Introduction
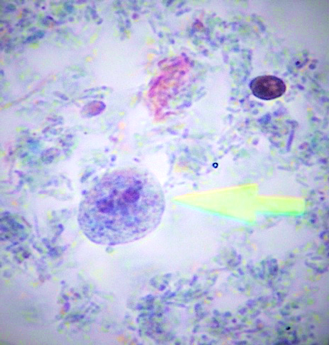
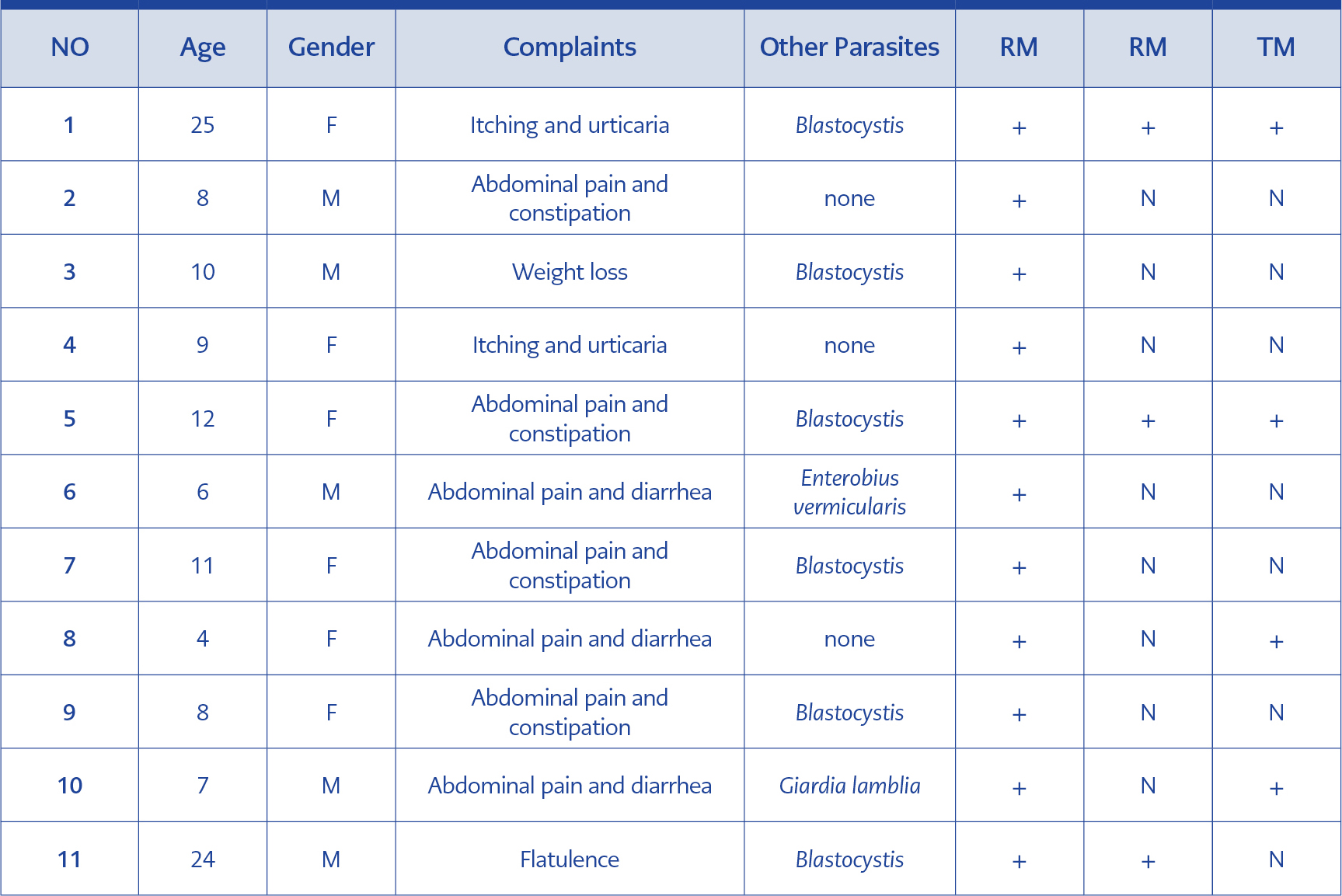
Figure 1. Dientamoeba fragilis trophozoites. (Trichrome stain, x1000; Archive of Manisa Celal Bayar University School of Medicine Department of Medical Parasitology)
Dientamoeba fragilis is an intestinal protozoon classified among the flagellates inhabiting in the caecum and colonic lumen of humans (1, 2). It has long been regarded as a commensal microorganism until when clinical reports indicate its association with gastrointestinal and dermatological case (3-8). Laboratory diagnosis of D. fragilis infections has long been limited to the microscopic examination of permanent stained fecal smears (Figure 1). Since this method has several disadvantages such as discontinuous shedding feature of D. fragilis, its rapid degeneration outside the body and need for expert microscopist for the evaluation of smears, many laboratories could not offer routine testing for D. fragilis. This may be the reason for the lack of concordant prevalence data for D. fragilis for decades (1, 4, 9).
Until the implementation of Polymerase chain reaction (PCR) in parasitology laboratories, culture has long been the most sensitive method to diagnose D. fragilis infection, mostly for research projects (4, 10). In vitro culture techniques to isolate and identify parasitic organisms have long been regarded as valuable tools, not only for research but also for clinical laboratories (11). Many clinical laboratories do not apply culture for routine diagnosis, because the methods for in vitro culture are often complex, their quality control is difficult, and it may take more than a day for a final decision (12). On the other hand, culture methods are relatively cost-effective and highly sensitive in the identification and isolation of certain parasites, and they are also useful in supplying vast numbers of living organisms for serological, immunological and drug trial studies (12, 13).
D. fragilis culture is maintained in the media also used for the cultivation of Entamoeba spp. (10, 13). According to Dobell, D. fragilis is one of the easiest of the human intestinal amoebae to isolate and grow in vitro (14), thus, it has already been cultivated in several media, such as Dobell’s, Boeck-Drbohlav’s, Robinson’s and Talis’s medium (TM), as well as Loeffler’s slope medium (4, 6, 10, 15, 16). A common feature of these media is being diphasic, including a solid and a liquid phase. The solid phase is mostly made of serum, agar or egg extracts, while the liquid phase contains a combination of solutions. Rice starch is the common carbohydrate source in these media (10). In addition, all are xenic media, where parasite growth is achieved in an unknown composition of microorganisms (12). Among these, RM has been commonly used in the culture of many intestinal parasites including D. fragilis (8-10). DM is an old medium including a solid phase of inspissated horse serum slant with an overlay of egg white diluted in Ringer’s solution and supplemented with rice starch (10). TM is also a diphasic egg medium developed primarily to obtain Entamoeba histolytica (15). The trials revealed that his formula might supply D. fragilis and Blastocystis spp. in huge numbers as well.
This study aims to compare the efficacies of three different culture media, RM, DM and TM, in the identification of D. fragilis in stool samples, in terms of both sensitivity and parasite load.
Materials and Methods
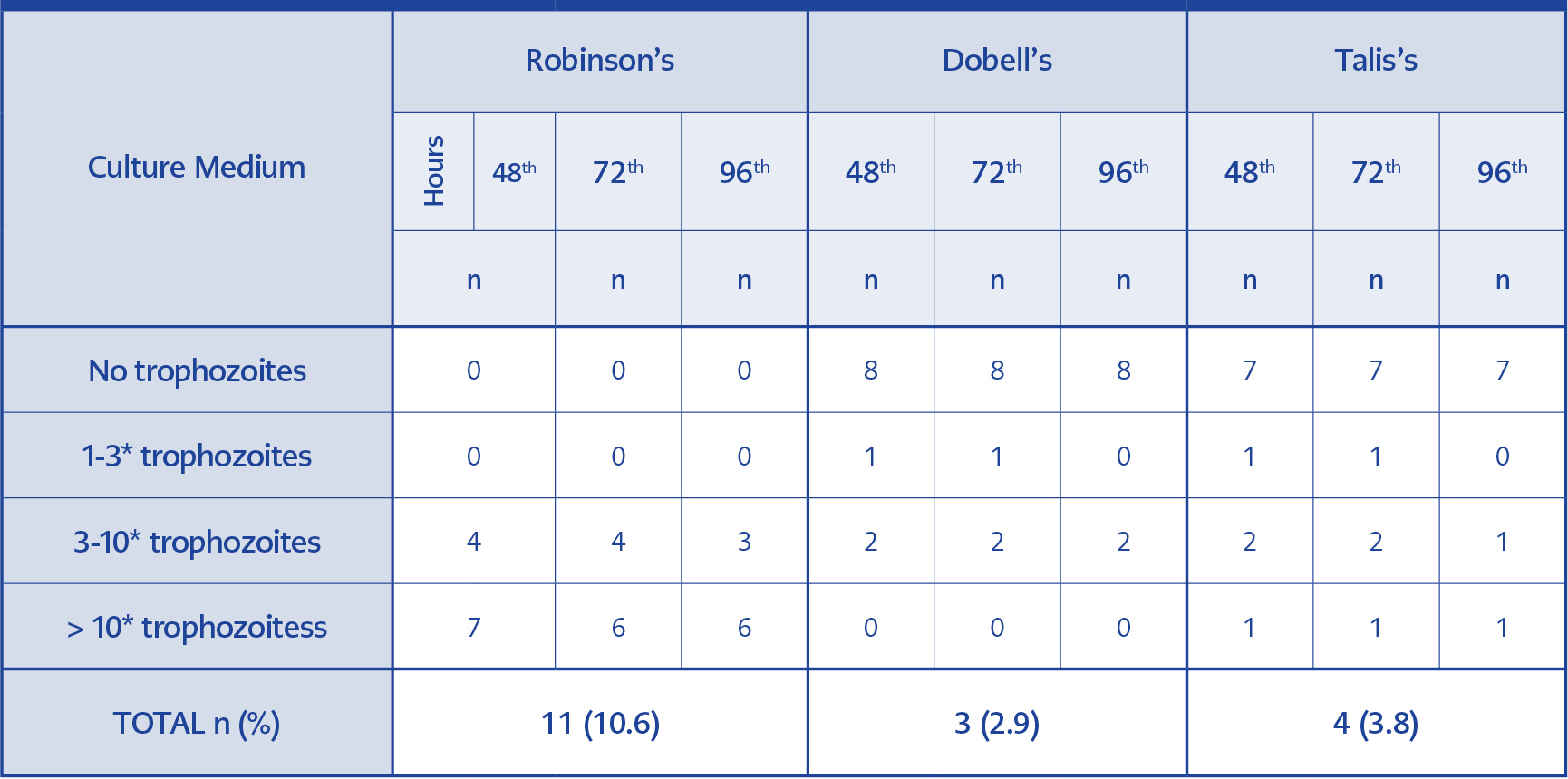
A total of 104 fresh stool samples submitted to Parasitology Laboratory of Manisa Celal Bayar University Hospital and found to be negative for D. fragilis trophozoites after the examination of trichrome-stained smears were enrolled in the study. Their fresh stool samples were inoculated in the culture tubes of RM, DM, and TM. RM and TM were incubated at 37°C, while DM was incubated at 41°C, as suggested previously (14). Robinson‘s medium was prepared and applied as described (10). The contents, preparation, and application of DM and TM were described in Table 1 and Table 2, respectively. Recovery of D. fragilis trophozoites was checked under the light microscope at x400 magnification, with a drop of culture sediment at 48, 72 and 96 hours after inoculation. Samples with no recovery at 96th hours were considered negative and subcultured once again to avoid false-negative samples, as suggested (17). The subcultures were also examined for reproduction at 48, 72 and 96 hours after inoculation. Trichrome-stained smears of the cultures were then examined for confirmation of the positives.
The number of D. fragilis trophozoites, in each examination field, were classified under x400 magnification as follows: (+) one to three trophozoites; (++) three to ten trophozoites; (+++) more than ten trophozoites. The sensitivity and specificity values of Dobell’s medium and TDEM were assessed compared to Robinson’s medium.
Manisa Celal Bayar University, School of Medicine Ethics Committee approved the study (Registration number: 2003-49). Written informed consent of the participants was obtained following a detailed explanation of the study protocol.
Statistical analysis
Chi-squared tests (with Yates’ correction) and Fisher’s exact test were employed for comparison of categorical variables. The Sensitivity and specificity of TM and DM were calculated with comparison to RM. The data analyses were done using Statistical Package for Social Sciences version 10.0® (SPSS Inc.; Chicago, IL, USA), and statistical significance was set as <0.05.
Results
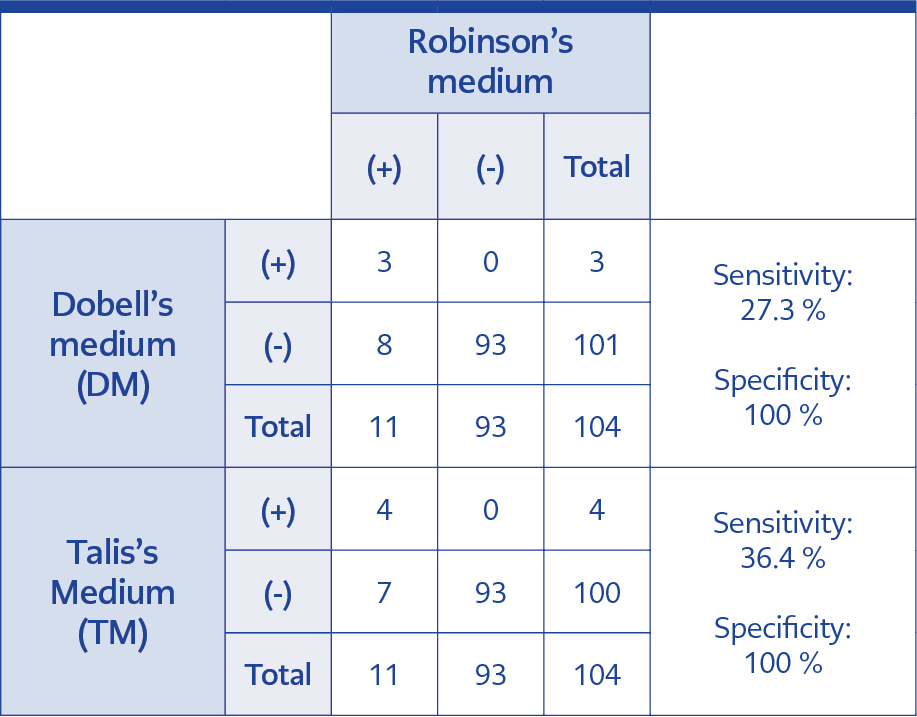
Table 3. The assessment of the efficacies of Dobell’s medium and Talis’s medium, compared to Robinson’s medium.
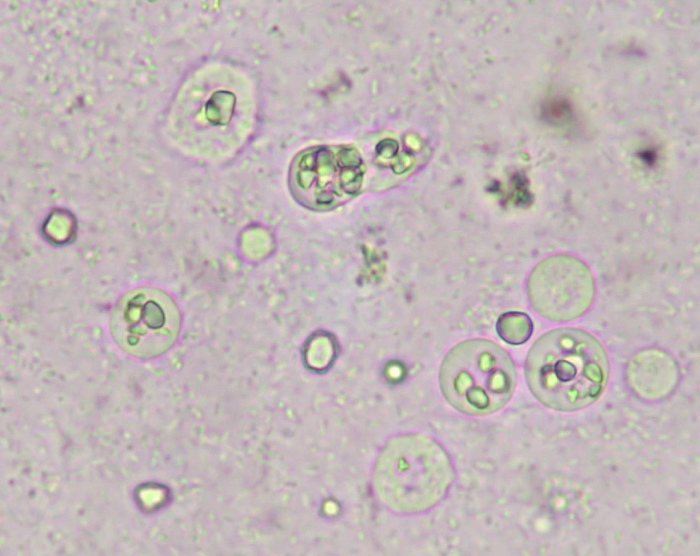
Figure 2. Dientamoeba fragilis trophozoites in culture. Note that D. fragilis trophozoites ingested rice starch while Blastocystis spp. not. Confirmation with microscopic examination of trichrome-stained smears are required (x400; Archive of Manisa Celal Bayar University School of Medicine Department of Medical Parasitology)
The mean age of the participants age of the participants was 22.2 (±6.0; age range 3-79), and there were more females (n=55; 52.9%) in the study group. Three of every four participants (n=78; 75.0%) were referred to parasitology laboratory from Gastroenterology department of internal medicine and paediatrics, followed by dermatology (n=16; 15.4%) and allergy (10, 9.6%) departments. One-third of the participants (n=34; 32.7%) described no symptoms, while abdominal pain and diarrhea were the most common symptoms followed by itching and/or urticaria (19 and 17 individuals, respectively) (Table 3).
Assessment of the study group showed trophozoites of D. fragilis were detected in 11 of 104 (10.6%) stool samples. All 11 samples were successfully cultivated in RM, while 3 and 4 of these 11 samples were positive in DM and TM, respectively (Table 4). Two samples were cultivated in all media. Trophozoites of D. fragilis were identified as round, rice-starch containing organisms, developing leaf-like pseudopodia after 10 minutes at room temperature (Figure 2). All positive samples were confirmed with microscopic examination of their trichrome-stained smears. More parasites were identified in RM tubes, even after 96 hours (Table 5). The sensitivity and values of both DM and TM compared to RM were shown in Table 4.
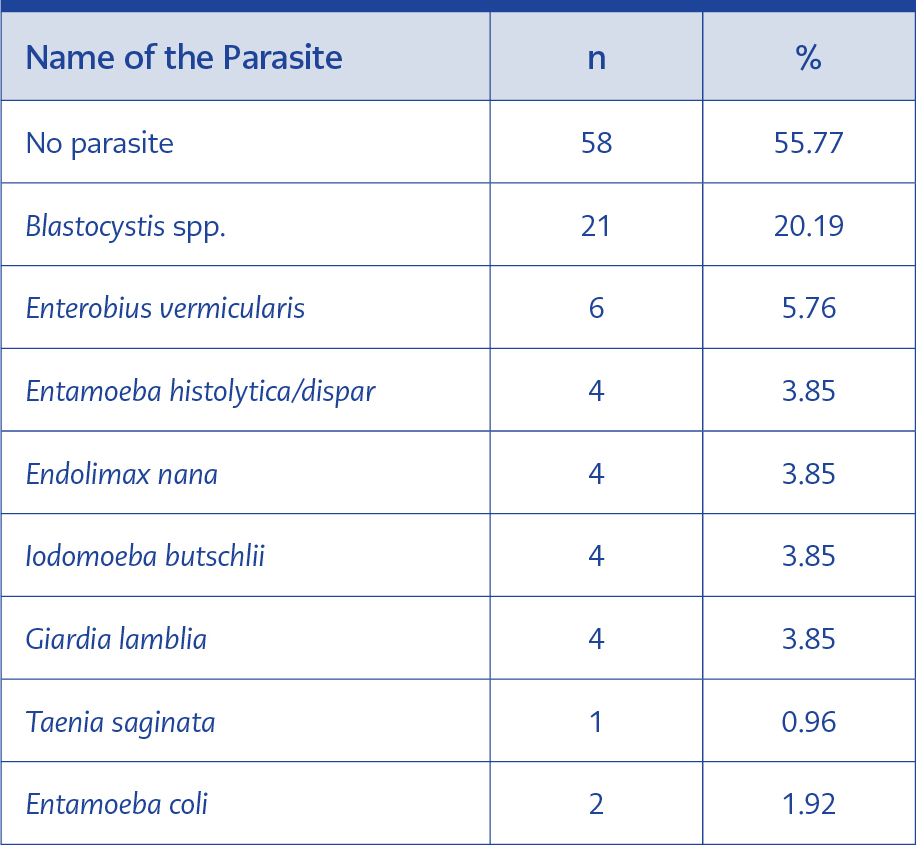
More than half of the stool samples had no parasites detected during the routine examination (n= 58; 55.8%). The leading parasite was Blastocystis spp. (n=21; 20.2%), followed by Enterobius vermicularis (n=6; 5.8%). Among the culture-positive samples, 6 of 11 RM, 2 of 4 TM and all 3 DM-positive samples had Blastocystis spp. (Table 5).
Discussion
According to some authors, the detection rate of D. fragilis has been improved dramatically by culture (18, 19). In a comparative study of diagnostic methods of protozoal diseases with 1.066 patients, the incidence of D. fragilis was found to be almost 2% and 39.3% by microscopy alone and microscopy with culture, respectively (17). Stool culture was found to be less labour-intensive than trichrome staining plus the isolate can also be used for molecular typing and cryopreservation; it was, therefore, suggested that it should be used to diagnose D. fragilis infection (16, 18, 19). Using microscopy alone, the detection rate of D. fragilis was found to be 3.3% in the Parasitology Laboratory of Manisa Celal Bayar University in 2003; this figure rose to 5.2% after the addition of culture even just to individual patients (unpublished data).
Despite regarded as a sensitive and specific method, culture was not common in routine diagnosis of intestinal parasites due to its being labour-intensive and time-consuming (4, 10, 17). After PCR protocols were launched to detect D. fragilis, it has been noticed that old methods have dramatically underestimated its prevalence; for example, in a Danish study involving PCR examination of 22,000 fecal samples of individuals in a period of three years, D. fragilis prevalence was found to be 43% in general population, while it was reaching 71% in children (20). However, PCR is still an expensive method for many laboratories as it requires a high-standard molecular laboratory, expensive equipment and regular supply of consumable products. The costs of conventional PCR and Real-Time PCR were calculated as 15 USD and 26.5 USD, while the cost of microscopy was 3.5 USD per stool sample (6). In our experience, the long-term costs of culture methods were almost the same compared to microscopic examinations of permanent stained smears and significantly cheaper than PCR. Therefore, culture is a reasonable option for laboratories with limited financial resources.
Successful cultivation of D. fragilis has already been managed in several xenic media, including RM, DM, TM, Boeck-Drbohlav’s medium and Loeffler’s slope medium (4, 10, 15, 16). Cultivation in axenic medium, where the microbiological content of the medium is predetermined, is essential for all kinds of studies, especially for the ones targeting the virulence and antimicrobial efficacy testing; however, cultivation of D. fragilis in an axenic culture medium has not been maintained yet, and this is a significant drawback for antimicrobial efficacy trials and studies on its pathogenicity (4, 21).
The xenic medium developed by Robinson in 1968 has been the most preferred medium for the cultivation of E. histolytica, as well as the other species of Entamoeba, Endolimax nana, Blastocystis spp. and D. fragilis (10; 13). It is labour-intensive and requires various chemicals that are used to prepare fresh solutions. In some D. fragilis-positive cultures, excessive reproduction of Blastocystis spp. were also noticed in RM, which were harmful to the maintenance of D. fragilis trophozoites (10). It may not be a problem in our study group as more than half D. fragilis-positive samples in RM were Blastocystis-positive as well. Although successful in short-term culture studies, Robinson’s medium seems unsuccessful for long-term culture of D. fragilis (19).
Dobell’s medium has been used successfully for the culture of Entamoeba spp. and D. fragilis (14). An important detail about successful cultivation in DM is that the incubation should be at 41°C, not at 37°C (14). In our experience, preparation of Dobell’s medium was a long and complicated process, and our results yielded its lower sensitivity in the recovery of D. fragilis trophozoites. On the other hand, no excessive reproduction of Blastocystis spp. isolates was noted in DM tubes.
This is probably the first study that used TM to identify D. fragilis in stool samples. It was developed to culture Entamoeba histolytica, but D. fragilis was also cultivated successfully in the trials (15). Preparation of TM was relatively easier compared to RM and DM, but its sensitivity was found to be poorer. Despite the presence of acriflavine in its formula, excessive reproduction of Blastocystis spp. was noted in TM tubes, as in RM.
The detection of protozoa may be obscured due to limited growth in initial culture tubes; therefore at least two passages should be performed in clinically suspected cases of dientamoebiasis. It was reported that the detection rate of culture among patients with known D. fragilis infection was 40% and 80% after the first and second passages, respectively (17). In our study, 2 of the 11 positive samples in Robinson’s medium and none in the other media were diagnosed in the second passages.
Our findings indicate that culture is a useful alternative to PCR for the identification of D. fragilis in stool samples. It takes at least two days to decide whether there is D. fragilis in the stool, which makes it an unfavorable diagnostic method for routine laboratories, while it provides vast numbers of trophozoites, which is essential for research projects involving serological and molecular assessments for D. fragilis. Of the three culture media, Robinson’s medium was the most sensitive and reliable one, although it was more labour-intensive compared to DM and TM. The highest number of trophozoites were recovered even at the 96th hour after inoculation in RM, compared to DM and TM.
In conclusion, culture method is still a cost-effective method that may be preferred in laboratories with limited financial resources where PCR is not available. Considering the obscurities about its pathogenicity, anti-microbial response and genotypic as well as the proteomic profile of D. fragilis, culture is a good option to obtain piles of trophozoites for further assessments.