Introduction
Coronavirus disease 2019 (COVID-19) emerged in Wuhan, China, at the end of 2019 with pneumonia cases and spread worldwide in a short time, leading to a pandemic. The cause of the disease was severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The World Health Organization (WHO) declared a pandemic on March 11, 2020. The WHO reported 293,750,692 cases and 5,454,131 deaths worldwide as of January 2022 (1).
The course of COVID-19 infection can vary from asymptomatic disease to severe pneumonia, respiratory failure, and critical disease. The disease is diagnosed by detecting viral RNA by the polymerase chain reaction (PCR) method. The most common symptoms of the disease have been reported as fever, weakness, cough, phlegm, muscle aches, loss of appetite, and shortness of breath (2). However, there are also patients with atypical presentations involving gastrointestinal, neurological, cardiac, and dermatological complaints (3-6). Although parotitis development during the course of COVID-19 is rare, cases that reveal this relationship have been reported in the literature (7-9). This report presented a case of bilateral acute non-suppurative parotitis associated with COVID-19.
Case Presentation
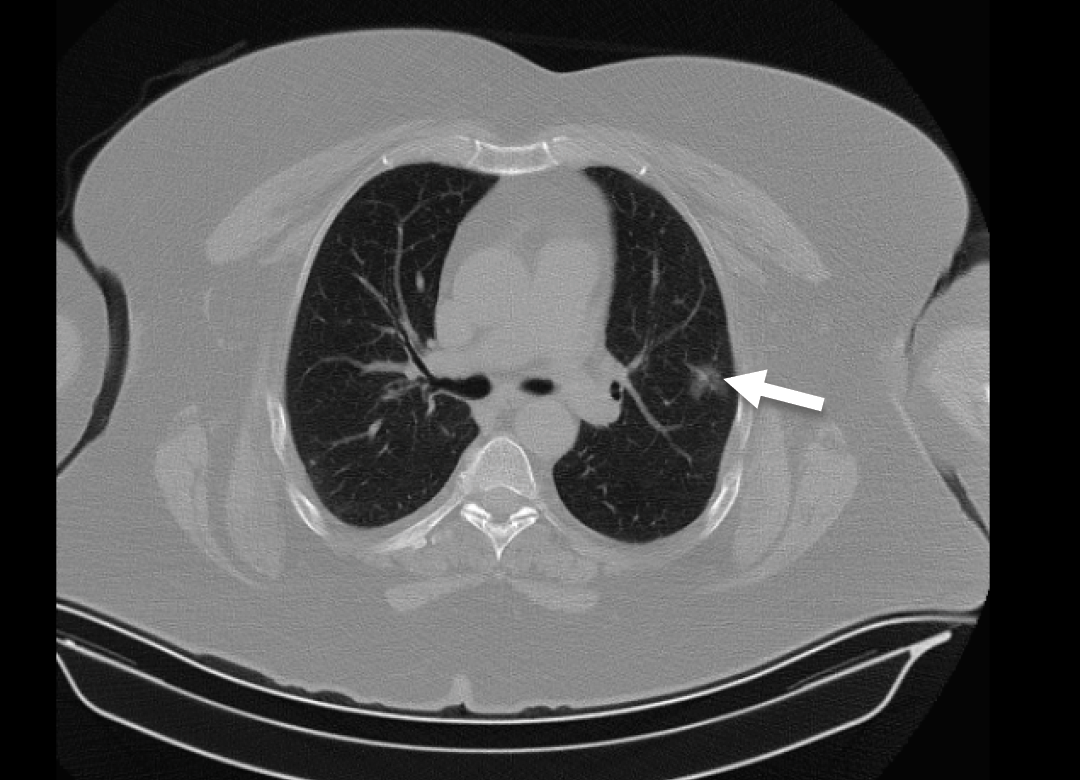
A 63-year-old female patient was admitted to the emergency room with complaints of cough, shortness of breath, muscle-joint pains, and weakness. She had previously known diagnoses of diabetes mellitus and hypertension. It was learned that the patient tested positive for the SARS-CoV-2 PCR test six days ago and had been taking favipiravir treatment at home for five days, but her complaints had worsened despite this treatment. Her temperature was 36.8 °C, and her vital signs were stable. Arterial oxygen saturation by pulse oximetry (SpO2) was 91%. There were no pathological findings in the physical examination. In her blood tests, the following levels were measured; leukocyte count: 3810/mm3, platelet: 126,000/mm3, creatinine: 1.12 mg/dL, AST: 26 u/L, ALT: 19 u/L, CRP: 60 mg/L, procalcitonin: 0.06 ng/mL, ferritin: 82 µg/L, D-dimer: 319 ng/mL. Thorax computed tomography (CT) scan showed an area in the lingular segment of the left lung with a ground glass density about 2 cm in size (Figure 1). The patient was admitted to the ward for follow-up and treatment. Methylprednisolone, lansoprazole, inhaled beta-2 agonist, and inhaled corticosteroid treatments were initiated for the patient who had dyspnea and hypoxia on pulse oximetry. Enoxaparin treatment was added to the treatment, as it was recommended for all hospitalized patients diagnosed with COVID-19 by the Ministry of Health guidelines for COVID-19(10).
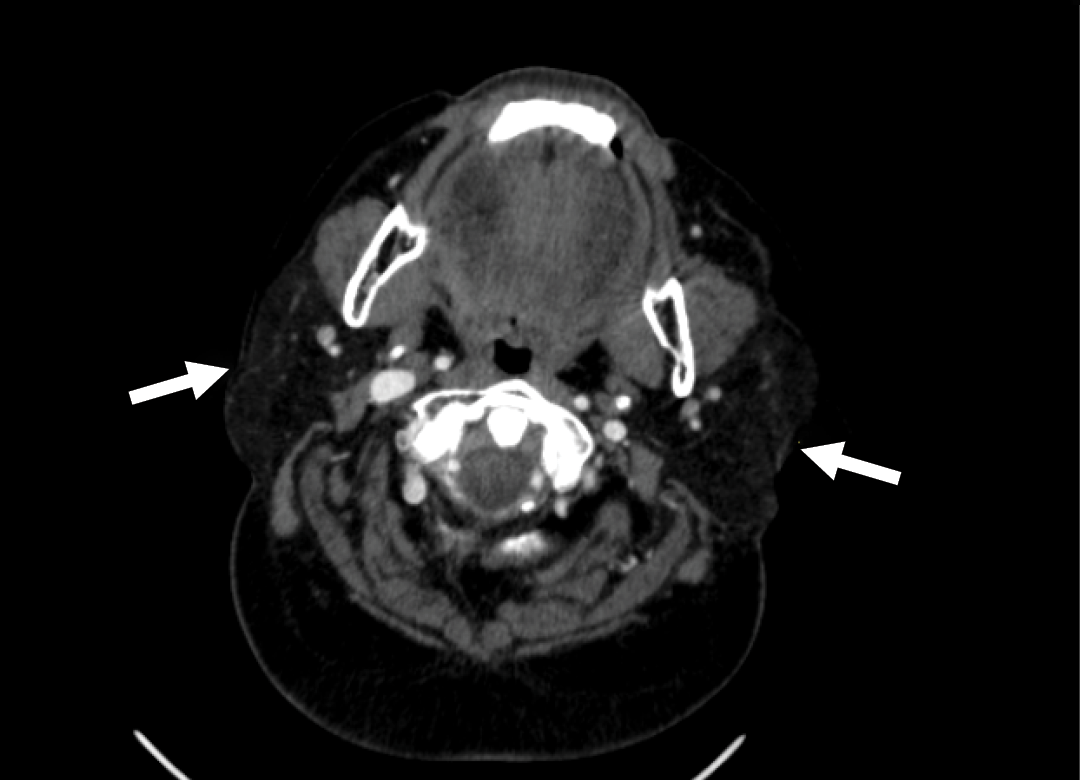
Figure 2. Edema, size increase, and decreased density in both parotid glands on the contrast-enhanced neck CT (arrows).
On the seventh day of her hospitalization, swelling, tenderness and pain developed in both submandibular areas and parotid gland sites. In the ultrasonography, the thickness of both parotid glands was increased, and the parenchyma was observed with heterogeneous appearance with edema. It was interpreted as parotitis. In the contrast-enhanced neck CT, edema, size increase, and decreased density was detected in both parotid glands (Figure 2). Meanwhile, bacterial parotitis was not considered primarily for the patient as she had no fever, leukocytosis, high CRP level, and also no pus leaking from the parotid canal was observed with the parotid massage. The serological tests were negative for acute infection for mumps, measles, rubella, cytomegalovirus, Epstein-Barr virus (EBV), viral hepatitis, and human immunodeficiency virus (HIV). Multiplex PCR tests for respiratory pathogens taken by nasopharyngeal sampling were negative for influenza virus A-B, parainfluenza virus, rhinovirus, enterovirus, human metapneumovirus, adenovirus, non-SARS-CoV-2 coronaviruses. COVID-19 infection was considered the cause of acute parotitis. Upon improvement of the patient who was treated with symptomatic and anti-inflammatory treatment, the patient was discharged.
Discussion
The etiology of acute parotitis includes infections (especially viral infections) and other noninfectious inflammatory causes. Mumps is the most common viral cause of acute parotitis. Other viral pathogens include influenza, parainfluenza, HIV, coronaviruses, enteroviruses, EBV, herpesvirus, and coxsackievirus. Viral parotitis is characterized by swelling and pain in the parotid gland after a few days of prodromal period with flu-like complaints. About 75% of cases are bilateral (11).
Saliva samples of patients with COVID-19 have a high viral load for SARS-CoV-2 (12). An animal study noted that SARS-CoV shows tropism on epithelial cells containing angiotensin-converting enzyme II (ACE-2) in the salivary glands and that the virus may infect the salivary glands (13).
In the literature, cases of acute non-suppurative parotitis due to COVID-19 have been reported (7-9, 14, 15). In addition, there is the case of a 4-year-old boy reported within our country (16). In all cases, complete recovery was reported with symptomatic treatment. As far as we know, our case is the first case of COVID-19-related parotitis reported from our country in the adult age group. After the negative results of the examinations regarding factors that may cause viral parotitis, it was thought that acute parotitis was COVID-19 related. In this study, unlike the cases in the literature, parotitis development was observed not in the early period after SARS-CoV-2 tested positive but about 12 days later.
Additionally, while almost all reported cases were unilateral, our case demonstrated a bilateral course. Because our case was bilateral, there were no systemic findings such as fever, no laboratory findings that may be associated with bacterial infection, and no abscesses in imaging; thus, viral parotitis was primarily considered. We initiated symptomatic treatment and anti-inflammatory treatment. Without any antibacterial treatments, clinical improvement was achieved.
Conclusion
As a result, as long as the COVID-19 pandemic continues unabated, patients with atypical presentations and clinical manifestations will continue to increase. COVID-19-related parotitis cases are one of these. This case report emphasized that acute non-suppurative parotitis may develop due to COVID-19 infection. Therefore, it should be kept in mind that identifying and providing the isolation of these cases and other similar cases of atypical COVID-19 infection shall contribute to reducing the spread of the disease.