Introduction
In the COVID-19 pandemic, it is essential to estimate the immune responses to various COVID-19 vaccinations to estimate the efficacy of the vaccines. During the ongoing pandemic, different vaccine types and vaccination strategies affect the cellular and molecular elements of innate and adaptive immune systems. Therefore, testing the effectiveness of COVID-19 vaccines in different vaccination schedules is necessary.
Memory B cells pose CD19+, CD20+, and CD27+ receptors. The IgG-positive memory cells induce the formation of IgG-secreting plasma cells even in the absence of T-cell help (1). The CD38+ B cells can be activated faster than CD38-memory B cells which are important in secondary immune response to antigens (2).
Initially, Turkey adopted two doses of inactivated COVID-19 vaccination as the primary vaccination strategy. In the course of the pandemic, the booster vaccination with inactivated vaccine, CoronaVac (Sinovac Life Sciences, China), or mRNA vaccine, BNT162b2 (Pfizer-BioNTech), is added to the vaccination schedule. The population was vaccinated either with CoronaVac or BNT162b2 booster. We aimed to investigate the effects of homologous and heterologous vaccination on the expression levels of different B cell subtypes.
Materials and Methods
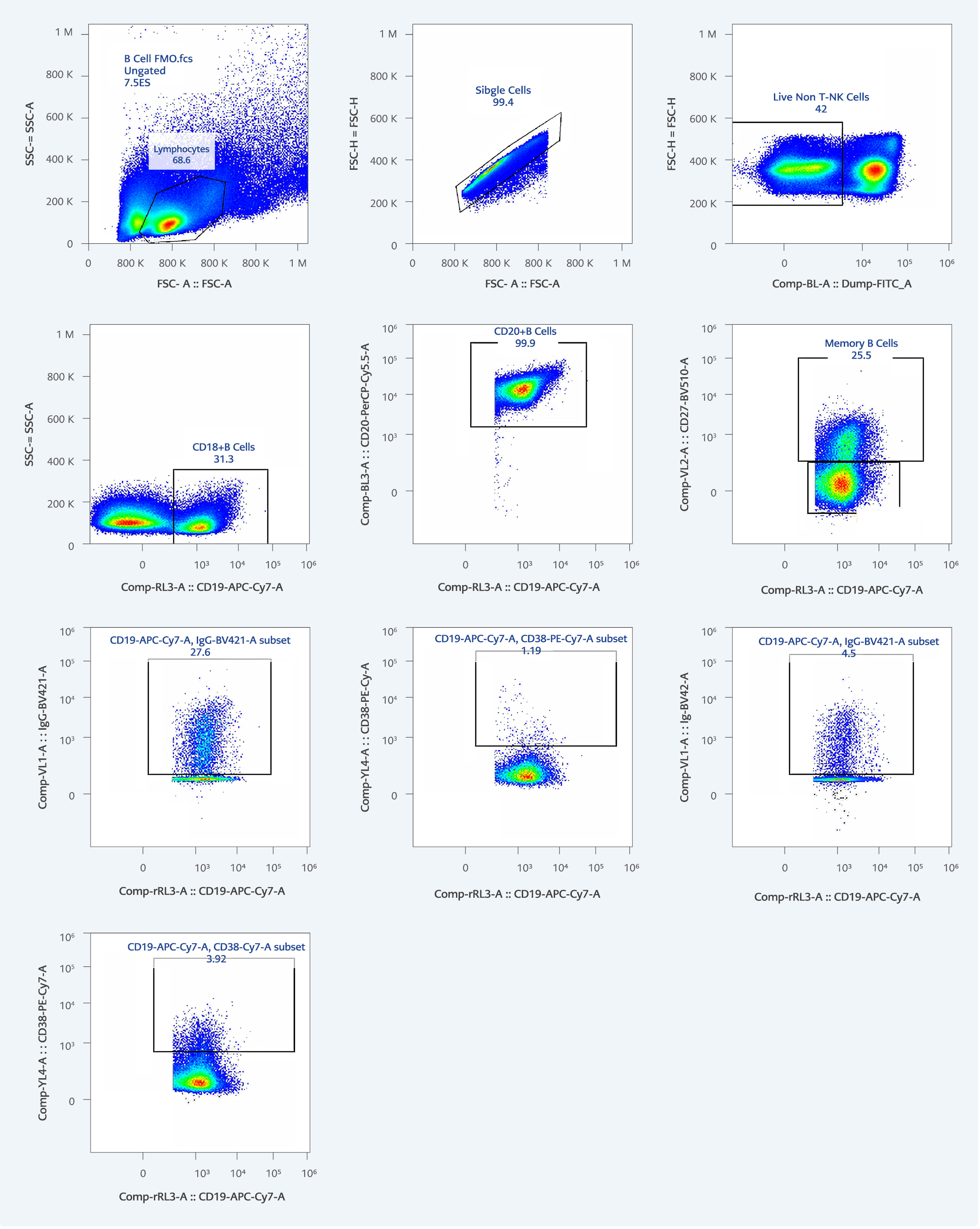
Whole blood samples were collected 3-5 months after two doses of CoronaVac vaccination (baseline) from 35 individuals from a tertiary hospital. Among these, in the first month, 28 received BNT162b2 booster, and seven of them received CoronaVac booster. In the third month, blood samples were collected from six BNT162b2 receivers and four CoronaVac receivers. The longitudinal blood samples were obtained from the same participants one and three months after the boosters. Peripheral blood mononuclear cells (PBMCs) were isolated by lymphoprep density gradient centrifugation (3). PBMCs were frozen at -80°C until use, after thawing cells were stained with viability dye-zombie green (Biolegend-423112) and then with specific B cell markers, CD19-APC-Cy7 (Biolegend-302218), CD20-PerCp-Cy5.5 (Biolegend-302326), IgG-brilliant violet 421 (Biolegend-410704), CD27-brilliant violet 501 (Biolegend-302836), CD38-PE-Cy7 (Biolegend-303516), CD14-FITC (Biolegend-325604), CD56-FITC (Biolegend-318304) and CD3-FITC (Biolegend-344804). The samples were run using Attune flow cytometer (Thermo Fisher Scientific Inc, USA). By gating, CD19+CD20+CD27-, CD19+CD20+CD27+, CD19+CD20+CD27+IgG+, and CD19+CD20+CD27+CD38+ B cell ratios were determined (4). In the gating respectively, lymphocytes, single cells, live non-T-NK cells, CD19+ B cells, CD20+ B cells, memory B cells, CD19+ B cells expressing IgG, and CD19+B cells expressing CD38 cell populations were analyzed as shown in Figure 2. This study complies with the Declaration of Helsinki and is approved by the Koç University Ethical Committee with decision number 2021.151.IRB.055 on August 10, 2021. The informed consent forms were collected from all participants.
Statistical Analysis
For comparison of independent groups, an unpaired non-parametric test, the Wilcoxon rank-sum (Mann-Whitney U) test was performed. Prism 8.0.2 software (GraphPad Software, USA) was used for the analysis and visualization. Statistical significance was set as p<0.05. The statistical analysis was performed according to our previous study (8).
Results
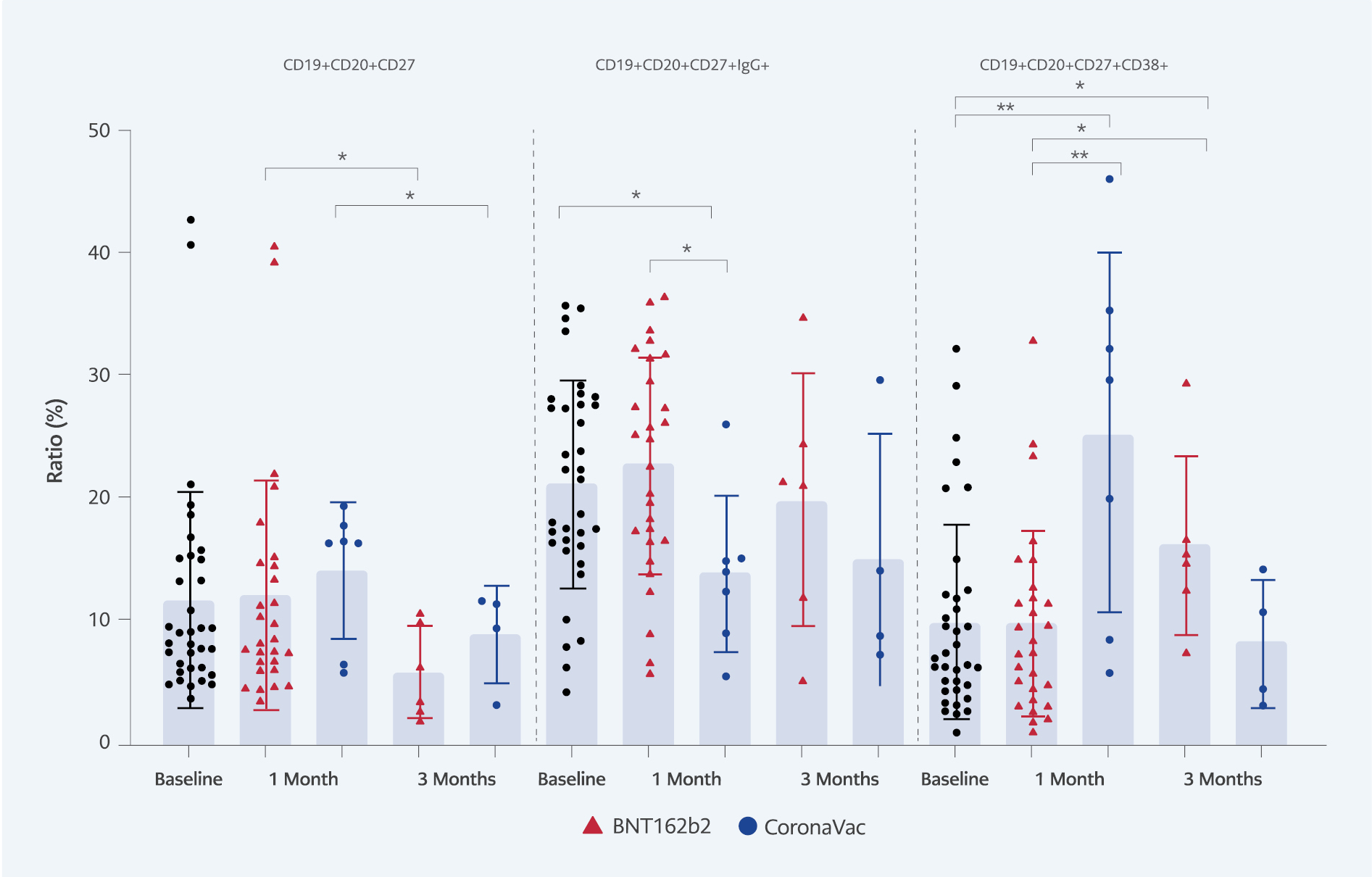
Figure 1. The ratio of the B cell subtypes (CD19+CD20+CD27+, CD19+CD20+CD27+IgG+, CD19+CD20+CD27+CD38+) in baseline, one month and three months after the booster vaccination.
The mean of baseline naïve B cells (CD19+CD20+CD27-) was 72.05%. The baseline level of memory B cells (CD19+CD20+CD27+) was 21.21%, and after booster doses with both vaccine types, there was no significant change in the memory B cell ratios in one month. However, a significant decrease was detected in BNT162b2 and CoronaVac groups in three months (5.98%, 9.00%), respectively. In the IgG expressing memory B cell population (CD19+CD20+CD27+IgG+), the mean level of the cells was significantly higher with BNT162b2 booster (22.70%) than CoronaVac (13.96%) in one month (p=0.01). In the effector memory B cell population (CD19+CD20+CD27+CD38+), the baseline level was 10.05% and the cell ratio significantly increased to 25.44% in one month after CoronaVac (p=0.0072). However, the CD19+CD20+CD27+CD38+ cell ratio declined to 8.24% in three months. BNT162b2 booster stimulated effector B cell population CD19+CD20+CD27+CD38+ in three months (16.21%) (Figure 1).
The flow gating strategy is shown in Figure 2.
Discussion
This study presented significant stimulation of CD19+CD20+CD27+CD38+ effector memory B cell populations after CoronaVac and BNT162b2 boosters. The CoronaVac booster stimulated cells in one month, but the number of cells decreased in three months. The CD19+CD20+CD27+CD38+ cell stimulation with BNT162b2 was slower than CoronaVac. The higher ratio of IgG-expressing B cells was detected after BNT162b2. Although there was no change in CD19+CD20+CD27+CD38+ memory B cell ratio in one month, a significantly higher memory B cell population than baseline and CoronaVac booster was observed in three months. Higher expression of CD38 shows that when fast and high amounts of IgG antibody will be produced (5, 6). After a full three doses of BNT162b2 vaccination, the subsequent increase in memory b cell subpopulation was shown (5). For the SARS-CoV-2 specific total memory B cells after two doses of CoronaVac and one dose of BNT162b2 booster, a significant decrease was shown by Prasithsirikul et al. (7).
In conclusion, we suggest that both CoronaVac and BNT162b2 boosters stimulate effector memory B cell responses. CoronaVac booster induces fast but unsustainable effector memory B cell response. Even though the memory B cell response is slower after the BNT162b2 booster, the high proportion of the cells in three months indicates a sustained long-term B cell memory activity after the BNT162b2 booster. Our study indicated that an additional BNT162b2 booster dose for individuals who already had CoronaVac vaccination is recommended. The long-term efficacy of booster against variants should be tested in the future.