Introduction
Chronic lymphocytic leukemia (CLL) is characterized by the clonal proliferation of mature-appearing neoplastic B lymphocytes in lymphoid tissues, including peripheral blood, bone marrow, lymph nodes, spleen, and liver (1). Hypogammaglobulinemia, along with functional impairments in T cells and certain subgroups (such as neutrophils and monocytes), suppresses both humoral and cellular immunity in CLL (2). Furthermore, in CLL, the activation of the B-cell receptor (BCR) promotes the uncontrolled proliferation of malignant B cells.
Ibrutinib, a kinase inhibitor used in the treatment of refractory CLL, irreversibly inhibits Bruton’s tyrosine kinase, the cytoplasmic enzyme of the BCR. By inhibiting B-cell proliferation, development, and viability, it disrupts the effects of the tumor microenvironment accompanied by immunosuppression, which leads to increased susceptibility to opportunistic infections such as disseminated nocardiosis (3).
This report discusses a case of disseminated nocardiosis (pneumonia and brain abscess) caused by Nocardia farcinica in a patient treated with ibrutinib for refractory CLL.
Case
A 67-year-old male patient presented with complaints of fever, cough, sputum production, shortness of breath, hoarseness, weight loss, and loss of appetite for one month. His medical history revealed a diagnosis of CLL, chronic obstructive pulmonary disease (COPD), coronary artery disease, and a two-year history of ibrutinib use.
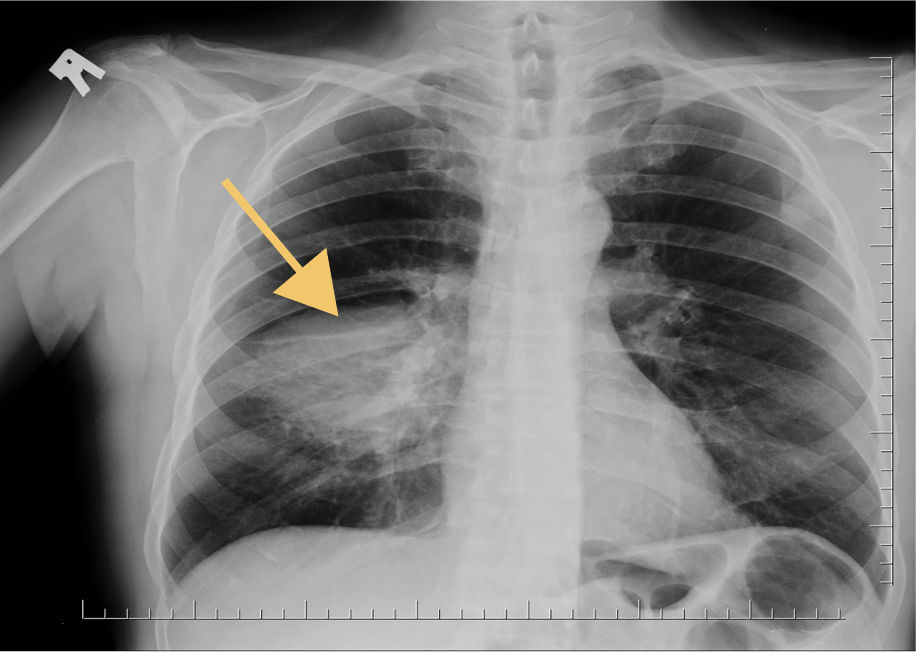
On physical examination, the patient’s general condition was good, and vital signs were within normal limits. A chest X-ray revealed consolidation in the right middle lobe (Figure 1), which was interpreted as consistent with lobar pneumonia. The patient was hospitalized and treated with levofloxacin and piperacillin-tazobactam. Upon observing a clinical and laboratory response to the initiated treatment, the patient was discharged with a prescription for levofloxacin.
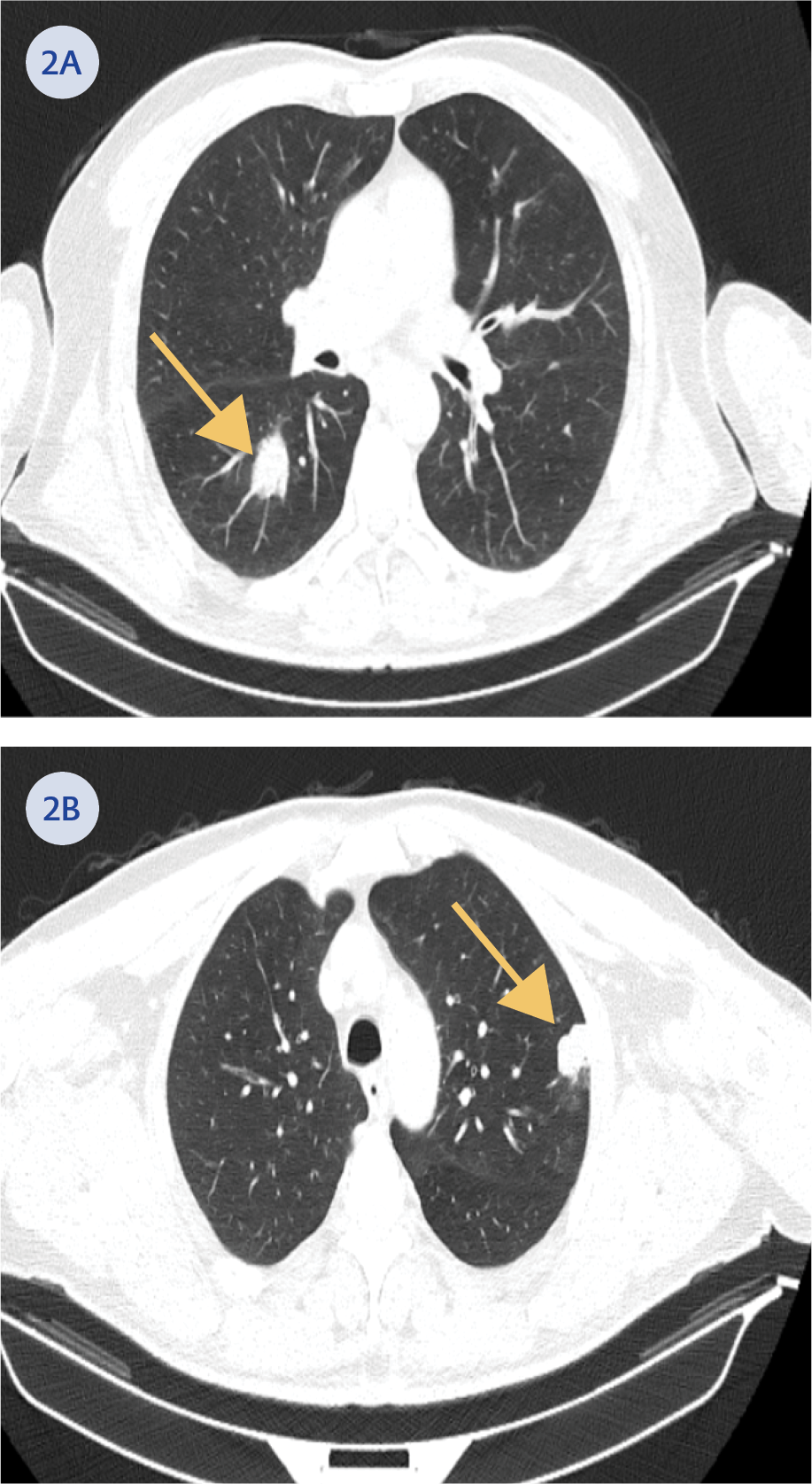
During a follow-up visit one week later, the patient reported persistent complaints of cough and fatigue. Physical examination revealed decreased breath sounds in the right middle lobe of the lung, and chest X-ray demonstrated persistent consolidation. The electronic personal health record system (e-Nabız) was reviewed. A thoracic computed tomography (CT) (Figures 2A, 2B) performed at an external center one month earlier revealed mass-like lesions in the lungs. To exclude possible malignancy and opportunistic infections, the patient was hospitalized and underwent bronchoscopy. During the procedure, bronchoalveolar lavage samples were collected from the right bronchial system (Table 1).
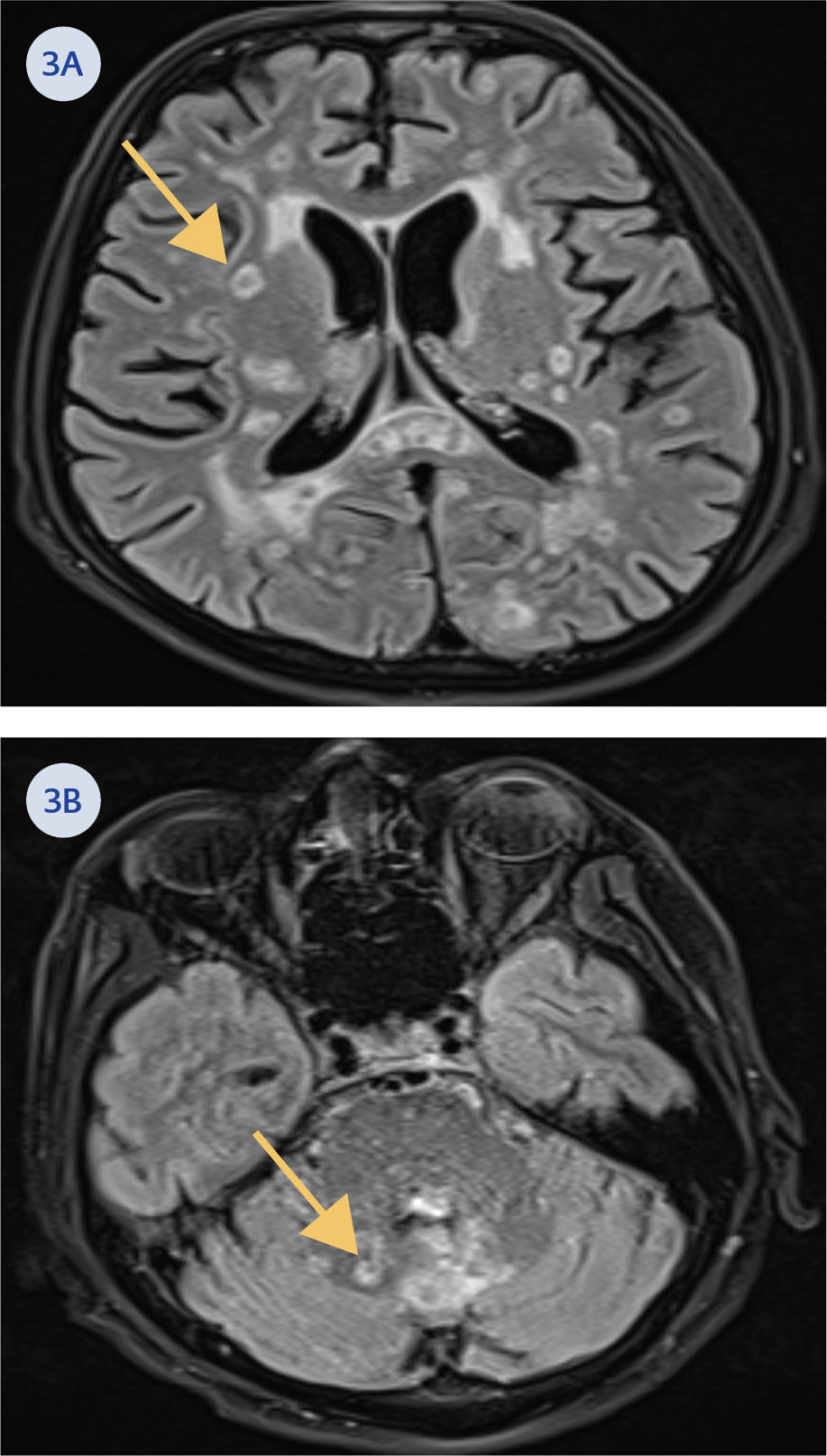
On the second day of hospitalization, the patient developed a fever (38.6°C), agitation, altered consciousness, and loss of balance. Blood and urine cultures were obtained, and contrast-enhanced brain magnetic resonance imaging (MRI) (Figures 3A and 3B), abdominal ultrasonography (USG), and transthoracic echocardiography (TTE) were performed. No growth was observed in blood and urine cultures, and the USG and TTE findings were normal. However, brain MRI findings raised suspicion of multiple millimetric pyogenic abscesses, leading to a lumbar puncture for cerebrospinal fluid (CSF) samples (Table 1).
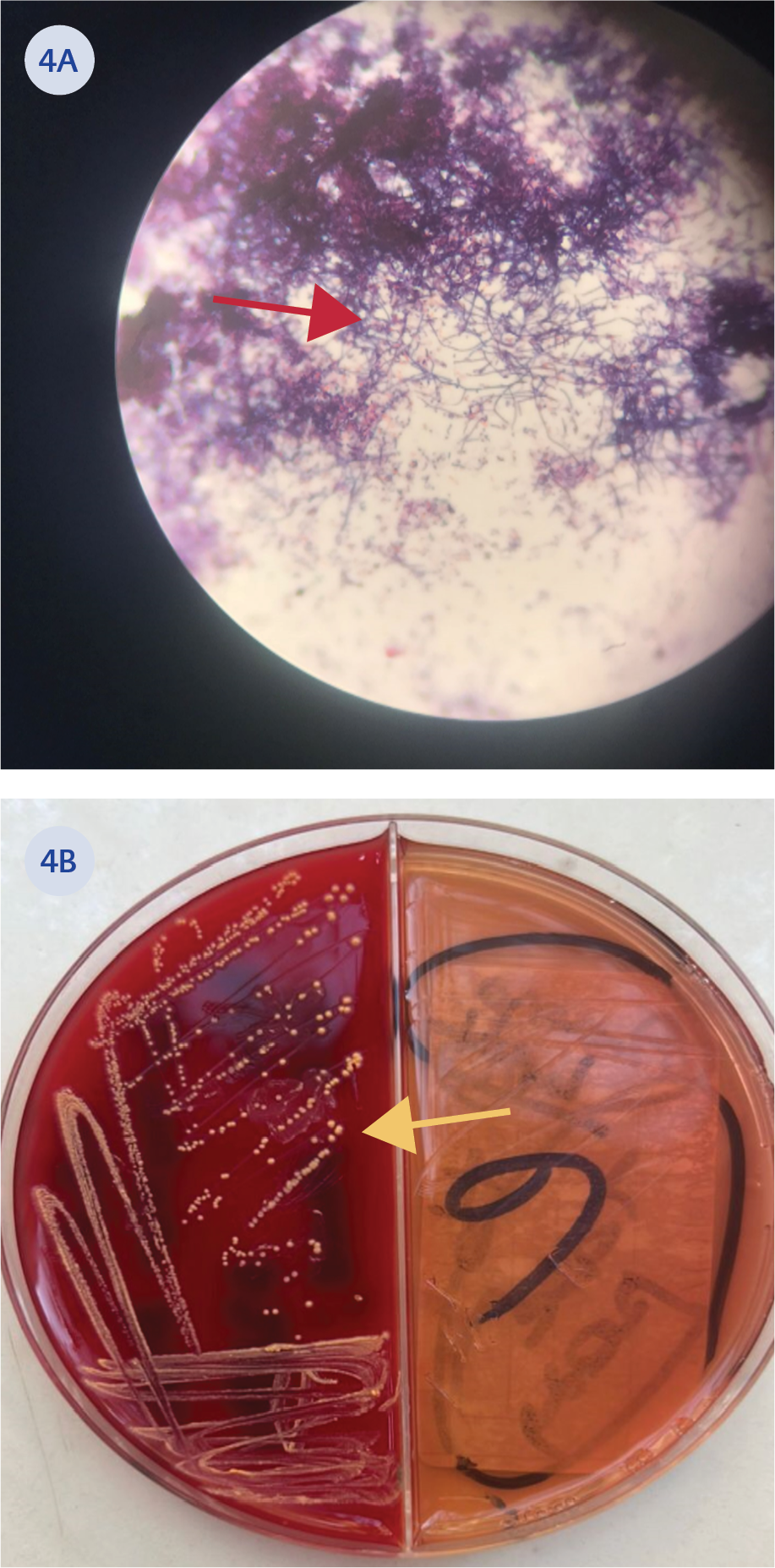
Figure 4. A) Brain tissue biopsy image of weakly stained bacilli with B) Nocardia colonies on blood agar
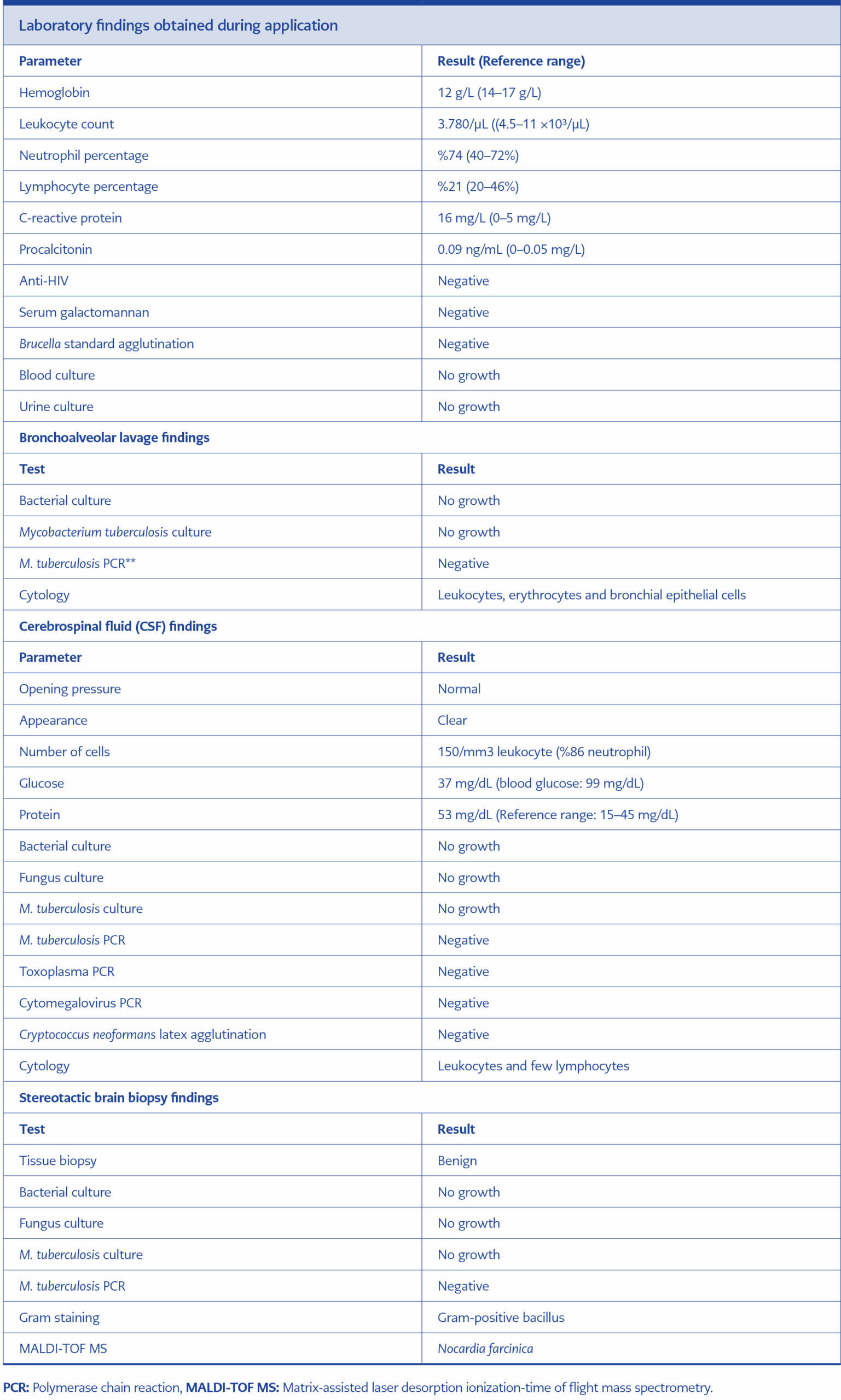
The MRI findings were considered suggestive of toxoplasmosis or aspergillosis. Therefore, the treatment was revised to include meropenem, linezolid, trimethoprim/sulfamethoxazole, and voriconazole. On the eighth day of empirical antibiotic treatment, stereotactic biopsy was performed on the brain lesions. Tissue Gram staining revealed Gram-positive bacilli, modified Erlich Ziehl-Neelsen staining demonstrated weakly acid-fast filamentous branching bacilli, and growth was observed on blood agar (Figures 4A and 4B). Microorganism identification from colonies grown on blood agar was performed using the matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) method, which identified the organism as N. farcinica. Antibiogram results indicated susceptibility to linezolid and intermediate susceptibility to ciprofloxacin. It was resistant to doxycycline, rifampicin, and trimethoprim/sulfamethoxazole.
The treatment was revised to include imipenem, trimethoprim/sulfamethoxazole, and amikacin. However, the patient’s condition progressively deteriorated, and he died on the 30th day of treatment.
Discussion
It has been reported that at least 50 species of Nocardia spp. cause diseases in humans, particularly in temperate and tropical climates. Nocardia spp. is an opportunistic bacterium found in the environment and not part of the human flora. It is considered a pathogen with a high risk of morbidity and mortality, especially in immunocompromised patients. Infection typically begins in the lungs following inhalation and subsequently spreads hematogenously to the central nervous system and other internal organs (4). The use of corticosteroids, calcineurin inhibitors, tyrosine kinase inhibitors, tumor necrosis factor-alpha (TNF-α) inhibitors, B and T cell suppressants, a history of organ transplantation, malignancy (solid or hematologic), human immunodeficiency virus (HIV) infection, primary immunodeficiency, COPD, and diabetes mellitus are recognized risk factors for nocardiosis (4, 5).
According to a report by the U.S. Centers for Disease Control and Prevention (CDC) examining 765 isolates between 1995 and 2004, the most significant pathogenic Nocardia species in humans were identified as Nocardia nova complex, Nocardia brasiliensis, N. farcinica, Nocardia cyriacigeorgica, Nocardia crevicatena, and Nocardia abscessus (6). In another report from Spain examining 1119 isolates between 2005 and 2014, the most significant pathogens were identified as N. cyriacigeorgica, N. nova, N. abscessus, N. farcinica, and Nocardia carnea.
In a study by Başaran et al. (7) involving nine cases, five strains identified at the species level were reported as N. cyriacigeorgica (two strains), N. farcinica, Nocardia asteroides, and N. abscessus. All cases occurred in immunosuppressive patients. However, CLL and ibrutinib use were not mentioned among the risk groups. In another study by Guliyeva et al. (4) involving 16 cases, 13 patients were diagnosed with pulmonary nocardiosis, while the others were diagnosed with cutaneous, bone, or cerebral nocardiosis. Similarly, it did not include patients with CLL or those using ibrutinib. Vanlı et al. (8) reported a case of cerebral Nocardia abscess, likely secondary to pulmonary nocardiosis, in a patient with CLL receiving corticosteroid therapy. The condition was successfully treated with surgical and antimicrobial interventions. However, it was not specified whether ibrutinib was used in the case.
AlHaj Issa et al. (9) reported a case of cerebral nocardiosis in a patient with CLL who was administered ibrutinib. Nocardia species was not isolated, but diagnosis was made histopathologically. Dotson et al. (10) also described a case of disseminated nocardiosis (including skin, lymph nodes, lungs, and brain) and bronchopulmonary aspergillosis coinfection in a patient diagnosed with CLL who was treated with ibrutinib. Nocardia species was not isolated, but diagnosis was made clinically.
There is no clear consensus on the optimal antimicrobial management of nocardiosis. However, combination therapy with trimethoprim/sulfamethoxazole, imipenem, and amikacin has been reported to be superior in the treatment of disseminated nocardiosis (5). In our case, a similar combination was used due to the involvement of both the lungs and the brain.
Conclusion
It is important to recognize that nocardiosis can develop in patients with CLL and those receiving ibrutinib for CLL. This case is significant due to the potential contribution of ibrutinib use to the development of disseminated nocardiosis, with lung and brain involvement. The absence of similar case reports and publications from Türkiye and the potential contribution of this case to the literature further emphasize its importance.