the left retropharynx consistent with an abscess with ipsilateral lymphadenopathy (Figure 2A, Figure 2B). The patient was taken back to the OR for incision and drainage (I&D). A copious amount of pus was expressed and sent for microbiologic cultures (Figure 3A).
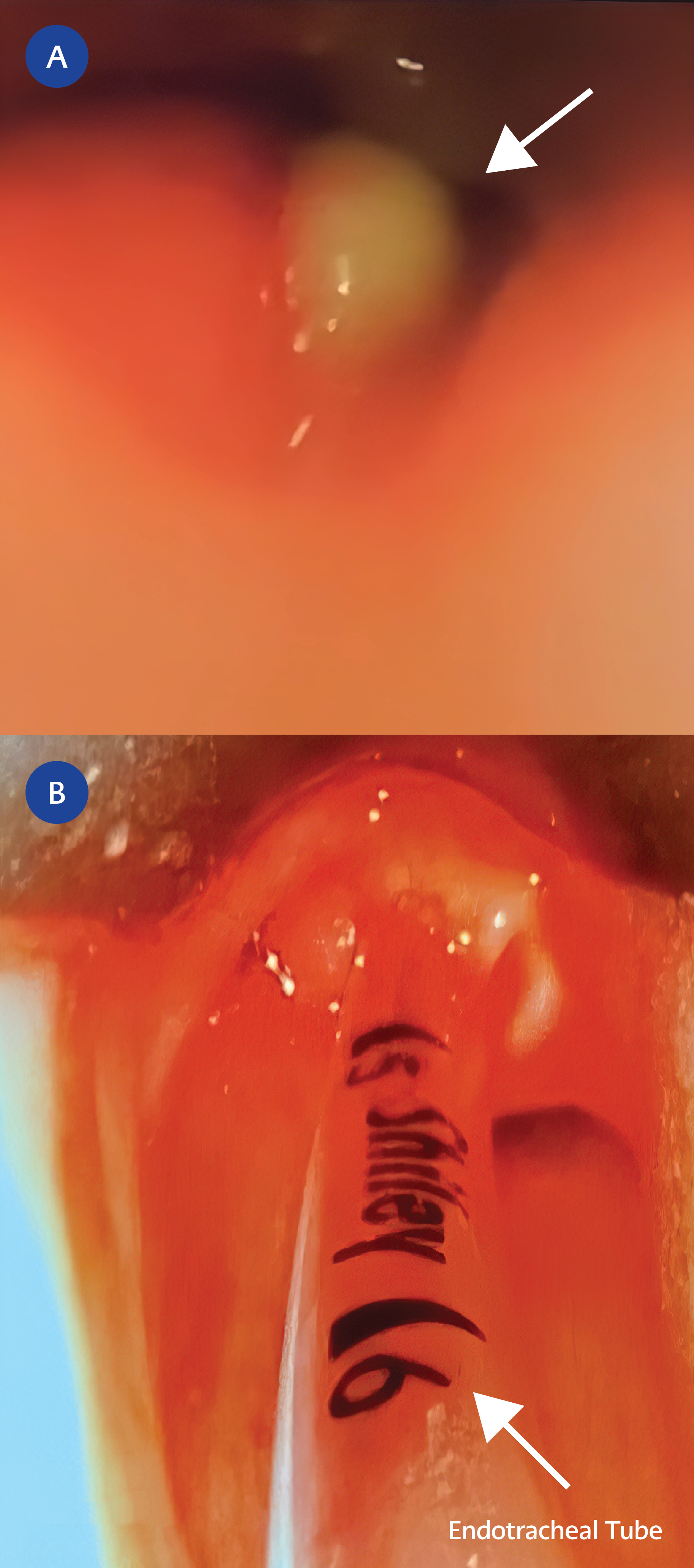
Figure 3. A. Status post incision and drainage of left retropharyngeal abscess with copious purulence (white arrow). B. Bedside direct laryngoscopy findings forty-eight hours after incision and drainage showing near resolution of the abscess and a more patent airway
The patient remained afebrile with improving leukocytosis following I&D. Abscess culture grew 4+ methicillin-resistant Staphylococcus aureus (MRSA), and the patient was continued on intravenous vancomycin. The abscess culture showed that the isolate was highly susceptible to clindamycin, gentamicin, tetracycline, trimethoprim-sulfamethoxazole, and vancomycin. As the drug of choice for invasive MRSA infections remains vancomycin, we continued therapy with this agent, optimizing therapeutic trough levels to maintain between 10-15 mcg/mL. Of note, initial nasal surveillance for MRSA screening was also positive. The patient remained intubated for forty-eight hours post-operatively, and a bedside direct laryngoscopy showed mild left lateral oropharyngeal swelling and slight eschar. Exploration of the abscess cavity revealed no further purulence (Figure 3B).
The patient was successfully extubated 72 hours after I&D on HD five and continued to recover. He was transitioned to enteral clindamycin for a total of 10 days. He had no additional bradycardic or apneic events, especially with position changes. He was discharged to home on HD eleven. Infectious diseases elicited a recent history of MRSA digit cellulitis in the patient’s father with recommendations for the decolonization of the father and all other family members, including the patient, with mupirocin applied to the nares twice a day for five days. Additional recommendations for decolonization with 2% chlorhexidine gluconate wipes were also provided as part of standard PICU protocol for the patient during hospitalization. Further detailed recommendations, including hygiene measures for MRSA colonization, were provided to the patient’s family during outpatient pediatric infectious disease follow-up.
Discussion
This clinical case is the first report of an infant with hemodynamically significant and positional bradycardia from an evolving RPA. There have been previous documented case reports of MRSA RPAs, with a mean age of 5.6 months (5-8). Most frequently, the causative agents of RPA include S. aureus and group A β-hemolytic streptococcus (4,9-14). RPAs generally develop secondary to a precursory infection but can result from trauma (15). In our neonate, normal resuscitative measures were taken during the newborn period, and routine nasal suctioning was employed in the weeks prior to admission. Most children will have a constellation of symptoms, including fever, toxic appearance, irritability, and respiratory distress. In our case, the patient exhibited irritability with cardiopulmonary changes instigated by positional head-turning to the left. The etiology of the positional bradycardia with the left head turn was likely due to the compression of the carotid bulb triggering the baroreceptor reflex via the glossopharyngeal nerve.
On admission to the PICU, a broad differential diagnosis was considered due to the irregularity of symptoms, including sepsis/meningoencephalitis, seizure disorder, central hypoventilation syndrome, and eventually structural airway disease such as tracheoesophageal fistula. Due to the finding of positional bradycardia and after having concurrently ruled out other causes, airway malformation or obstruction was highly suspected despite negative neck ultrasound findings. Although ultrasound may have some utility in diagnosing deep space infections in pediatric patients, its diagnostic sensitivity in this population is limited to a small number of case reports (16). Ultrasound is also highly operator dependent. Computed tomography (CT) is usually the preferred imaging modality for detecting RPA but is not without reservations in the pediatric population due to radiation exposure. Sensitivity ranges from 69-100%, with a more variable specificity ranging from 45-100% (17-20). Previously, lateral neck x-ray was utilized in diagnosing an RPA, but currently, there is no consensus on its utility and a paucity of recent literature to support its use (21).
Complications of RPA include airway compromise, perforation, mediastinitis, septic shock, and aspiration pneumonia (8). In this report, our patient required intubation to secure the airway. After an oropharyngeal mass was found in the OR with direct laryngoscopy, the patient underwent an MRI to assist in delineating mass characteristics and diagnosis. MRI provided superior soft tissue definition and clearer distinction of vascular involvement compared to CT scan. MRI helped distinguish the mass from a paraganglioma or other tumor, aortic arch/great vessel anomaly, and an abscess without subjecting the patient to radiation.
Notable in the patient’s family history was that the father had a recent MRSA hand infection with near amputation. We suspect that the infant’s invasive MRSA infection was potentially a result of an unfortunate combination of MRSA nasal colonization, young age, and a more virulent strain of S. aureus. Maternal nasal screening was not performed, and no additional strain typing was performed on the patient’s intraoperative MRSA cultures. However, in one large study from a university-affiliated neonatal intensive care unit (NICU), the USA300 strain of MRSA identified via pulsed-field gel electrophoresis (PFGE) was found to be most common amongst colonized or infected neonates. There is a strong association between strains found in neonates causing subsequent infections; therefore, many NICUs and PICUs have implemented universal decolonization as an important MRSA infection prevention strategy (22).
Lastly, underlying primary immunodeficiency was considered, but less likely, given the presentation, father’s history of recent MRSA infection, and previous normal newborn screen (including severe combined immunodeficiency [SCID] by measurement of T-cell receptor excision circles). Nonetheless, further immunological testing was performed as an outpatient, and the patient had normal quantitative immunoglobulin levels, T-cell and B-cell subsets, and a negative DHR (dihydrorhodamine 123 test) flow testing for chronic granulomatous disease. Additionally, repeat nasal surveillance cultures for MRSA were negative for ongoing bacterial colonization with MRSA at 12 weeks post-hospitalization and after the initial decolonization protocol was completed.
In conclusion, we present a highly unusual case of a MRSA RPA masquerading as positional bradycardia followed by apnea. This clinical case provides an abundance of learning: 1) Although ENT was consulted earlier in this patient’s course, early phlegmon or abscess can be difficult to visualize via physical examination or flexible endoscopy, especially in awake infants. Persistent symptoms and disease progression warranted re-evaluation and, ultimately, operative endoscopy despite prior negative findings. 2) Children often do not follow the textbook (bradycardia followed by apnea vs. the opposite). This case was remarkably challenging due to the protracted history presented and unusual symptoms; critical thinking of sequential events will almost always help narrow one’s primary differential diagnosis. 3) A multidisciplinary team approach and collaboration (in our case – between critical care medicine, ENT, infectious diseases, and neurology) is undeniably paramount in diagnosing rare pediatric illnesses.